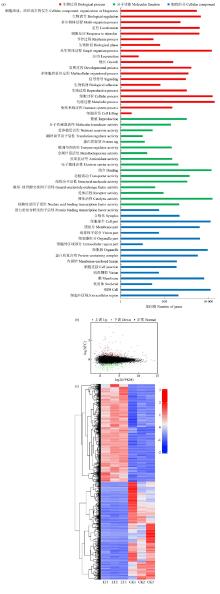
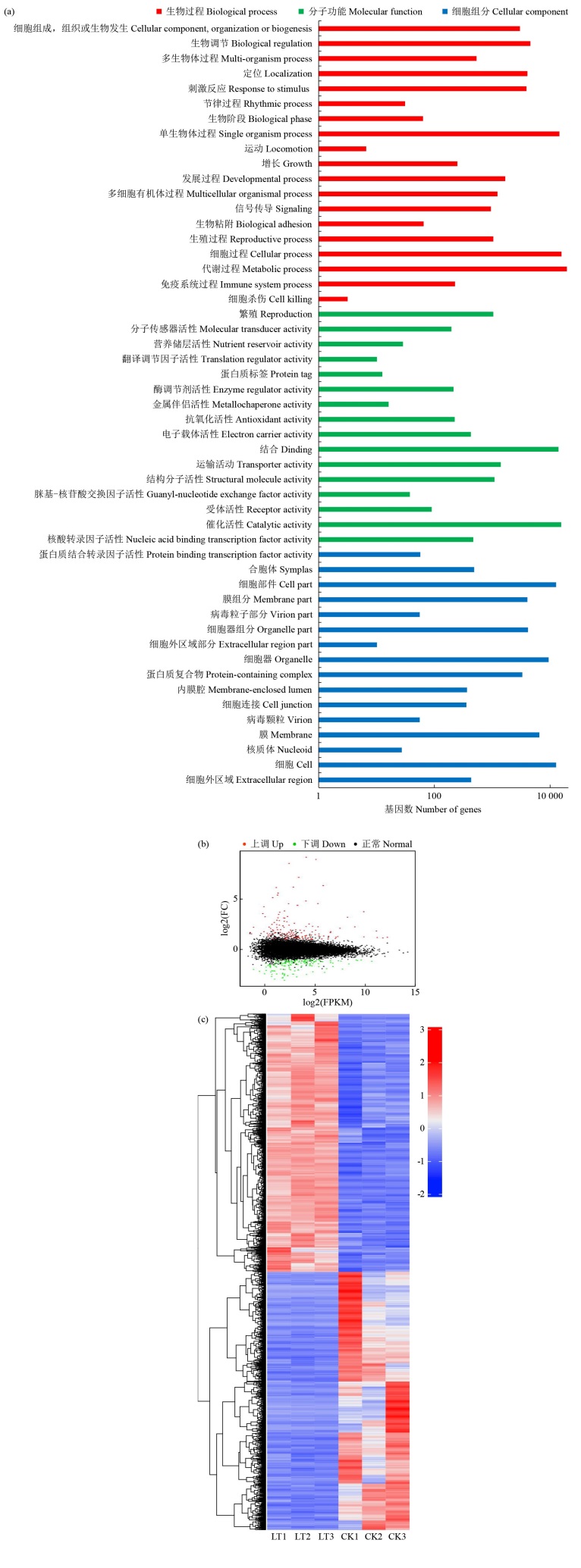
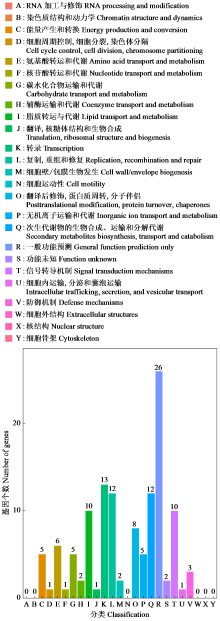
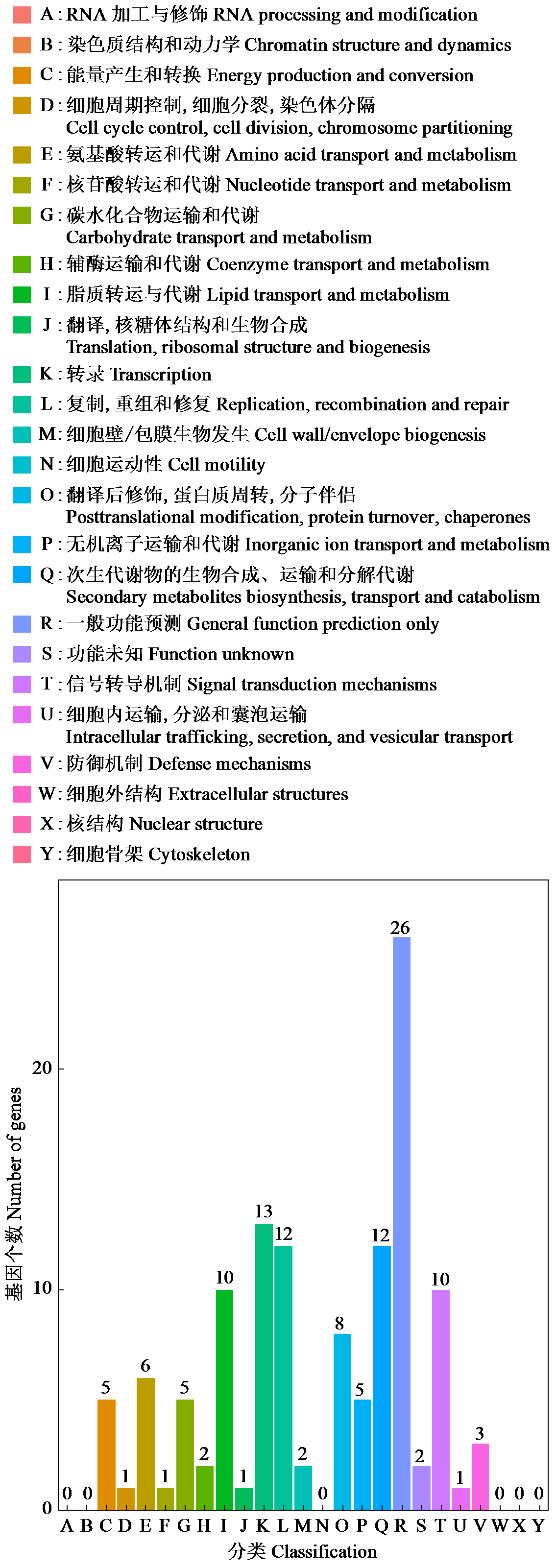
Crops ›› 2025, Vol. 41 ›› Issue (4): 95-103.doi: 10.16035/j.issn.1001-7283.2025.04.012
Previous Articles Next Articles
Analysis of the Transcriptome and Jasmonic Acid Signal Transduction in Chewing Cane Leaves under Cold Stress
Chen Minghui1( ), Xu Yu2, Huang Zhiqiang2, Wang Junqing1, Zhang Baoqing3(
), Xu Yu2, Huang Zhiqiang2, Wang Junqing1, Zhang Baoqing3( )
)
- 1School of Chemistry and Environmental Engineering, Pingdingshan University / Henan Key Laboratory of Germplasm Innovation and Utilization of Eco-Economic Woody Plant, Pingdingshan 467000, Henan, China
2Pingdingshan Forestry Bureau, Pingdingshan 467000, Henan, China
3Guangxi Key Laboratory of Sugarcane Genetic Improvement / Sugarcane Research Institute of Guangxi Academy of Agricultural Sciences / Key Laboratory of Sugarcane Biotechnology and Genetic Improvement (Guangxi), Ministry of Agriculture and Rural Affairs, Nanning 530007, Guangxi, China
| [1] | Shi Y T, Ding Y L, Yang S H. Cold signal transduction and its interplay with phytohormones during cold acclimation. Plant & Cell Physiology, 2015, 56(1):7-15. |
| [2] | 乌凤章, 王贺新, 徐国辉, 等. 木本植物低温胁迫生理及分子机制研究进展. 林业科学, 2015, 51(7):116-128. |
| [3] | Chinnusamy V, Zhu J, Zhu J K. Cold stress regulation of gene expression in plants. Trends Plant Science, 2007, 12(10):444-451. |
| [4] |
张赞培, 谷月营, 尚旭岚, 等. 自然低温下23个青钱柳家系耐寒性初步评价. 南京林业大学学报(自然科学版), 2024, 48(4):85-92.
doi: 10.12302/j.issn.1000-2006.202208024 |
| [5] | 李丽, 达晓伟, 张悦婧, 等. 逆境胁迫对拟南芥细胞质、叶绿体和细胞外ATP水平的影响. 兰州大学学报(自然科学版), 2023, 59(6):846-852. |
| [6] | 罗健东, 邱梦青, 周慧敏, 等. 菠萝AcZFP1的克隆及其在低温胁迫下的功能分析. 园艺学报, 2024, 51(3):495-508. |
| [7] | 罗玉霞, 许涛, 戴倩, 等. 过表达F-box基因BoFBX117对甘蓝低温耐受性的影响. 农业生物技术学报, 2024, 32(1):70-79. |
| [8] |
Lyons R, Manners J M, Kazan K. Jasmonate biosynthesis and signaling in monocots: a comparative overview. Plant Cell Reports, 2013, 32(6):815-827.
doi: 10.1007/s00299-013-1400-y pmid: 23455708 |
| [9] |
Weber H. Fatty acid-derived signals in plants. Trends in Plant Science, 2002, 7(5):217-224.
doi: 10.1016/s1360-1385(02)02250-1 pmid: 11992827 |
| [10] |
Chini A, Monte I, Zamarreno A M, et al. An OPR3-independent pathway uses 4,5-didehydrojasmonate for jasmonate synthesis. Nature Chemical Biology, 2018, 14(2):171-178.
doi: 10.1038/nchembio.2540 pmid: 29291349 |
| [11] | Ruan J J, Zhou Y X, Zhou M L, et al. Jasmonic acid signaling pathway in plants. International Journal of Molecular Sciences, 2019, 20(10):2479-2479. |
| [12] |
Wasternack C. Jasmonates: an update on biosynthesis, signal transduction and action in plant stress response, growth and development. Annals of Botany, 2007, 100(4):681-697.
doi: 10.1093/aob/mcm079 pmid: 17513307 |
| [13] | Du S G, Cui M, Cai Y L, et al. Metabolomic analysis of chilling response in rice (Oryza sativa L.) seedlings by extractive electrospray ionization mass spectrometry. Environmental and Experimental Botany, 2020, 180:104231. |
| [14] | Chang J J, Guo Y L, Zang Z X, et al. CBF-responsive pathway and phytohormones are involved in melatonin-improved photosynthesis and redox homeostasis under aerial cold stress in watermelon. Acta Physiologiae Plantarum, 2020, 42(10):109205-109242. |
| [15] | Pan X X, Wu H, Hu M Y, et al. Global analysis of gene expression profiles in glutinous rice 89-1 (Oryza sativa L.) seedlings exposed to chilling stress. Plant Molecular Biology Reporter, 2021, 39(3):1-14. |
| [16] | Li L J, Ma H Y, Lu X C, et al. TMT-Based quantitative proteomic revealed metabolic changes of Jasmonic acid in regulating the response of Malus baccata (L.) Borkh. roots to low root-zone temperature. Journal of Plant Growth Regulation, 2021, 41(2):1-16. |
| [17] | Hu Y R, Jiang L Q, Wang F, et al. Jasmonate regulates the inducer of cbf expression C-repeat binding factor/DRE binding factor 1 cascade and freezing tolerance in Arabidopsis. Plant Cell, 2013, 25(8):2907-2924. |
| [18] | 陈明辉, 张志录, 程世平, 等. 低温胁迫对不同品种果蔗膜脂过氧化及渗透调节物质的影响. 作物杂志, 2016(6):53-57. |
| [19] | 陈明辉, 程世平, 张志录, 等. 不同品种果蔗幼苗对低温的生理响应及耐寒性评价. 华南农业大学学报, 2018, 39(2):40-46. |
| [20] | 陈明辉, 张志录, 程世平, 等. 低温胁迫对4个果蔗品种幼苗根系活力和保护酶活性的影响. 江苏农业科学, 2018, 46(3):116-119. |
| [21] | 陈明辉, 程世平, 张志录, 等. 低温胁迫下不同果蔗品种光合及荧光特性的变化及耐寒性评价. 热带作物学报, 2018, 39(3):465-471. |
| [22] | Love M I, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biology, 2014, 15(12):1-21. |
| [23] | Kenneth J L, Thomas D S. Analysis of relative gene expression data using real-time quantitative PCR and the 2-∆∆CT method. Methods, 2001, 25(4),402-408. |
| [24] |
Varshney R K, May G D. Next-generation sequencing technologies: opportunities and obligations in plant genomics. Briefings in Functional Genomics, 2012, 11(1):1-2.
doi: 10.1093/bfgp/els001 pmid: 22345600 |
| [25] | 朱鹏锦, 宋奇琦, 谭秦亮, 等. 3个甘蔗品种响应低温胁迫的转录组分析. 广西科学, 2023, 30(2):267-276. |
| [26] | 唐仕云, 杨丽涛, 李杨瑞, 等. 低温胁迫处理对甘蔗转录因子表达的影响. 分子植物育种, 2020, 18(7):2152-2160. |
| [27] | 杨玉婷. 甘蔗响应低温胁迫的转录组分析与冷响应基因挖掘. 福州: 福建农林大学, 2022. |
| [28] |
唐仕云, 杨丽涛, 李杨瑞. 低温胁迫下不同甘蔗品种的转录组比较分析. 生物技术通报, 2018, 34(12):116-124.
doi: 10.13560/j.cnki.biotech.bull.1985.2018-0522 |
| [29] | Yang H L, Guo Z J, Jiang L, et al. How the wild sugarcane resource Miscanthus floridulus responds to low-temperature stress: a physiological and transcriptomic analysis. Sugar Tech, 2022, 25(2):398-409. |
| [30] | Dharshini S, Hoang N V, Mahadevaiah C, et al. Root transcriptome analysis of Saccharum spontaneum uncovers key genes and pathways in response to low-temperature stress. Environmental and Experimental Botany, 2020, 171:103935-103935. |
| [31] | Park J W, Benatti T R, Marconi T, et al. Cold responsive gene expression profiling of sugarcane and Saccharum spontaneum with functional analysis of a cold inducible saccharum homolog of NOD26-like intrinsic protein to salt and water stress. PLoS ONE, 2015, 10(5):e0125810. |
| [32] | 郭晋敏, 杨升, 刘星, 等. 秋茄低温胁迫转录组分析及脱落酸信号途径基因挖掘. 林业科学研究, 2023, 36(2):39-49. |
| [33] |
Yang H L, Wang T J, Yu X H, et al. Enhanced sugar accumulation and regulated plant hormone signalling genes contribute to cold tolerance in hypoploid Saccharum spontaneum. BMC Genomics, 2020, 21(1):507.
doi: 10.1186/s12864-020-06917-z pmid: 32698760 |
| [34] |
Liu W H, Wang H Y, Chen Y P, et al. Cold stress improves the production of artemisinin depending on the increase in endogenous jasmonate. Biotechnology and Applied Biochemistry, 2017, 64(3):305-314.
doi: 10.1002/bab.1493 pmid: 26988377 |
| [35] | Ming R H, Zhang Y, Wang Y, et al. The JA-responsive MYC2-BADH-like transcriptional regulatory module in Poncirus trifoliata contributes to cold tolerance by modulation of glycine betaine biosynthesis. New Phytologist, 2020, 229(5):2730-2750. |
| [36] | Zhao M L, Wang J N, Shan W, et al. Induction of jasmonate signalling regulators MaMYC2s and their physical interactions with MaICE 1 in methyl jasmonate-induced chilling tolerance in banana fruit. Plant Cell and Environment, 2013, 36(1):30-51. |
| [37] |
Wasternack C, Hause B. Jasmonates:biosynthesis, perception, signal transduction and action in plant stress response, growth and development. Annals of Botany, 2013, 111(6):1021-1058.
doi: 10.1093/aob/mct067 pmid: 23558912 |
| [1] | Dou Weize, Jiang Wen, Feng Yichuan, Jin Zhuo, Quan Xueli, Wu Songquan. Transcriptome Analysis Reveals Mechanisms of Calycosin- 7-O-β-D-Glucoside Accumulation in Astragalus membranaceus Adventitious Roots through Hydrogen Peroxide [J]. Crops, 2024, 40(1): 48-56. |
| [2] | Wu Qihua, Chen Diwen, Zhou Wenling, Ao Junhua, Huang Ying, Huang Zhenrui, Li Shuang, Sun Donglei. Effects of Reducing Phosphorus Application in High-P Soils on the P Efficiency of Chewing Cane and Soil Enzyme Activity [J]. Crops, 2021, 37(3): 91-98. |
| [3] | Song Li,Yibo He,Manman Lu,Hongjian Liu,Junxian Liu,Limin Liu,Kunxing Yu,Xin Liu. Effects of Planting Density on Yield Traits and Commercial Characters of Guiguozhe No.1 [J]. Crops, 2017, 33(4): 78-83. |
| [4] | Minghui Chen,Zhilu Zhang,Shiping Cheng,Xing Huang,Baoqing Zhang. Effects of Low Temperature Stress on Lipid Peroxidation and Osmotic Regulation Substances of Chewing Cane Seedlings [J]. Crops, 2016, 32(6): 53-57. |
|
||