Crops ›› 2025, Vol. 41 ›› Issue (5): 74-85.doi: 10.16035/j.issn.1001-7283.2025.05.011
Previous Articles Next Articles
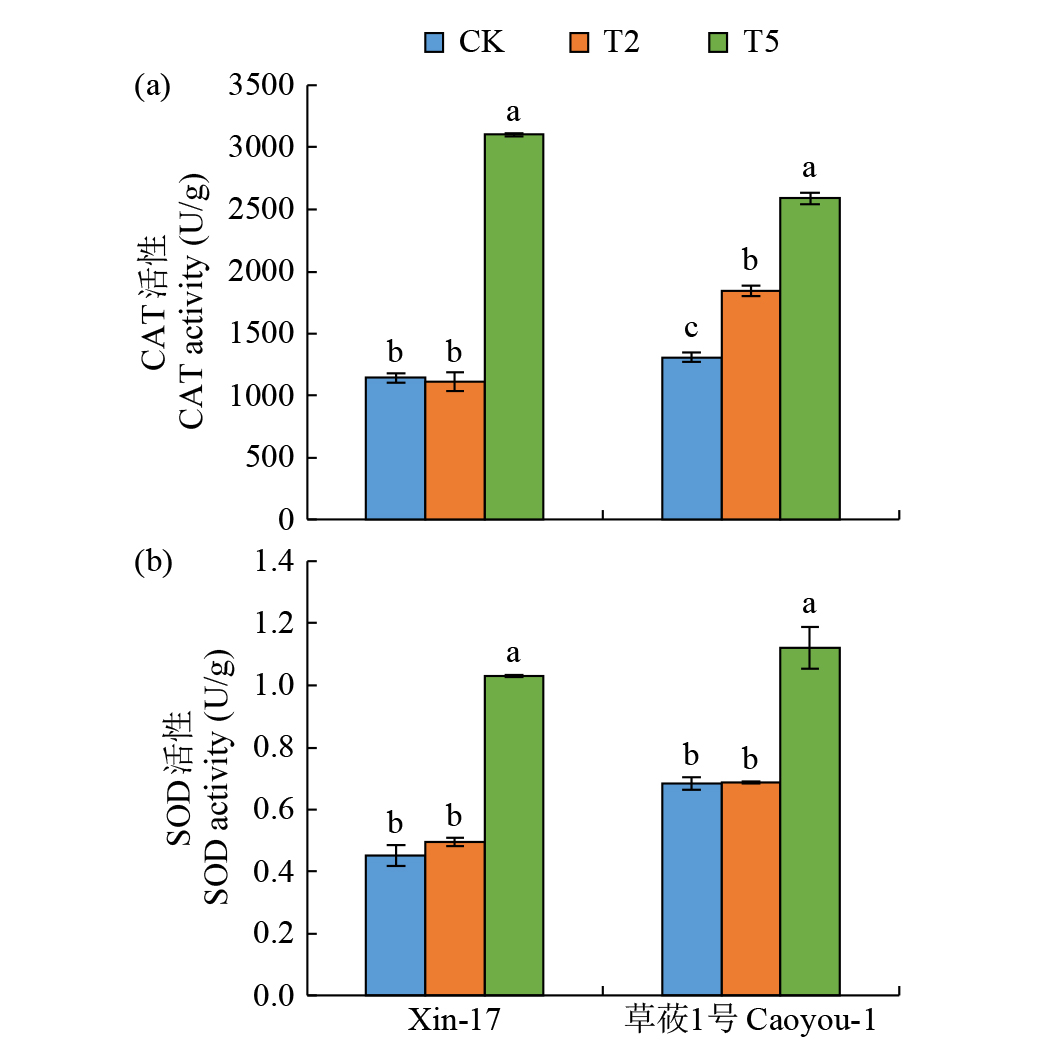
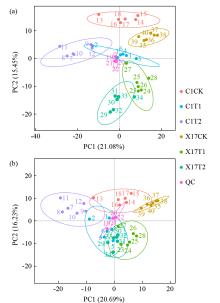
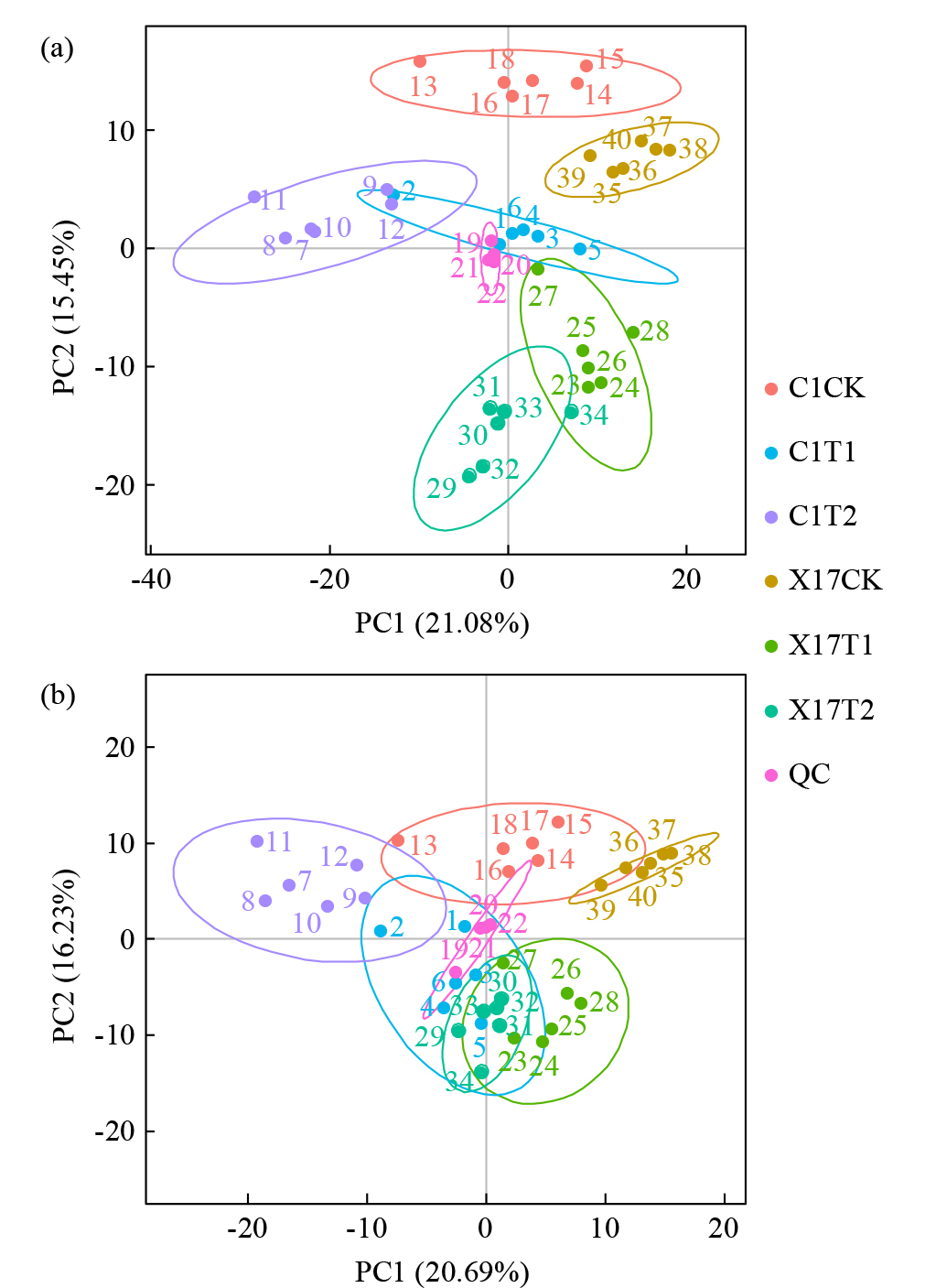
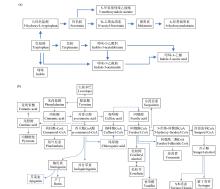
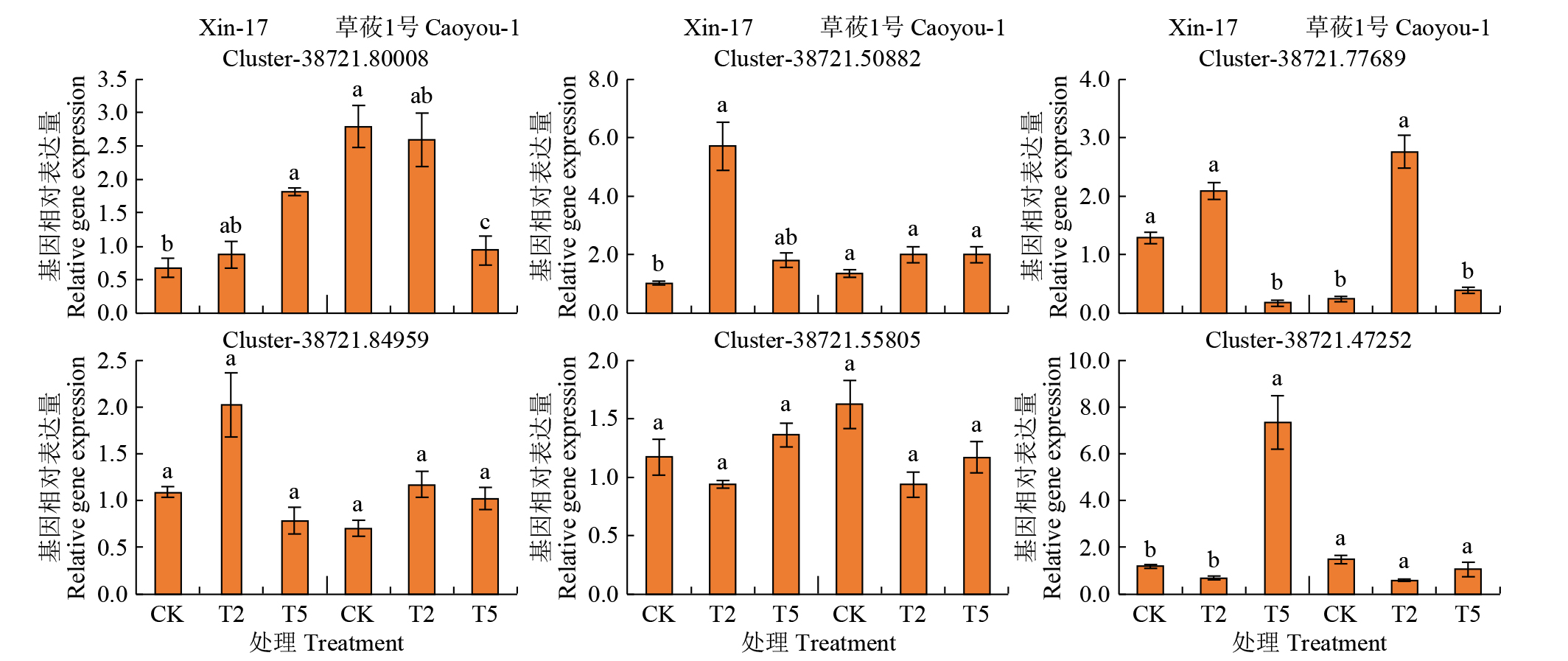
Effects of Compound Saline-Alkali Stress on Growth and Metabolism of Oat
Zhao Zhou( ), Zhang Li, Gao Xinlei, Qiu Hongyu
), Zhang Li, Gao Xinlei, Qiu Hongyu
College of Agriculture ,Chifeng University, Chifeng 024000 Inner Mongolia, China
| [1] | Food and Agriculture Organization of the United Nations.The state of food and agriculture 2021. (2021-12-12)[2024-08-26].https://www.fao.org/3/cb4476en/cb4476en.pdf. |
| [2] | Han M F, et al. Soil salinization research in China: advances and prospects. Journal of Geographical Sciences, 2014, 24(5):943-960. |
| [3] | Chhabra R. Classification of salt-affected soils. Arid Land Research and Management, 2004, 19(1):61-79. |
| [4] | Qadir M, Oster J D, Schubert S, et al. Phytoremediation of sodic and saline-sodic soils. Advances in Agronomy, 2007, 96(7):197-247. |
| [5] |
Flowers T J, Munns R, Colmer T D. Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Annals of Botany, 2015, 115(3):419-431.
doi: 10.1093/aob/mcu217 pmid: 25466549 |
| [6] |
Munns R, Tester M. Mechanisms of salinity tolerance. Annual Review of Plant Biology, 2008, 59:651-681.
doi: 10.1146/annurev.arplant.59.032607.092911 pmid: 18444910 |
| [7] | Bui E N. Soil salinity: a neglected factor in plant ecology and biogeography. Journal of Arid Environments, 2013, 92:14-25. |
| [8] | Bhattarai S, Biswas D, et al. Morphological, physiological, and genetic responses to salt stress in alfalfa: a review. Agronomy, 2020, 10(4):577. |
| [9] |
Julkowska M M, Testerink C. Tuning plant signaling and growth to survive salt. Trends in Plant Science, 2015, 20(9):586-594.
doi: 10.1016/j.tplants.2015.06.008 pmid: 26205171 |
| [10] | Sanchez D H, Siahpoosh M R, Roessner U, et al. Plant metabolomics reveals conserved and divergent metabolic responses to salinity. Physiologia Plantarum, 2008, 132(2):209-219. |
| [11] | Kim J K, Bamba T, Harada K, et al. Time-course metabolic profiling in Arabidopsis thaliana cell cultures after salt stress treatment. Journal of Experimental Botany, 2007, 58(3):415-424. |
| [12] | Richter J A, Behr J H, Erban A, et al.Ion-dependent metabolic responses of Vicia faba L. to salt stress. Plant Cell and Environment, 2019, 42(1):295-309. |
| [13] | Rengasamy P. Soil processes affecting crop production in salt-affected soils. Functional Plant Biology, 2010, 37:613-620. |
| [14] |
Liu L, Wang B, Liu D, et al. Transcriptomic and metabolomic analyses reveal mechanisms of adaptation to salinity in which carbon and nitrogen metabolism is altered in sugar beet roots. BMC Plant Biology, 2020, 20(1):138.
doi: 10.1186/s12870-020-02349-9 pmid: 32245415 |
| [15] | Pang Q Y, Zhang A Q, Zang W, et al. Integrated proteomics and metabolomics for dissecting the mechanism of global responses to salt and alkali stress in Suaeda corniculata. Plant and Soil, 2016, 402(1/2):379-394. |
| [16] |
De Oliveira D F, Lopes L S, Gomes-Filho E. Metabolic changes associated with differential salt tolerance in sorghum genotypes. Planta, 2020, 252(3):34.
doi: 10.1007/s00425-020-03437-8 pmid: 32761417 |
| [17] | Fang C Y, Luo J. Metabolic GWAS-based dissection of genetic bases underlying the diversity of plant metabolism. Plant Journal, 2019, 97(1):91-100. |
| [18] | Carrera F P, Noceda C, Maridueña-Zavala M G, et al. Metabolomics, a powerful tool for understanding plant abiotic stress. Agronomy, 2021, 11(5):824. |
| [19] | Alawiye T T, Babalola O O. Metabolomics: current application and prospects in crop production. Biologia, 2020, 76(1):227-239. |
| [20] | Guo R, Shi L X, Ding X M, et al. Effects of saline and alkaline stress on germination, seedling growth, and ion balance in wheat. Agronomy Journal, 2010, 102(4):1252-1260. |
| [21] |
Fercha A, Capriotti A L, Caruso G, et al. Shotgun proteomic analysis of soybean embryonic axes during germination under salt stress. Proteomics, 2016, 16(10):1537-1546.
doi: 10.1002/pmic.201500283 pmid: 26969838 |
| [22] | Yan G, Shi Y J, Chen F F, et al. Physiological and metabolic responses of Leymus chinensis seedlings to alkali stress. Plants, 2022, 11(11):1494. |
| [23] | Strychar R, Webster F H, Wood P J.World Oat Production, Trade, and Usage. Minnesota:American Association of Cereal Chemists International, 2011. |
| [24] | Chen O, Mah E, Dioum E, et al. The role of oat nutrients in the immune system: a narrative review. Nutrients, 2021, 13(4):1048. |
| [25] | Han L P, Wang W H, Eneji A E, et al. Phytoremediating coastal saline soils with oats: accumulation and distribution of sodium, potassium, and chloride ions in plant organs. Journal of Cleaner Production, 2015, 90:73-81. |
| [26] |
Gerona M E B, Deocampo M P, Egdane J A, et al. Physiological responses of contrasting rice genotypes to salt stress at reproductive stage. Rice Science, 2019, 26(4):207-219.
doi: 10.1016/j.rsci.2019.05.001 |
| [27] |
Ribba T, Garrido-Vargas F, O'brien J A. Auxin-mediated responses under salt stress: from developmental regulation to biotechnological applications. Journal of Experimental Botany, 2020, 71(13):3843-3853.
doi: 10.1093/jxb/eraa241 pmid: 32433743 |
| [28] | Ryu H, Cho Y. Plant hormones in salt stress tolerance. Journal of Plant Biology, 2015, 58(3):147-155. |
| [29] | Liu J, Zhu T T, et al. The role of melatonin in salt stress responses. International Journal of Molecular Sciences, 2019, 20 (7):1735. |
| [30] | Zhan H S, Nie X J, Zhang T, et al. Melatonin: a small molecule but important for salt stress tolerance in plants. International Journal of Molecular Sciences, 2019, 20(3):709. |
| [31] | Varghese N, Alyammahi O, Nasreddine S, et al. Melatonin positively influences the photosynthetic machinery and antioxidant system of Avena sativa during salinity stress. Plants, 2019, 8 (12):610. |
| [32] | Mannino G, Pernici C, Serio G, et al. Melatonin and phytomelatonin: chemistry, biosynthesis, metabolism, distribution and bioactivity in plants and animals - an overview. International Journal of Molecular Sciences, 2021, 22(18):9996. |
| [33] | 翁文凤, 伍小方, 张凯旋, 等. 过表达FtbZIP5提高苦荞毛状根黄酮积累及其耐盐性. 作物杂志, 2021(4):1-9. |
| [34] | 张旭丽, 王瑞军, 郗小倩, 等. 干旱胁迫及复水对黄芪幼苗生长、生理特性及次生代谢产物积累的影响. 作物杂志, 2024 (5):204-211. |
| [1] | Wu Huijuan, Geng Xiaoli, Li Deming, Zhou Dongchang, Fu Ping, Liu Qian, Du Xiaocun. Effects of Foliar Spraying with Different Iron Fertilizers on Seed Yield and Its Components in Oat [J]. Crops, 2025, 41(5): 233-238. |
| [2] | Wang Liang, Wang Rui, Zhu Jincheng, Sang Yuwei, Shi Biao, Guo Jiashuai, Jiao Huimin, He Zongling, Shui Yong. Analysis of Salt Tolerance in 50 Peanut Varieties (Lines) at Seedling Stage [J]. Crops, 2025, 41(5): 35-41. |
| [3] | Zhang Jindong, Wang Cheng, Lu Huan, Zeng Lingling, Zhang Gongliang, Sun Haoyue, Liu Yue, Yang Helin, Hou Xiaomin. Response of Mung Bean Genotypes to Exogenous Brassinosteroids under Saline-Alkali Stress [J]. Crops, 2025, 41(5): 54-60. |
| [4] | Chen Ping, Luo Yuanyuan, Wang Juan, Sun Quan, Ma Lingfang, Ma Wenli, Xie Jingbo. Response and Tolerance Evaluation of Oat to Mixed Saline-Alkali Stress at Germination Stage [J]. Crops, 2025, 41(5): 61-66. |
| [5] | Sheng Bin, Lin Zhihao, Wu Zhijian, Zhao Yiming, Ye Xueling, Lü Honghao, Liu Guangyang, Xu Donghui. Research Progress on the Role of Nano-Fertilizers in Horticultural Crop Cultivation [J]. Crops, 2025, 41(5): 93-101. |
| [6] | Zhou Qi, Zhang Jing, Wang Zhenlong, Shi Zhiguo, Deng Chaochao, Chang Hao, Liu Yang, Zhou Yanfang. Effects of Green Manure Incorporation and Nitrogen Fertilizer Reduction on Soil Quality, Oat Yield and Quality in Hexi Irrigation District of Gansu Province [J]. Crops, 2025, 41(4): 188-196. |
| [7] | Chu Peiyu, Han Xicai, Wang Pan, Li Duo, Zhu Hao, Jin Xijun, Song Xinling, Cao Hongxun, Xia Zunmin, Wang Xiaonan. Effects of CaCl2 on Growth and Physiology of Flax Seedlings Under Water Stress [J]. Crops, 2025, 41(4): 206-213. |
| [8] | Tao Zuhao, Wang Weiqin, Zheng Huabin, Xiang Jun, Tang Qiyuan. Effects of Water, Fertilizer and Chemical Regulation on Seedling Quality of Mechanized Casting Transplanting in Late Rice [J]. Crops, 2025, 41(4): 224-230. |
| [9] | Zhang Zhihan, Yao Jie, Zhang Zhantian, Bian Fuhua, Zhang Ziran, Chen Ping, Chen Haining, Liu Baoyou. Effects of 5-Aminolevulinic Acid on Cucumber Seedling Growth and Soil Enzyme Activities under Drought Stress [J]. Crops, 2025, 41(4): 245-250. |
| [10] | Li Jiahao, Jia Yonghong, Lian Shihao, Liu Yue, Yu Shan, Tian Wenqiang, Wang Ziqian, Zhang Jinshan, Shi Shubing. Effects of Prohexadione-Calcium and Phosphorus Application Rate on the Growth, Dry Matter Accumulation, and Yield of Winter Wheat [J]. Crops, 2025, 41(3): 165-171. |
| [11] | Liang Hui, Zhang Jianxin, Xue Lihua, Jia Keke. Effects of Drip Irrigation Amount on Root Growth and Yield of Xinnongdou 2 under the Condition of the Postpone of Water and Nitrogen [J]. Crops, 2025, 41(3): 233-240. |
| [12] | Huang Ming, Fu Xinxin, Zhang Zhenwang, Zhang Jun, Li Youjun. Effects of Seed Sizes on Seed Germination, Seedling Characteristics and Drought Resistance of Dryland Wheat [J]. Crops, 2025, 41(3): 255-262. |
| [13] | Zhao Lingling, Li Guifang, Cheng Chu, Zheng Mingjie, Hu Min, Zhu Jianfeng, Shen Ayi, Shen Aga, Wang Junzhen, Shao Meihong. Preliminary Report on Introduction Experiment of New Buckwheat Varieties in Zhejiang Province [J]. Crops, 2025, 41(2): 86-92. |
| [14] | Lei Yun, Liu Yueyan, Wang Jianjian. Effects of CO2 Enrichment and Phosphorus Level on Seedling Growth and Nutrient Element Absorption of Capsicum anmuum L. [J]. Crops, 2025, 41(2): 189-195. |
| [15] | Zhang Lina, Yang Wenping, Su Miao, Zhang Zhixuan, Li Junhui, Aamir Ali, Chen Jie, Gao Zhiqiang, Yang Zhenping. Effect of Foliar Application of Selenium onNutritional Quality of Oat Grain [J]. Crops, 2025, 41(2): 196-206. |
|
||