Crops ›› 2025, Vol. 41 ›› Issue (5): 155-164.doi: 10.16035/j.issn.1001-7283.2025.05.021
Previous Articles Next Articles
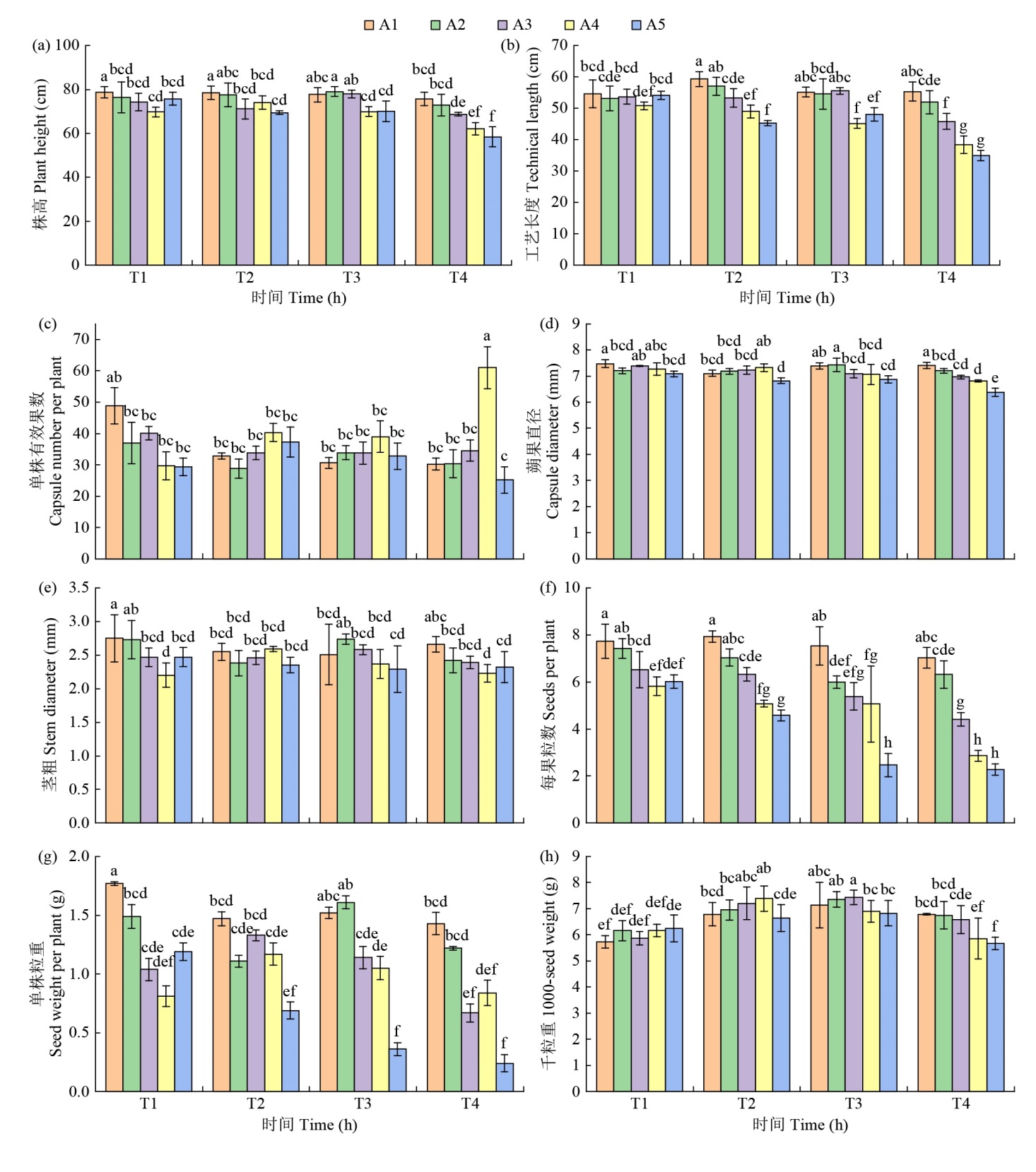
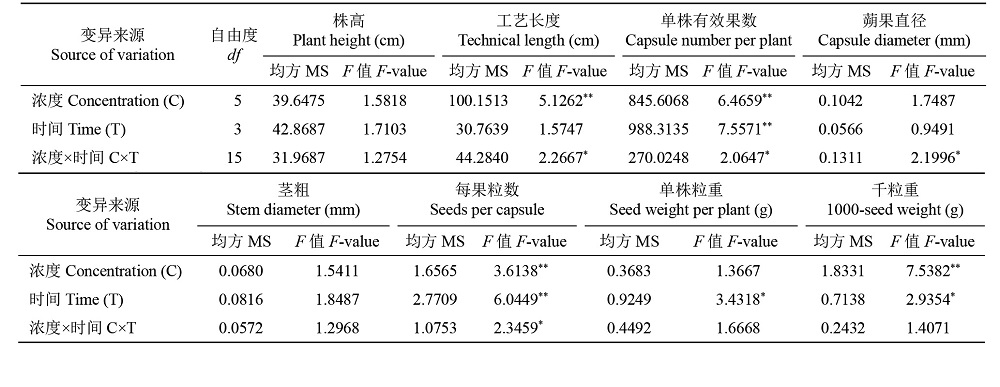
Effects of Chemical Mutagens on Seed Germination and Main Agronomic Traits of M1 Generation of Flax ʻDingya 23ʼ
Li Ying( ), Zhao Yongwei, Chen Ying, Ma Weiming(
), Zhao Yongwei, Chen Ying, Ma Weiming( ), Li Wenzhen, Zhang Haijie
), Li Wenzhen, Zhang Haijie
Dingxi Academy of Agricultural Sciences Dingxi 743000, Gansu, China
| [1] |
谢启光, 徐小冬. 植物生物钟在农业生产中应对全球变暖的应用. 植物学报, 2024, 59(4):635-650.
doi: 10.11983/CBB23136 |
| [2] | 邓欣. 亚麻分子标记的开发及产量相关性状的关联分析. 北京: 中国农业科学院, 2013. |
| [3] | 伊六喜, 斯钦巴特尔, 贾霄云, 等. 胡麻种质资源、育种及遗传研究进展. 中国麻业科学, 2017, 39(2):81-87. |
| [4] | 王斌, 赵利, 侯静静. 化学诱变剂EMS对胡麻种子的诱变效应. 寒旱农业科学, 2022(10):73-77. |
| [5] |
Chantreau M, Grec S, Gutierrez L, et al. PT-Flax (phenotyping and TILLinG of flax): development of a flax (Linum usitatissimum L.) mutant population and TILLinG platform for forward and reverse genetics. BMC Plant Biology, 2013, 13:159.
doi: 10.1186/1471-2229-13-159 pmid: 24128060 |
| [6] | You F M, Cloutier S, Rashid K Y, et al. Flax (Linum usitatissimum L.) genomics and breeding//Advances in plant breeding strategies:industrial and food crops. Berlin: Springer International Publishing, 2019:277-317. |
| [7] | 乔广军. 诱变在我国亚麻育种及资源创新中的利用. 中国麻业, 2006, 28(1):17-20. |
| [8] | 张瑞成, 李魏, 潘素君, 等. 化学诱变在种质资源改良上的应用. 分子植物育种, 2017, 15(12):5189-5196. |
| [9] | 刘翔. EMS诱变技术在植物育种中的研究进展. 激光生物学报, 2014, 23(3):197. |
| [10] | 钮力亚, 于亮, 付晶, 等. 叠氮化钠在农作物育种中的应用. 河北农业科学, 2010, 14(12):52-53,57. |
| [11] | 王芳, 魏章焕, 陆兴苗, 等. 化学诱变甘薯新品种甬紫薯1号的选育. 浙江农业学报, 2015, 27(12):2061-2064. |
| [12] | 陈灿. 化学诱变对水稻诱变后代组织结构及农艺性状和生理特性的影响. 长沙:湖南农业大学, 2010. |
| [13] | 颜志勤. 三种化学诱变剂在杉木育种中的应用研究. 福州:福建农林大学, 2013. |
| [14] | 曹冠男. EMS诱导4种小麦效应研究及突变体的筛选. 杨凌:西北农林科技大学, 2018. |
| [15] | 张晓勤, 薛大伟, 周伟辉, 等. 用甲基磺酸乙酯(EMS)诱变的大麦浙农大3号突变体的筛选和鉴定. 浙江大学学报(农业与生命科学版), 2011, 37(2):169-174. |
| [16] | 杨建胜, 孙万仓, 刘自刚, 等. EMS诱变对北方白菜型冬油菜农艺性状与品质的影响. 干旱地区农业研究, 2018, 36(1):213-220. |
| [17] | Kumari A, Paul S, Sharma V. Genetic diversity analysis using RAPD and ISSR markers revealed discrete genetic makeup in relation to fibre and oil content in Linum usitatissimum L. genotypes. Nucleus, 2018, 61(1):45-53. |
| [18] | Pan G, Chen A G, et al. Genome-wide development of simple sequence repeats database for flax (Linum usitatissimum L.) and its use for genetic diversity assessment. Genetic Resources and Crop Evolution, 2020, 67(4):865-874. |
| [19] | Zhang J P, Wang L M, et al. Genomic comparison and population diversity analysis provide insights into the domestication and improvement of flax. Science, 2020, 23(4):100967. |
| [20] | 刘彩月. 胡麻EMS突变体库的构建及抗ALS类除草剂与高油酸突变体的鉴定分析. 呼和浩特:内蒙古大学, 2023. |
| [21] |
冯学金, 郭秀娟, 杨建春, 等. 诱变技术在亚麻育种中的应用. 核农学报, 2017, 31(7):1310-1316.
doi: 10.11869/j.issn.100-8551.2017.07.1310 |
| [22] | 赵利, 王斌. 60Co-γ射线辐射胡麻种子的诱变效应研究. 中国油料作物学报, 2021, 43(5):834-842. |
| [23] | 曹亚萍, 武银玉, 范绍强, 等. EMS诱变技术在小麦上的应用. 激光生物学报, 2019, 28(5):394. |
| [24] | 赵福永, 郑娇, 何芳, 等. EMS与NaN3对甘蓝型油菜和芥菜型油菜诱变的效果. 江西农业学报, 2010, 22(9):6-9. |
| [25] | 黎诗艳, 阮景军, 范昱, 等. EMS诱变剂处理对苦荞种子萌发及主要农艺性状的影响. 分子植物育种, 2021, 19(3):914-922. |
| [26] | 杜园园, 刘永忠, 李万星, 等. 大豆EMS化学诱变处理条件分析. 安徽农业科学, 2012, 40(35):16995-16996. |
| [27] | 刘建霞, 张梦丽, 李慧, 等. 苦荞麦种子与幼苗对叠氮化钠诱变的响应. 江苏农业科学, 2019, 47(10):85-88. |
| [28] | 李娟宁, 吕英, 赵桂琴, 等. 化学诱变剂EMS和MNU对燕麦种子萌发和幼苗生长的影响. 草原与草坪, 2021, 41(3):108-118. |
| [29] |
许燕, 谢永平, 郑楚群, 等. EMS诱变花生珍珠红1号创制优良新种质. 核农学报, 2020, 34(7):1369-1377.
doi: 10.11869/j.issn.100-8551.2020.07.1369 |
| [30] |
蔺豆豆, 赵桂琴, 柴继宽, 等. 叠氮化钠诱变燕麦M1代的主要性状分析. 草地学报, 2022, 30(3):587-593.
doi: 10.11733/j.issn.1007-0435.2022.03.010 |
| [31] | 姜振峰, 刘志华, 李文滨, 等. 叠氮化钠对大豆M1的生物学诱变效应. 核农学报, 2006, 20(3):208-210. |
| [32] | 李燕红, 高世庆, 任扬, 等. 小麦核质互作杂交种农艺性状和籽粒性状的杂种优势分析. 麦类作物学报, 2021, 41(10):1228-1237. |
| [33] | 彭波, 徐庆国, 陈灿. 三种化学诱变剂对不同水稻品种的生物学效应研究. 湖南人文科技学院学报, 2007, 24(4):22-26. |
| [1] | Zhu Lingyu, Yang Qiaohui, Liu Yixun, Yuan Jian, Wang Minghua, Xiang Dabing, Zou Liang, Wang Junzhen, Fan Yu. Photosynthetic Characteristics and Agronomic Traits of Wild and Cultivated Oats during Filling Process [J]. Crops, 2026, 42(1): 167-174. |
| [2] | Gao Yanmei, Feng Pengrui, Chen Weiwei, Zhang Meng, Zhang Yongqing. Physiological Response of Quinoa Seedlings with Drought Resistance to Drought Stress [J]. Crops, 2026, 42(1): 182-188. |
| [3] | Yang Wengao, Yuan Wenjue, Li Zhaoguang, He Guiqing, He Qiongji, Wang Rui, Li Yan, Ye Lei, Hou Zhijiang. Effects of Winter Sowing Date on Agronomic Traits and Yield of Quinoa in Northwestern Yunnan, China [J]. Crops, 2026, 42(1): 257-265. |
| [4] | Liu Xiaohan, Tang Yujie, Liu Xinyu, Qiao Zhengyan, Shi Guishan, Yu Miao, Li Yang, Wang Nai, Qi Hongying, Chen Bingru. Analysis of Combining Ability and Heterosis of Main Traits in Core Parents of Sorghum for Spring-Sown Early-Maturing Regions [J]. Crops, 2026, 42(1): 47-53. |
| [5] | Gao Fengyun, Yi Liuxi, Zhou Yu, Siqinbateer , He Ruichao, Jia Xiaoyun. Analysis of Genetic Diversity of Flax Germplasm Resources [J]. Crops, 2026, 42(1): 60-71. |
| [6] | Li Wenjun, Guo Yanping, Yang Shenghua, Shao Yang. Analysis of Genetic Diversity of Main Agronomic Traits of 513 Faba Bean Germplasm Resources [J]. Crops, 2026, 42(1): 85-93. |
| [7] | Yu Huaxian, An Rudong, Tao Lianan, Lang Rongbin, Bian Xin, Zhang Yu, Liu Xinlong, Liu Jiayong, Zhao Liping, Liu Hongbo, Zhang Gemin, Zhang Baoqing. Comprehensive Evaluation of Reciprocal Cross Hybrids between 57NG208 and Nanjian Chewing Cane Based on Agronomic Traits [J]. Crops, 2026, 42(1): 94-103. |
| [8] | Chen Zhihao, Wang Ting, Chang Xuhong, Wang Yanjie, Liu Xiwei, Yang Yushuang, Wang Yujiao, Wang Demei, Zhao Guangcai. Comprehensive Analysis of Yield and Quality Traits of Wheat Lines in the Northern Huang-Huai Winter Wheat Region [J]. Crops, 2025, 41(6): 148-155. |
| [9] | Qin Nana, Huang Linhua, Chen Ying, Wang Shengmou, Xie Yong, Miao Kai, Li Wanming, Qi Lan. Effects of Foliar Propionyl Brassinolide Application on Photosynthesis, Agronomic Traits and Yield of Summer Soybean [J]. Crops, 2025, 41(6): 164-171. |
| [10] | Yan Xiaowen, Liang Junchao, Zeng Pan, Zhou Hongying, Wang Zhiqi, Le Meiwang, Sun Jian. Effects of Late Sowing on Main Agronomic Traits and Yield of Autumn Sesame [J]. Crops, 2025, 41(6): 189-194. |
| [11] | Yang Shan, Zhang Zhi, Guo Yueqi, Luo Rui, Deng Jianglong, Guo Xiaoyi, Zhou Lihong. Effect of Lees-Based Microbial Inoculants on the Yield of Liquor-Making Sorghum and Soil Physicochemical Properties [J]. Crops, 2025, 41(6): 210-215. |
| [12] | Chen Guoli, Xu Chaofeng, Wei Changmin, Wang Ruyin, Zhang Yanfang, Li Haoyuan, Zhang Jun. Analysis of Genotype and Environmental Interaction Effects of New Silage Corn Varieties in Henan Province [J]. Crops, 2025, 41(6): 91-99. |
| [13] | Zhao Caixia, Baima Yangzhen, Yang Guanghuan, Tang Lin. Identification and Cluster Analysis of Agronomic Traits in Different Types of Rapeseed Resources [J]. Crops, 2025, 41(5): 120-127. |
| [14] | Wu Liguo, Li Xiaohui, Zhao Qing, Chen Xiaolong, Pan Jing, Liu Wangqing, Bai Haibo, Li Qianrong. Genetic Diversity Analysis of Agronomic Traits of Spring Wheat Varieties (Lines) in Ningxia [J]. Crops, 2025, 41(5): 147-154. |
| [15] | Du Hanmei, Tan Lu, Li Shengchun, Wang Qinghai, Xu Zhou, Wu Dandan, Wang Anhu. Cadmium Tolerance of Tartary Buckwheat during Germination Stage and Its Effects on Physiological Characteristics of Seedlings [J]. Crops, 2025, 41(5): 209-220. |
|
||