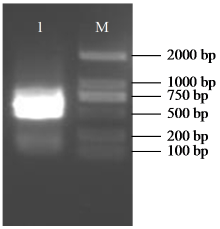
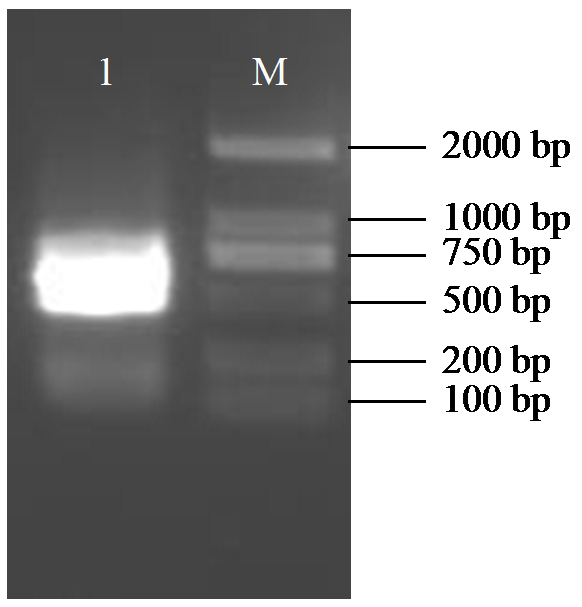
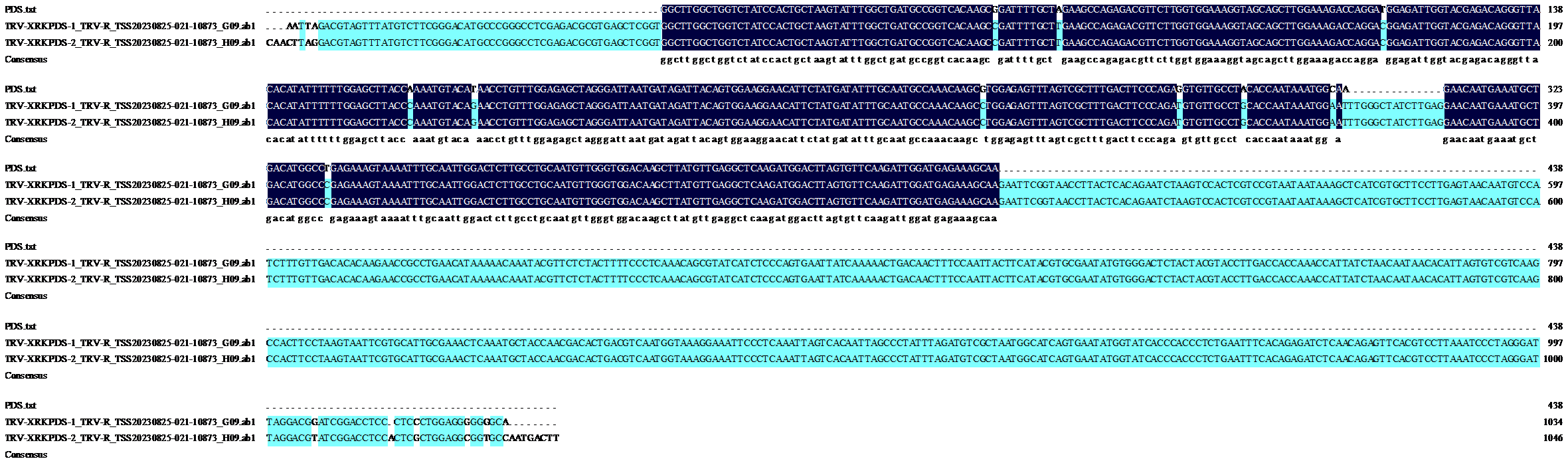
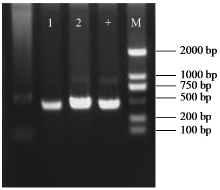
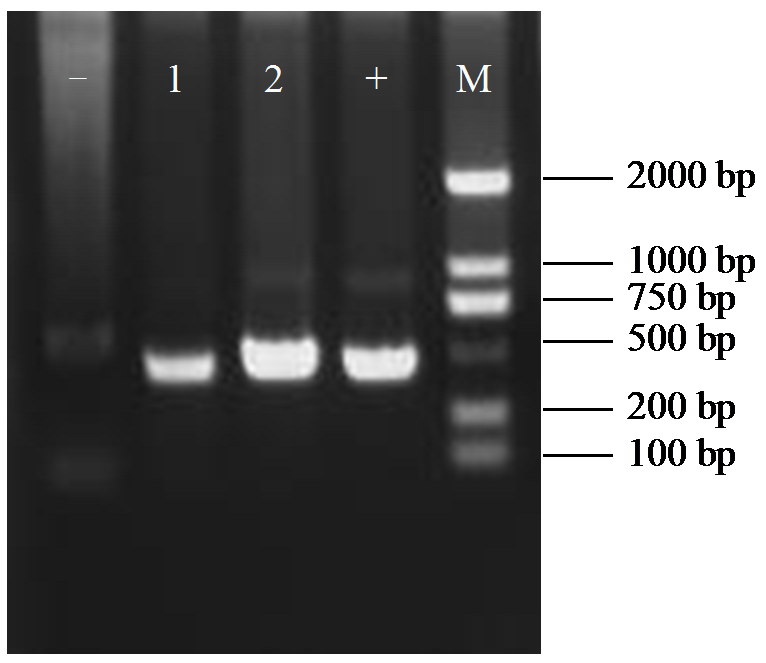
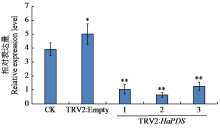
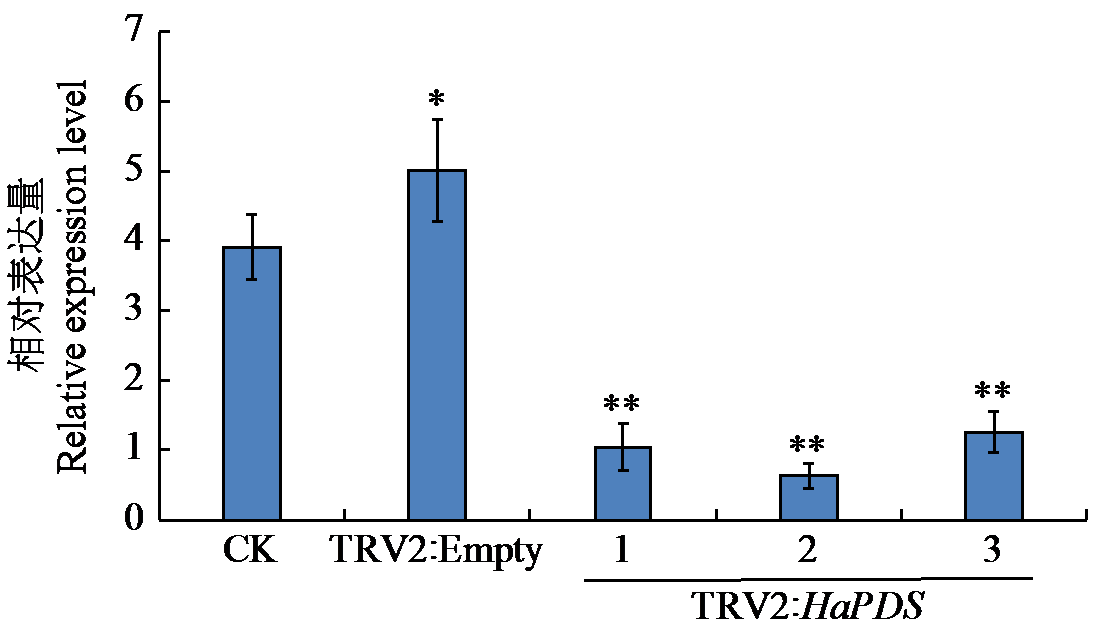
Crops ›› 2025, Vol. 41 ›› Issue (6): 45-50.doi: 10.16035/j.issn.1001-7283.2025.06.006
Previous Articles Next Articles
Construction of a TRV-Mediated VIGS System in Sunflower
Zhou Fei1( ), Huang Xutang1, Xie Pengyuan1, Wang Jing1, Liu Yan1, Cui Jiawei2, Tang Lili1, Wang Wenjun1(
), Huang Xutang1, Xie Pengyuan1, Wang Jing1, Liu Yan1, Cui Jiawei2, Tang Lili1, Wang Wenjun1( )
)
- 1
Industrial Crops Institute ,Heilongjiang Academy of Agricultural Sciences Harbin 150086, Heilongjiang, China
2Institute of Agricultural Science of the 10th Division ,Xinjiang Production and Construction Corps Beitun 836000, Xinjiang, China
| [1] | Htyvusevich P, Navaselsky I, Talkachova Y, et al. Sodium influx and potassium efflux currents in sunflower root cellls under high salinity. Frontiers in Plant Sciences, 2021, 11:613936. |
| [2] |
Nelson G J. Dietary fat, trans fatty acids, and risk of coronary heart disease. Nutrition Reviews, 1998, 56(8):250-252.
pmid: 9735680 |
| [3] | Mohamed S, Boehm R, Schnabl H. Particle bombardment as a strategy for the production of transgenic high oleic sunflower (Helianthus annuus L.). Journal of Applied Botany & Food Quality, 2012, 80(2):171-178. |
| [4] | 潘多, 张嵩玥, 刘芳伊, 等. 病毒诱导的基因沉默技术用于植物色素代谢机制的研究进展. 生物工程学报, 2023, 39(7):579-2599. |
| [5] | 马伟, 王熙豪, 张雪妍. 基于PDS基因家族的海马齿VIGS体系的构建. 农业生物技术学报, 2024, 32(4):949-960. |
| [6] | Bennypaul H S, Mutti J S, Rustgi S, et al. Virus-induced gene silencing (VIGS) of genes expressed in root, leaf, and meiotic tissues of wheat. Functional & Integrative Genomics, 2012, 12 (1):143-156. |
| [7] |
Purkayastha A, Mathur S, Verma V, et al. Virus-induced gene silencing in rice using a vector derivedrus-induced gene silencing in rice using a vector derive from a DNA virus. Planta, 2010, 232(6):1531-1540.
doi: 10.1007/s00425-010-1273-z pmid: 20872012 |
| [8] |
Singh D K, Lee H K, Dweikat I, et al. An efficient and improved method for virus-induced gene silencing in sorghum. BMC Plant Biology, 2018, 18(1):123.
doi: 10.1186/s12870-018-1344-z pmid: 29914379 |
| [9] |
Xu H, Xu L F, Yang P P, et al. Virus-induced phytoene desaturase (PDS) gene silencing using tobacco rattle virus in Lilium×formolongi. Horticultural Plant Journal, 2019, 5(1):31-38.
doi: 10.1016/j.hpj.2018.10.002 |
| [10] | 郭燕, 刘志达, 康立茹, 等. 基于PDS基因的番茄VIGS高效沉默体系的优化. 作物杂志, 2023(2):46-50. |
| [11] |
Deng X B, Elomaa P, Nguyen C X, et al. Virus‐induced gene silencing for Asteraceae—a reverse genetics approach for functional genomics in Gerbera hybrida. Plant Biotechnology Journal, 2012, 10(8):970-978.
doi: 10.1111/pbi.2012.10.issue-8 |
| [12] | 刘俊飞, 杨晓娟, 潘根, 等. 基于TRV病毒的工业大麻(Cannabis sativa L.)VIGS体系的优化. 中国麻业科学, 2023, 45(5):197-205,214. |
| [13] | 王心宇, 吕坤, 蔡彩平, 等. TRV病毒介导的基因沉默体系在棉花中的建立及应用. 作物学报, 2014, 40(8):1356-1363. |
| [14] | Gao X Q, Robert C, Britt J, et al. Agrobacterium-mediated virus-induced gene silencing assay in cotton. Journal of Visualized Experiments, 2011, 54:e2983. |
| [15] |
张景霞, 王芙蓉, 高阳, 等. VIGS技术及其在棉花功能基因组研究中的应用进展. 棉花学报, 2015, 27(5):469-473.
doi: 10.11963/issn.1002-7807.201505011 |
| [16] | Kumagai M H, Donson J, della-Cioppa G, et al. Cytoplasmic inhibition of carotenoid biosynthesis with virus-derived RNA. Proceedings of the National Academy of Sciences of the United States of America, 1995, 92(5):1679-1683. |
| [17] |
Ryu C M, Anand A, Li K, et al. Agrodrench: a novel and effective agroinoculation method for virus-induced gene silencing in roots and diverse solanaceous species. The Plant Journal, 2004, 40 (2):322-331.
doi: 10.1111/tpj.2004.40.issue-2 |
| [18] |
Yan H X, Fu D Q, Zhu B Z, et al. Sprout vacuum-infiltration: a simple and efficient agroinoculation method for virus-induced gene silencing in diverse solanaceous species. Plant Cell Reports, 2012, 31(9):1713-1722.
doi: 10.1007/s00299-012-1285-1 |
| [19] |
付偲僮, 司未佳, 刘颖, 等. TRV介导的小报春基因沉默技术体系的建立. 生物技术通报, 2022, 38(4):295-302.
doi: 10.13560/j.cnki.biotech.bull.1985.2021-0924 |
| [20] |
Deng X B, Bashandy H, Ainasoja M, et al. Functional diversification of duplicated chalcone synthase genes in anthocyanin biosynthesis of Gerbera hybrida. New Phytologist, 2014, 201(4):1469-1483.
doi: 10.1111/nph.2014.201.issue-4 |
| [21] | 唐宜, 李凌飞, 王小菁. 非洲菊花瓣瞬时表达和病毒诱导的基因沉默(VIGS)系统的建立. 植物生理学报, 2017, 53(3):505-512. |
| [22] | 张洪义. 万寿菊(Tagetes erecta)VIGS体系的建立与优化. 武汉:华中农业大学, 2016. |
| [23] | 刘钰婷. 瓜叶菊舌状花花斑形成中关键基因的功能解析. 北京: 北京林业大学, 2020. |
| [24] | 黄书怡. 观赏向日葵花瓣颜色形成的分子机理. 杨凌:西北农林科技大学, 2023. |
| [25] |
Qu J, Ye J, Geng Y F, et al. Dissecting functions of KATANIN and WRINKLED 1 in cotton fiber development by virus-induced gene silencing. Plant Physiology, 2012, 160(2):738-748.
doi: 10.1104/pp.112.198564 |
| [1] | Ma Jun, Zhang Xianglei, An Chengjie, Yang Xuewen, Zhang Bolin, Zhang Wu, Wang Pengdong. Chloroplast Genome Characteristics and Codon Bias Analysis of Anti-Broomrape Confectionery Sunflower Hybrid Jingkui 1408 [J]. Crops, 2025, 41(6): 83-90. |
| [2] | Wang Heya, Luo Jingjing, Meng Ling, Ai Haifeng, Wang Bin, Li Huaisheng, Xu Jingpeng, Xu Xiangyang. Yield Sensitivity Analysis of Edible Sunflower Varieties in Ta'e Basin [J]. Crops, 2025, 41(3): 30-37. |
| [3] | Di Na, Zheng Xiqing, Wang Jing, Han Haijun, Li Na. Study on the Difference of Physiological Response of Sunflower to Broomrape Parasitism [J]. Crops, 2025, 41(2): 123-127. |
| [4] | Mu Jianguo, Wang Peng, Liu Yantao, Cui Jiawei, Chen Yanfang, Wan Sumei, Chen Guihong. Effects of Different Harvesting Periods on the Commerciality and Yield of Edible Sunflower [J]. Crops, 2024, 40(5): 146-151. |
| [5] | Liu Chunhui, Du Ruixia, Wang Yongxing, Yang Qinfang, Shan Feibiao, Xu Fuxun, Zhao Yuxin, Chen Shushu, Liu Wei. Relationship between Meteorological Factors and DUS Test Measurement Traits of Sunflower [J]. Crops, 2024, 40(3): 156-162. |
| [6] | Du Chao, Li Jun, Wang Gang, Wu Xuerui, Ren Zhiyuan, Zhang Junfeng, Bao Haizhu, Wen Aiqing. Effects of High Ridge Mulching Drip Irrigation on the Growth and Water Use of Sunflower in Moderate Saline-Alkali Land in Hetao Irrigation Region [J]. Crops, 2024, 40(1): 111-116. |
| [7] | Lü Zengshuai, Dong Hongye, Wang Peng, Duan Wei, Liu Shengli, Liu Yantao. Progress in Mechanism of Herbicide Resistance and Breeding of Sunflower [J]. Crops, 2024, 40(1): 16-22. |
| [8] | Wu Sheng, Duan Yu, Zhang Tingting, An Hao, Zhang Jun, Liang Junmei, Zhang Sheng. Relationships between Dry Matter Accumulation, Transport and Yield of Confectionary Sunflower and Response to Water and Nitrogen Interactions [J]. Crops, 2023, 39(6): 243-251. |
| [9] | Li Hepeng, Zhang Yunhua, Meng Qinglin, Ma Ligong, Yu Hongtao, Li Haiyan, Li Yichu, Liu Jia, Shi Fengmei, Yang Fan, Liu Liang. Screening of Inducers for Sunflower Sclerotinia sclerotiorum and Application of Hypersensitive Protein [J]. Crops, 2023, 39(6): 257-260. |
| [10] | Ling Yibo, Wang Binjie, Hu Yimin, Heinar·Madithermic mann, Chen Nianlai. Responses of Dry Matter Translocation and Yield Formation to Planting Density and Row Spacing of Sunflower [J]. Crops, 2023, 39(5): 197-203. |
| [11] | Yi Bing, Liu Jingang, Song Dianxiu, Wang Dexing, Zhao Mingzhu, Liu Xiaohong, Sun Enyu, Cui Liangji. Study on Land Productivity and Interspecific Competition of Sunflower and Millet Intercropping in Arid Areas [J]. Crops, 2023, 39(5): 219-223. |
| [12] | Zhu Kongyan, Han Shengcai, Zhao Rong, Wen Yujie, Hu Haochi, Qiao Yimin, Lu Jiafeng, Cao Kai, Xu Zhenghan, Bao Haizhu, Gao Julin. Isolation and Identification of Endophytes from Sunflower Seeds [J]. Crops, 2023, 39(5): 280-284. |
| [13] | Guo Yan, Liu Zhida, Kang Liru, Bao Tingting, Yang Xu, Zhao Jun, Zhang Zhiwei. Optimization of Efficient Silencing System of Tomato VIGS Based on PDS Gene [J]. Crops, 2023, 39(2): 46-50. |
| [14] | Jia Xiuping, Mao Xuhui, Liang Gensheng, Liu Runping, Liu Feng, Wang Xingzhen. Analysis of Physiological and Biochemical Mechanism and Growth and Development Characteristics of Saline and Alkali Resistance in Sunflower [J]. Crops, 2022, 38(5): 146-152. |
| [15] | Shi Bixian, Tao Jianfei, Gao Yan, Xie Huihong, Abulimiti·Aierken , Cheng Pingshan, Maitituersun·Sadike , Sha Hong. Effects of Different Planting Densities on the Morphological Traits and Yields of Three Confectionery Sunflower Varieties [J]. Crops, 2022, 38(5): 195-200. |
|
||