Crops ›› 2023, Vol. 39 ›› Issue (2): 138-144.doi: 10.16035/j.issn.1001-7283.2023.02.020
Previous Articles Next Articles
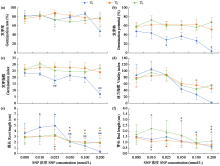
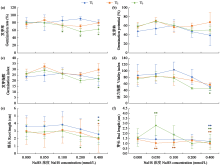
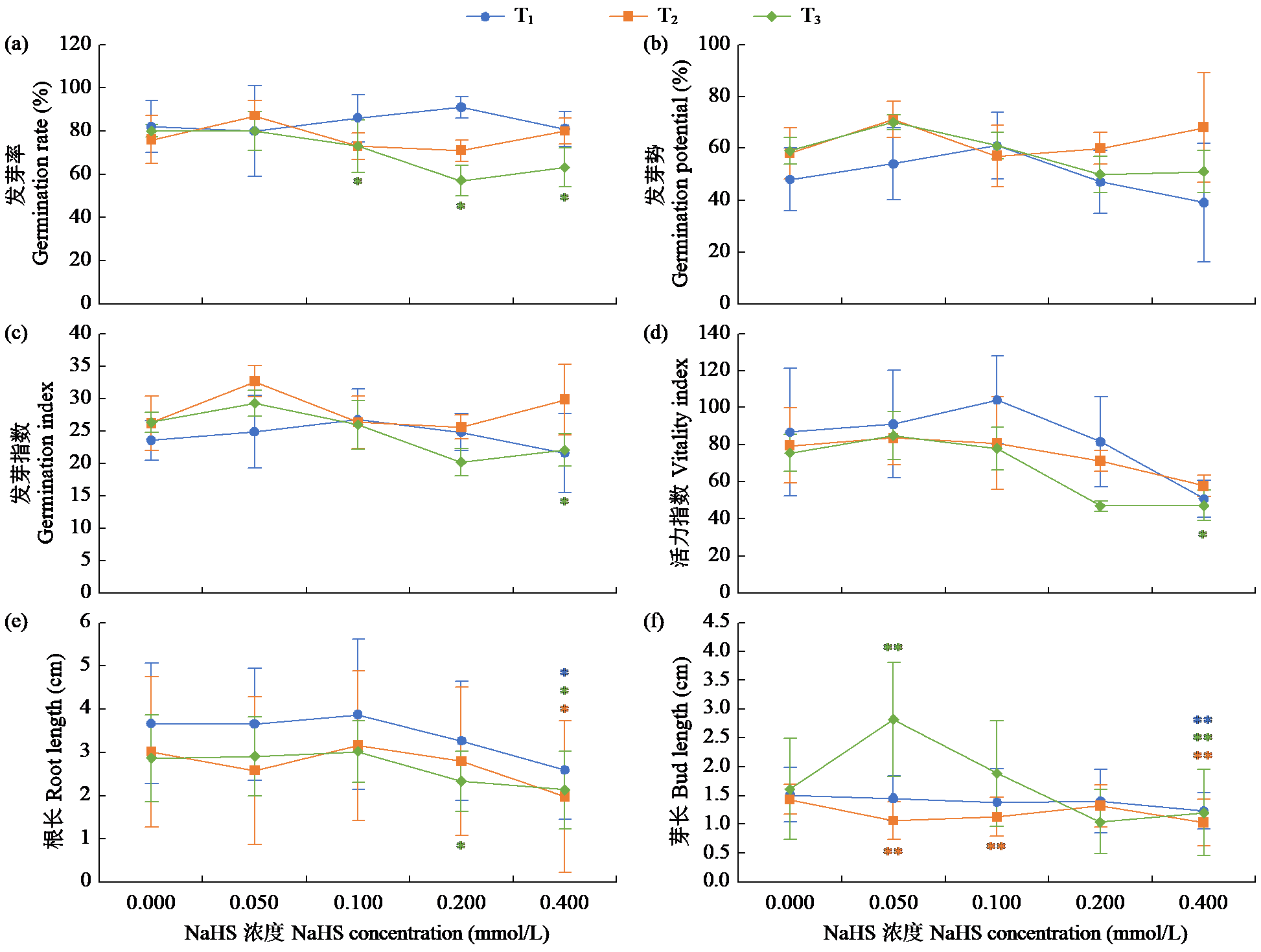
Effects of Exogenous Nitric Oxide and Hydrogen Sulfide on the Germination of Foxtail Millet Seeds
He Shuiling( ), Zhao Xia, Wu Mingqi, Wang Dongsheng(
), Zhao Xia, Wu Mingqi, Wang Dongsheng( )
)
- College of Life Sciences, Shanxi Normal University, Taiyuan 030032, Shanxi, China
| [1] |
Garima P, Gopal M, Kajal K. Genome-wide development and use of microsatellite marker for large-scale genotyping applications in foxtail millet. DNA Research, 2013, 20(2):197-207.
doi: 10.1093/dnares/dst002 pmid: 23382459 |
| [2] | 孙加梅, 王雪梅, 王东健, 等. 谷子种质资源遗传多样性研究. 山东农业科学, 2013, 45(3):33-37. |
| [3] |
孙玉莹, 邱雪梅, 叶芯妤, 等. 植物中硫化氢和一氧化氮信号的交互作用. 生物技术通报, 2020, 36(8):153-161.
doi: 10.13560/j.cnki.biotech.bull.1985.2020-0084 |
| [4] | 崔文玉, 许新月, 张仁堂, 等. 硫化氢和一氧化氮的交互作用对香蕉采后品质及抗氧化体系的影响. 食品与发酵工业, 2020, 46(13):166-173. |
| [5] | 解梦洁, 贺烽, 张丽萍, 等. H2S和NO在大白菜抵抗高温胁迫中的作用. 农业环境科学学报, 2018, 37(6):1079-1085. |
| [6] |
Zhu L Q, Du H Y, Wang W, et al. Synergistic effect of nitric oxide with hydrogen sulfide on inhibition of ripening and softening of peach fruits during storage. Scientia Horticulturae, 2019, 256:108591.
doi: 10.1016/j.scienta.2019.108591 |
| [7] | 刘建新, 王鑫, 雷蕊霞. 外源一氧化氮供体SNP对黑麦草种子萌发和幼苗生长的影响. 生态学杂志, 2007(3):393-398. |
| [8] |
刘建新, 刘瑞瑞, 刘秀丽, 等. 外源H2S供体NaHS浸种对盐碱胁迫下裸燕麦种子萌发特性的影响. 植物研究, 2021, 41(6):870-877.
doi: 10.7525/j.issn.1673-5102.2021.06.004 |
| [9] | 周永斌, 殷有, 苏宝玲, 等. 外源一氧化氮供体对几种植物种子的萌发和幼苗生长的影响. 植物生理学通讯, 2005(3):316-318. |
| [10] | 戴知宁, 陈明堂, 刘颖, 等. 硝普钠浸种对低温胁迫下小麦种子萌发和幼苗生长的影响. 大麦与谷类科学, 2018, 35(2):36-41. |
| [11] | 刘锐锋, 郭希凯, 张华. 硫化氢对小麦种子萌发早期淀粉酶活性的影响. 安徽农业科学, 2010, 38(14):7218-7219,7226. |
| [12] | 黄菡, 郭莎莎, 陈良超, 等. 外源硫化氢对盐胁迫下茶树抗氧化特性的影响. 植物生理学报, 2017, 53(3):497-504. |
| [13] | 张爱慧, 朱士农. 外源一氧化氮对不同贮藏年限甘蓝、黄瓜种子萌发活力的影响. 江苏农业科学, 2012, 40(3):109-111. |
| [14] | 王晓颖, 朱慧森, 黄志超, 等. 不同浸种处理对野生草地早熟禾种子萌发特性的影响. 山西农业科学, 2020, 48(9):1418-1423. |
| [15] | 刘皓玥, 张晓萌, 杜玉贤, 等. 温度对4种秋眠型苜蓿种子萌发的影响. 今日畜牧兽医, 2021, 37(3):9-11. |
| [16] | 李应心, 柳夏艳, 孔令宇, 等. 四种有机活性物质对黄瓜种子萌发的影响. 腐植酸, 2021(4):13-19. |
| [17] |
Palmer R M, Ferrige A G, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature, 1987, 327:524-526.
doi: 10.1038/327524a0 |
| [18] | Ignarro L J, Buga G M, Wood K S, et al. Endothelium derived relaxing factor produced and released from artery and vein is nitric oxide. Proceedings of the National Academy of Sciences,USA, 1987, 84:9265-9269. |
| [19] |
Corpas F J, González-Gordo S, Cañas A, et al. Nitric oxide and hydrogen sulfide in plants:which comes first?. Journal of Experimental Botany, 2019, 70(17):4391-4404.
doi: 10.1093/jxb/erz031 |
| [20] |
Yamasaki H, Cohen M F. Biological consilience of hydrogen sulfide and nitric oxide in plants:Gases of primordial earth linking plant,microbial and animal physiologies. Nitric Oxide-Biology and Chemistry, 2016, 55/56:91-100.
doi: 10.1016/j.niox.2016.04.002 |
| [21] |
Hardeland R. Melatonin in plants-diversity of levels and multiplicity of functions. Frontiers in Plant Science, 2016, 7:198.
doi: 10.3389/fpls.2016.00198 pmid: 26925091 |
| [22] |
Ramesh S A, Tyerman S D, Gilliham M, et al. γ-Aminobutyric acid (GABA) signallingin plants. Cellular and Molecular Life Sciences, 2017, 74:1577-1603.
doi: 10.1007/s00018-016-2415-7 |
| [23] |
Vanstraelen M, Benková E. Hormonal interactions in the regulation of plant development. Annual Review of Cell and Developmental Biology, 2012, 28:463-487.
doi: 10.1146/annurev-cellbio-101011-155741 pmid: 22856461 |
| [24] | 郑光华. 种子生理学. 北京: 科学出版社, 2004. |
| [25] |
Barrero J M, Jacobsen J V, Talbot M J, et al. Grain dormancy and light quality effects on germinationin the model grass Brachypolium distachyon. The New Phytologist, 2012, 193(2):376-386.
doi: 10.1111/j.1469-8137.2011.03938.x |
| [26] | 宋占丽, 秦德明, 刁永强, 等. 不同浸种时间对毛叶苕子种子吸胀作用及萌发的影响. 种子科技, 2021, 39(13):29-30. |
| [27] | 曾燕, 彭运祥, 杨细林, 等. 浸种时间对直播杂交水稻种子萌发的影响. 农业科技通讯, 2020(12):87-90. |
| [28] | 罗阳, 蔺伟虎, 田沛. 浸种温度和光照时间对披碱草种子萌发的影响. 种子, 2021, 40(4):11-16,21. |
| [29] | 李欣勇, 黄迎, 张静文. 风车草种子休眠及萌发特性研究. 种子, 2021, 40(5):57-62. |
| [1] | Ma Jiyu, Wang Shuang, Li Yun, Guo Zhenqing, Wang Jian, Lin Xiaohu, Han Yucui. Effects of Planting Density on Agronomic Characteristics and Yield of Foxtail Millet [J]. Crops, 2023, 39(2): 222-228. |
| [2] | Wang Huimin, Li Minghao, Li Yun, Li Han, Wang Shuang, Lin Xiaohu, Han Yucui. Identification and Evaluation of Salt-Alkali Tolerance of Foxtail Millet Cultivars (Lines) at Germination Stage [J]. Crops, 2023, 39(2): 57-66. |
| [3] | Bian Shuhui, Xing Guofang, Liang Xin, Zhang Shuwei, Wang Jianing, Ye Haoyu. Effects of Different Forms Selenium and Dosage on Foxtail Millet Growth and Physiology at Seedling Stage [J]. Crops, 2023, 39(1): 152-157. |
| [4] | Wang Qi, Xu Yanli, Yan Peng, Dong Haosheng, Zhang Wei, Lu Lin, Dong Zhiqiang. Effects of Polyaspartic Acid-Chitosan on Agronomic Traits, Yield and Nitrogen Use of Spring Foxtail Millet [J]. Crops, 2023, 39(1): 58-67. |
| [5] | Zhao Xiaoqin, Jia Ruiling, Liu Junxiu, Liu Yanming, Wen Yinhua, Shi Lili, Zhang Juanning, Ma Ning. Agronomic Traits and Genetic Diversity Analysis of 120 Foxtail Millet Germplasms [J]. Crops, 2022, 38(6): 61-69. |
| [6] | Li Zhihua, Mu Tingting, Li Aijun. Breeding and Heterosis Analysis of Two Line Parents of Foxtail Millet Hybrids with Similar Growth Process [J]. Crops, 2022, 38(6): 75-81. |
| [7] | Ma Ke, Feng Lei, Zhao Xiatong, Zhang Liguang, Yuan Xiangyang, Dong Shuqi, Guo Pingyi, Song Xi’e. Effects of Sowing Distance and Sowing Amount on the Growth Characteristics and Yield of Zhangzagu 10 [J]. Crops, 2022, 38(4): 172-178. |
| [8] | Pang Xingyue, Wan Lin, Li Su, Wang Yuhang, Liu Chen, Xiao Xiaolu, Li Xinhao, Ma Ni. Effects of Exogenous SLs and Nano-K2MoO4 on Seed Germination of Brassica napus L. under Drought Stress [J]. Crops, 2022, 38(4): 214-220. |
| [9] | Lü Jianzhen, Ren Ying, Wang Hongyong, Zhang Tingjun, Ma Jianping, Zhao Kai. Comprehensive Phenotype Evaluation of 264 Major Foxtail Millet Bred Varieties (Lines) [J]. Crops, 2022, 38(4): 22-31. |
| [10] | Li Binghua, Wang Guiqi, Shi Zhigang, Liu Xiaomin, Xu Xian, Zhao Bochui, Cheng Ruhong. Sensitivity of Foxtail Millets (Setaria italica L.) and Weeds to Cyhalofop-Butyl [J]. Crops, 2022, 38(4): 262-266. |
| [11] | Yang Aojun, Chang Qiaoling, Wang Peng, Wang Fang, Gao Yanting, Zhou Guangkuo, Song Xiaojia, Wei Encheng. Effects of Exogenous 5-Aminolevulinic Acid on Seed Germination and Seedling Growth of Maize under Drought Stress [J]. Crops, 2022, 38(3): 194-199. |
| [12] | Qin Na, Zhu Cancan, Dai Shutao, Song Yinghui, Li Junxia, Wang Chunyi. Fine Mapping and Functional Analysis of Yellow Leaf Mutant ylm-1 in Foxtail Millet [J]. Crops, 2022, 38(3): 55-62. |
| [13] | Guo Yongxin, Zhou Hao, Sun Peng, Wang Yaqing, Ma Ke, Li Xiaorui, Dong Shuqi, Guo Pingyi, Yuan Xiangyang. Effects of Planting Patterns on Lodging Resistance and Yield of Zhangza 10 in Different Ecological Areas [J]. Crops, 2022, 38(2): 195-202. |
| [14] | Lu Jun, Qian Yu, Yang Liu, Wang Yong, Chen Yulan. Effects of Storage Time on Seed Germination and Physiological Characteristics of Tobacco Variety Honghuadajinyuan [J]. Crops, 2022, 38(2): 211-214. |
| [15] | Zhao Lirong, Ma Ke, Zhang Liguang, Tang Sha, Yuan Xiangyang, Diao Xianmin. Analysis of Agronomic Traits and Quality of Foxtail Millet Varieties in Different Ecological Regions [J]. Crops, 2022, 38(2): 44-53. |
|
||