VIGS(virus-induced gene silencing)是一种由病毒载体诱导的基因沉默技术,与转基因、基因敲除和反义抑制等方法相比,VIGS技术具有周期短、成本低和高通量等优势[3],因此,VIGS常被用作研究植物基因的功能[4]。VIGS主要是利用病毒载体携带目的基因侵染植物,在其复制和表达过程中形成双链RNA(double stranded RNA,dsRNA),dsRNA在细胞中被特异性核酸内切酶Dicer切割成21~24nt的小分子干扰RNA(small interfering RNA,siRNA)片段,siRNA被RNA聚合酶扩增,以单链的形式和一些蛋白结合形成RNA诱导复合体(RNA-induced silencing complex,RISC),这些复合体与目的基因mRNA特异性互作,导致其降解,从而发生转录后水平的基因沉默[5]。
八氢番茄红素脱氢酶(phytoene desaturase,PDS)是类胡萝卜素合成途径中的关键酶。当该基因被沉默后,类胡萝卜素合成途径被阻断,从而使得植物产生光漂白现象[6-7],即叶片变白。因此,PDS基因被作为报告基因在VIGS沉默体系中普遍应用。目前,应用于植物的VIGS载体包括RNA病毒载体、DNA病毒载体和卫星病毒载体等,不同载体在植物上的沉默株率有很大差异[8]。基于烟草脆裂病毒(Tobacco rattle virus,TRV)开发的VIGS载体是目前应用最广泛的病毒载体,具有沉默株率较高、病毒症状温和且沉默持续性较好等优势[9],但在不同寄主植物中基因沉默的效率及成功率存在差异。已有的研究结果表明,影响VIGS体系沉默株率的因素主要包括以下几个方面:第一,插入基因序列与靶基因序列的同源性越高,沉默效率越好[10];第二,插入基因片段的长度、方向和折叠方式影响基因沉默效率,有研究[11]表明,插入病毒载体的基因片段长度存在有效区间,最适片段长度应该控制在200~350bp;第三,环境因素直接影响病毒载体在植物细胞体内的复制,对于不同的沉默载体,沉默后植株的生长环镜对于基因沉默效率也有显著的影响,大量研究[12-13]表明,对于双分体病毒载体来说,随着培养温度的升高,VIGS病毒症状会随之减弱;第四,接种方法对VIGS沉默株率也有一定的影响,应该根据不同植物类型选择最优的接种方法。现有的接种方式有注射法[14]、灌根法[15]和真空浸润法[16]等。VIGS技术被广泛应用于番茄非生物胁迫(如干旱[17]、低温[18])或生物胁迫(如各类病原菌[19])等有害生物方面的基因功能验证。然而在这些研究中,番茄VIGS的沉默株率和沉默效率却各有差异。
本研究以番茄品种money maker为材料,研究不同农杆菌菌液浓度、不同的接种方法和接种后不同培养温度对番茄PDS基因沉默株率的影响,以期建立并优化出最适的VIGS沉默条件,为后续番茄目标基因的功能研究奠定技术基础。
1 材料与方法
1.1 试验材料
1.1.1 供试番茄、质粒及菌株
本研究选用番茄品种为money maker,由内蒙古自治区农牧业科学院惠赠。病毒载体为TRV,筛选基因为卡那霉素(Kanamycin),酶切位点选用Xba Ⅰ及Kpn Ⅰ。大肠杆菌(Escherichia coli)菌株为Trans1-T1,农杆菌(Agrobacterium tumefaciens)菌株为GV3101。
1.1.2 培养基配制
YEB固体培养基组成为蛋白胨5.00g,牛肉提取物5.00g,酵母提取物1.00g,蔗糖5.00g,MgSO4•7H2O 0.50g,琼脂(液体YEB不添加)15.00g,定容至1.00L。
MMA培养基组成为MES1.95g,MS盐5.00g,蔗糖20.00g,定容至1.00L,调节pH至5.7,高温灭菌冷却后加入乙酰丁香酮200μmol/L。
1.2 试验方法
1.2.1 接种液制备
将带有TRV1和TRV2、TRV1和TRV2-PDS载体的农杆菌,分别在含有Kana 50mg/L和Rif 25mg/L的YEB固体培养基中划线培养,于28℃培养2d。挑取单菌落至3.00mL液体YEB培养基中(Kana 50mg/L、Rif 25mg/L),28℃、200转/min过夜培养。取200μL上述菌液,加入15.00mL YEB液体培养基(Kana 50mg/L、Rif 25mg/L)中,28℃、200转/min培养20~24h。3000转/min离心10min,弃上清,在沉淀中加入MMA培养基,悬浮沉淀,调节OD600至0.50、1.00和1.50。将TRV1和TRV2、TRV1和TRV2-PDS的菌液等体积混合,室温静置3~4h,用于后续番茄叶片的接种。
1.2.2 基因表达量分析
采用RT-PCR的方法,利用RNAzol reagent(北京金百特生物技术有限公司)提取番茄总RNA,使用script III first strand cDNA synthesis kit(北京金百特生物技术有限公司)反转录成cDNA。用番茄PDS引物(F:TTTTGTGTT TGCCGCTCCAG,R:GTCGCTGCGAGATATCCA CT)和番茄GAPDH引物(F:GGGGAAAGTGCT ACCAGCAT,R:ACCCCATTCGTTGTCGTACC)进行PCR扩增,产物通过1.0%凝胶电泳检测后,用Quantity one软件定量分析。
1.2.3 接种方法
注射法接种:在子叶期,用无针头注射器吸取接种液,注入植株子叶背面,每个植株注射200μL,置于20℃黑暗处理24h后,转移至光照16h、黑暗8h、25℃的光照培养箱中继续培养,每个处理共32株幼苗,重复3次。
真空浸润法接种:把子叶期的幼苗浸没于接种液中,在真空压力为0.80MPa条件下静置2min,将植株移入营养钵中,置于20℃黑暗处理24h后,转移至光照16h、黑暗8h、25℃的光照培养箱中继续培养,每个处理共32株幼苗,重复3次。
注射接种+真空浸润法接种:在子叶期,先用注射法接种,置于20℃黑暗处理24h后,再将幼苗取出用真空浸润法接种进行处理,处理后放回光照16h、黑暗8h、25℃的光照培养箱中继续培养。每个处理共32株幼苗,重复3次。
1.2.4 温度处理
将经过注射法接种的番茄幼苗先置于20℃黑暗处理24h后,然后分别在不同温度下进行培养。处理一:光照16h、黑暗8h、25℃;处理二:光照16h、黑暗8h、22℃。每个处理共32株幼苗,重复3次。
1.3 数据处理
以白化植株出现的百分率标记为VIGS的沉默株率,计算公式为:植株沉默株率(%)=(白化株数/转化总株数)×100。
2 结果与分析
2.1 基于PDS基因VIGS沉默体系的建立
为了更好地评价VIGS的沉默株率,利用VIGS对番茄植株中PDS基因进行沉默,通过统计具有光漂白现象的植株数计算VIGS的沉默株率。采用注射法接种25d后,番茄植株出现了光漂白现象(图1)。
图1
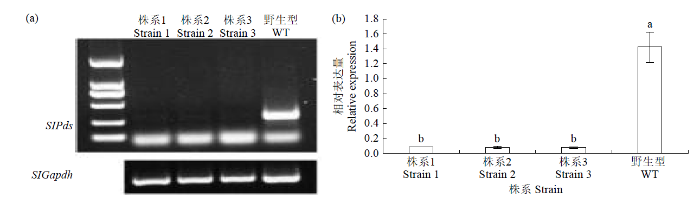
收集沉默植株的叶片,利用RT-PCR分析PDS基因的表达量,结果显示,与对照植株相比,3株番茄中PDS基因的表达量分别降低91.39%、90.91%和91.67%(图2),说明沉默体系能够对目标基因进行沉默。
图2
图2
光漂白植株中PDS基因表达量分析
不同小写字母表示差异显著(P < 0.05),下同
Fig.2
Analysis of PDS gene expression in photobleached plants
The different lowercase letters mean significant difference (P < 0.05), the same below
2.2 农杆菌不同的接菌浓度对VIGS沉默株率的影响
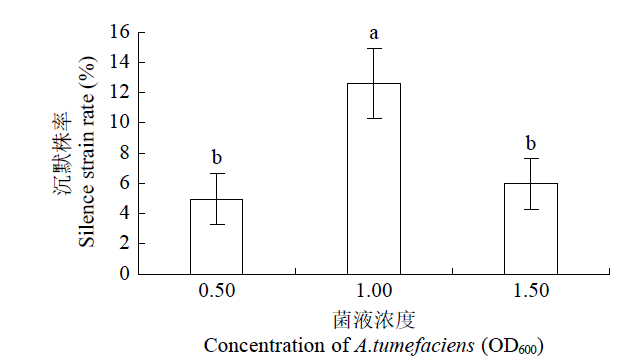
如图3所示,农杆菌的不同接菌浓度对番茄的VIGS沉默株率有显著影响。当农杆菌菌液OD600= 1.00时,PDS基因的沉默比例最高,为13%,而农杆菌菌液OD600=0.50和1.50时沉默比例仅为5%和6%。后续试验选用农杆菌菌液OD600=1.00来优化其他的条件。
图3
图3
农杆菌不同接种浓度对PDS基因沉默株率的影响
Fig.3
Effects of A.tumefaciens concentration on PDS gene silence strain rate
2.3 农杆菌不同接种方法对VIGS沉默株率的影响
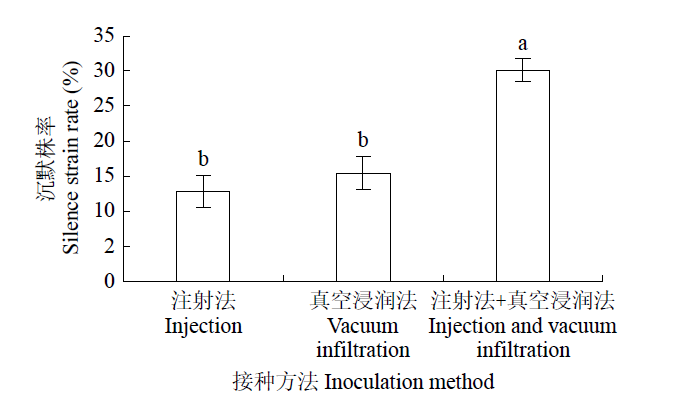
如图4所示,注射法或真空浸润法接种的沉默比例分别为13%和15%,二者没有显著差异,而注射法+真空浸润法目标基因的沉默株率高达30%,显著高于其他2种方法。
图4
图4
农杆菌不同接种方法对沉默株率的影响
Fig.4
Effects of A.tumefaciens different inoculation methods on silence strain rate
2.4 接种后的不同培养温度对VIGS沉默株率的影响
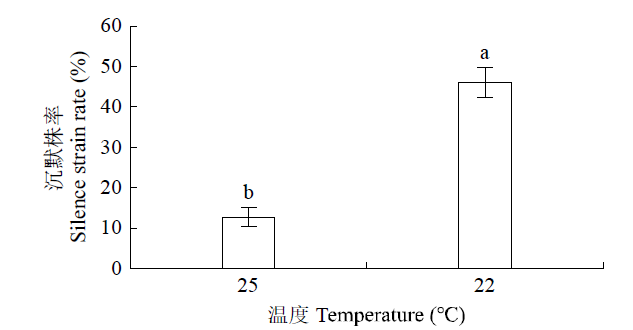
如图5所示,当接种植株培养温度为25℃时,目标基因的沉默株率为13%,而培养温度为22℃时,目标基因的沉默株率为46%,显著高于25℃的沉默株率,表明接种后植株的培养温度对基因沉默株率有影响。
图5
图5
不同温度处理对沉默株率的影响
Fig.5
Effects of different temperature treatments on silence strain rate
3 讨论
VIGS是研究植物基因功能的常规工具,利用该技术已经在番茄中鉴定或验证了上百种基因。TRV重组病毒载体因其沉默株率较高、病毒症状较为温和且沉默持续时间较长等独特优势,已成为番茄中应用最为广泛的VIGS沉默载体[20]。但目前这种技术仍然存在一定的缺陷,主要因为VGIS载体使用具有一定的宿主范围限制,其中大部分只是在烟草和番茄中应用。针对番茄VIGS体系的TRV病毒载体,研究者一直在对该体系进行优化,例如载体的改良、接种方式和植株的生长发育环境等方面。
目前已有的VIGS沉默方法分别是机械接种、金属离子轰击以及农杆菌介导,其中农杆菌介导相比于其他方法更为简单高效且应用最为广泛[21]。农杆菌介导的方法有多种,包括牙签刺伤法、真空渗透法、叶背注射法和高压喷枪法等,各有优缺点且诱导沉默效率和稳定性存在差异。有研究[22]证明,在烟草中温度可以通过次级siRNA调节VIGS的效率,在高温作用下,初级siRNA很快降解,不能获得持续稳定沉默植株。除此之外,许多抗病毒途径也是依赖于温度调节,例如AGOs、DCLs和RDRs[23-24]。对于病毒来说,侵入寄主细胞后,也需要在合适的温度进行增殖[25]。除低温依赖病毒外,大多数植物病毒在20℃~25℃都可以进行侵染[26]。本研究发现,培养温度为22℃时,VIGS的沉默株率显著增加,主要是由于低温条件下更适宜该病毒载体的侵染与扩增,同时避免了高温条件下植物体内抗病毒物质的大量积累,降低了siRNA的损失[24]。周婷[27]研究同样发现温度是影响VIGS体系的重要因素,在使用TRV病毒载体沉默马铃薯基因过程当中,20℃~24℃范围内成功沉默马铃薯PDS基因。同样,Tuttle等[28]研究发现,对于双分体病毒载体CLCrV在18℃~22℃比26℃~30℃条件下病毒的积累量高出10倍,基因的沉默效果在18℃~22℃条件下也明显优于26℃~30℃条件。因此,沉默植株的合适生长培养环境也对病毒载体在植物细胞内增殖起到促进作用。本研究同样发现,相对较低的培养温度有利于提升沉默株率,与之前的研究结果一致。
针对VIGS沉默体系,目前还存在持续性差、沉默株率低和沉默不均一等缺陷,需要不断进行体系优化,使其沉默株率更高。本研究发现除了温度是影响VIGS沉默株率的主要因素,农杆菌浓度以及接种方式对TRV介导的番茄VIGS体系沉默株率也存在一定程度的影响。
4 结论
在money maker品种中,农杆菌浓度、接种方式和接种后植株培养温度对VIGS沉默效果均具有显著影响,其中最适宜的农杆菌接种浓度OD值为1.00,最佳的接种方式是注射法+真空浸润法,接种后最适宜的培养温度为22℃。
参考文献
A simple and efficient Agrobacterium-mediated procedure for transformation of tomato
DOI:10.1007/s12038-009-0049-8
PMID:19805904
[本文引用: 1]

We describe a highly efficient and reproducible Agrobacterium-mediated transformation protocol applicable to several varieties of tomato (Solanum lycopersicum, earlier known as Lycopersicum esculentum). Conditions such as co-cultivation period, bacterial concentration, concentration of benzyl amino purine (BAP), zeatin and indole acetic acid (IAA) were optimized. Co-cultivation of explants with a bacterial concentration of 108 cells/ml for three days on 2 mg/l BAP, followed by regeneration on a medium containing 1 mg/ml zeatin resulted in a transformation frequency of 41.4%. Transformation of tomato plants was confirmed by Southern blot analysis and beta-glucuronidase (GUS) assay. The protocol developed showed very high efficiency of transformation for tomato varieties Pusa Ruby, Arka Vikas and Sioux. The optimized transformation procedure is simple, efficient and does not require tobacco, Petunia, tomato suspension feeder layer or acetosyringone.
VIGS-genomics goes functional
DOI:10.1016/j.tplants.2009.09.002 URL [本文引用: 1]
Applications and advantages of virus-induced gene silencing for gene function studies in plants
DOI:10.1111/tpj.2004.39.issue-5 URL [本文引用: 1]
病毒诱导的基因沉默(VIGS)研究进展
病毒诱导的基因沉默(Virus-induced gene silencing,VIGS)是一种RNA 介导的植物抗病毒机制,近年来已被用作研究植物基因功能的反向遗传学手段。与转基因、基因敲除、反义抑制等基因功能研究方法相比,VIGS 技术研究周期短,不需要遗传转化,具有低成本、高通量等优势。因此,VIGS已被广泛应用于植物抗病抗逆、生长发育以及代谢调控等生理途径相关基因的功能鉴定。综述了VIGS 的机制、技术发展、沉默效率的影响因素及其在植物基因功能鉴定中的应用,并对VIGS 在植物性状改良及植物保护方面的应用前景进行了展望。
Initiation and maintenance of virus-induced gene silencing
DOI:10.1105/tpc.10.6.937
PMID:9634582
[本文引用: 1]

The phytoene desaturase (PDS) gene of Nicotiana benthamiana was silenced in plants infected with potato virus X (PVX) vectors carrying PDS inserts, and a green fluorescent protein (GFP) transgene was silenced in plants infected with PVX-GFP. This virus-induced gene silencing (VIGS) is post-transcriptional and cytoplasmic because it is targeted against exons rather than introns of PDS RNA and against viral RNAs. Although PDS and GFP RNAs are most likely targeted through the same mechanism, the VIGS phenotypes differed in two respects. PDS mRNA was targeted by VIGS in all green tissue of the PVX-PDS-infected plant, whereas PVX-PDS was not affected. In contrast, VIGS of the GFP was targeted against PVX-GFP. Initially, VIGS of the GFP was initiated in all green tissues, as occurred with PDS VIGS. However, after 30 days of infection, the GFP VIGS was no longer initiated in newly emerging leaves, although it was maintained in tissue in which it had already been initiated. Based on these analyses, we propose a model for VIGS in which the initiation of VIGS is dependent on the virus and maintenance of it is virus independent.
Virus-induced gene silencing can persist for more than 2 years and also be transmitted to progeny seedlings in Nicotiana benthamiana and tomato
DOI:10.1111/pbi.2011.9.issue-7 URL [本文引用: 1]
Size constraints for targeting post-transcriptional gene silencing and for RNA- directed methylation in Nicotiana benthamiana using a potato virus X vector
DOI:10.1046/j.1365-313x.2001.00976.x URL [本文引用: 1]
Optimized cDNA libraries for virus-induced gene silencing (VIGS) using tobacco rattle virus
DOI:10.1186/1746-4811-4-1
[本文引用: 1]

Research in plant science laboratories often involves usage of many different species, cultivars, ecotypes, mutants, alleles or transgenic lines. This creates a great challenge to keep track of the identity of experimental plants and stored samples or seeds.
Geminivirus-mediated gene silencing from cotton leaf crumple virus is enhanced by low temperature in cotton
DOI:10.1104/pp.108.123869
PMID:18621976
[本文引用: 1]

A silencing vector for cotton (Gossypium hirsutum) was developed from the geminivirus Cotton leaf crumple virus (CLCrV). The CLCrV coat protein gene was replaced by up to 500 bp of DNA homologous to one of two endogenous genes, the magnesium chelatase subunit I gene (ChlI) or the phytoene desaturase gene (PDS). Cotyledons of cotton cultivar 'Deltapine 5415' bombarded with the modified viral vectors manifested chlorosis due to silencing of either ChlI or PDS in approximately 70% of inoculated plants after 2 to 3 weeks. Use of the green fluorescence protein gene showed that replication of viral DNA was restricted to vascular tissue and that the viral vector could transmit to leaves, roots, and the ovule integument from which fibers originate. Temperature had profound effects on vector DNA accumulation and the spread of endogenous gene silencing. Consistent with reports that silencing against viruses increases at higher temperatures, plants grown at a 30 degrees C/26 degrees C day/night cycle had a greater than 10-fold reduction in viral DNA accumulation compared to plants grown at 22 degrees C/18 degrees C. However, endogenous gene silencing decreased at 30 degrees C/26 degrees C. There was an approximately 7 d delay in the onset of gene silencing at 22 degrees C/18 degrees C, but silencing was extensive and persisted throughout the life of the plant. The extent of silencing in new growth could be increased or decreased by changing temperature regimes at various times following the onset of silencing. Our experiments establish the use of the CLCrV silencing vector to study gene function in cotton and show that temperature can have a major impact on the extent of geminivirus-induced gene silencing.
Enhancement of virus-induced gene silencing in tomato by low temperature and low humidity
Agrodrench:a novel and effective agroinoculation method for virus-induced gene silencing in roots and diverse solanaceous species
DOI:10.1111/tpj.2004.40.issue-2 URL [本文引用: 1]
Characterization of a brome mosaic virus strain and its use as a vector for gene silencing in monocotyledonous hosts
DOI:10.1094/MPMI-19-1229
PMID:17073305
[本文引用: 1]

Virus-induced gene silencing (VIGS) is used to analyze gene function in dicotyledonous plants but less so in monocotyledonous plants (particularly rice and corn), partially due to the limited number of virus expression vectors available. Here, we report the cloning and modification for VIGS of a virus from Festuca arundinacea Schreb. (tall fescue) that caused systemic mosaic symptoms on barley, rice, and a specific cultivar of maize (Va35) under greenhouse conditions. Through sequencing, the virus was determined to be a strain of Brome mosaic virus (BMV). The virus was named F-BMV (F for Festuca), and genetic determinants that controlled the systemic infection of rice were mapped to RNAs 1 and 2 of the tripartite genome. cDNA from RNA 3 of the Russian strain of BMV (R-BMV) was modified to accept inserts from foreign genes. Coinoculation of RNAs 1 and 2 from F-BMV and RNA 3 from R-BMV expressing a portion of a plant gene to leaves of barley, rice, and maize plants resulted in visual silencing-like phenotypes. The visual phenotypes were correlated with decreased target host transcript levels in the corresponding leaves. The VIGS visual phenotype varied from maintained during silencing of actin 1 transcript expression to transient with incomplete penetration through affected tissue during silencing of phytoene desaturase expression. F-BMV RNA 3 was modified to allow greater accumulation of virus while minimizing virus pathogenicity. The modified vector C-BMV(A/G) (C for chimeric) was shown to be useful for VIGS. These BMV vectors will be useful for analysis of gene function in rice and maize for which no VIGS system is reported.
VIGS介导的番茄转录因子SIDREB2基因的耐旱特征
SlDREB2 基因是利用rd29A 基因启动子的DRE 元件通过酵母单杂交技术从番茄幼苗(丽春)cDNA 文库中筛选得到的属于EREBP 家族的一个转录因子基因。利用构建病毒VIGS 载体,分别用含pBINTRB6、pTV00∷SlDREB2-1 和pTV00∷SlDREB2-2 重组载体的农杆菌GV3101 菌液灌根与叶片注射法侵染番茄植株,转化7 d 后进行病毒DNA 检测与验证。测定了干旱胁迫下侵染成功的番茄植株与野生型株系的生理指标,并通过Real-time qPCR 分析了VIGS 介导的SlDREB2 转录因子基因的表达特征。番茄植株干旱3 周胁迫处理后测定结果表明,野生番茄中脯氨酸和可溶性糖水平要高于被VIGS 病毒侵染的番茄植株,而丙二醛水平要低于被侵染植株,并且指标显示被pTV00∷SlDREB2-2 重组载体的农杆菌GV3101 菌液侵染的番茄植株抗旱性明显降低;复水1 周后,受VIGS 浸染株系丙二醛含量明显高于野生型,而可溶性糖与脯氨酸累积却低于野生型。Real-time qPCR 结果表明,经VIGS 病毒侵染过的番茄植株在胁迫处理时期SlDREB2 基因的相对表达量都明显低于野生型株系,表明番茄SlDREB2基因保守域末端459 bp 的特异片段具有较显著的沉默效果,说明VIGS 介导的SlDREB2 基因的表达受干旱诱导,该基因可以作为抗旱型作物品种改良的有价值基因。
Virus-induced gene silencing (VIGS) for functional analysis of MYB80 gene involved in solanum lycopersicum cold tolerance
DOI:10.1007/s00709-018-1302-5 [本文引用: 1]
病毒诱导的基因沉默在番茄功能基因组学研究中的应用进展
对近年来病毒诱导的基因沉默(virus-induced gene silencing,VIGS)技术在番茄果实发育、激素调控、植物—病原物互作、防御害虫及非生物胁迫应答相关基因功能研究方面的进展进行了综述。
Temperature modulates virus‐ induced transcriptional gene silencing via secondary small RNAs
DOI:10.1111/nph.v232.1 URL [本文引用: 1]
Temperature-dependent survival of turnip crinkle virus-infected arabidopsis plants relies on an rna silencing-based defense that requires DCL2,AGO2,and HEN1
DOI:10.1128/JVI.00497-12
URL
[本文引用: 1]

\n While RNA silencing is a potent antiviral defense in plants, well-adapted plant viruses are known to encode suppressors of RNA silencing (VSR) that can neutralize the effectiveness of RNA silencing. As a result, most plant genes involved in antiviral silencing were identified by using debilitated viruses lacking silencing suppression capabilities. Therefore, it remains to be resolved whether RNA silencing plays a significant part in defending plants against wild-type viruses. We report here that, at a higher plant growth temperature (26°C) that permits rigorous replication of\n Turnip crinkle virus\n (TCV) in\n Arabidopsis\n, plants containing loss-of-function mutations within the\n Dicer-like 2\n (\n DCL2\n ),\n Argonaute 2\n (\n AGO2\n ), and\n HEN1\n RNA methyltransferase genes died of TCV infection, whereas the wild-type Col-0 plants survived to produce viable seeds. To account for the critical role of DCL2 in ensuring the survival of wild-type plants, we established that higher temperature upregulates the activity of DCL2 to produce viral 22-nucleotide (nt) small interfering RNAs (vsRNAs). We further demonstrated that DCL2-produced 22-nt vsRNAs were fully capable of silencing target genes, but that this activity was suppressed by the TCV VSR. Finally, we provide additional evidence supporting the notion that TCV VSR suppresses RNA silencing through directly interacting with AGO2. Together, these results have revealed a specialized RNA silencing pathway involving DCL2, AGO2, and HEN1 that provides the host plants with a competitive edge against adapted viruses under environmental conditions that facilitates robust virus reproduction.\n
RDR6 has a broad-spectrum but temperature-dependent antiviral defense role in Nicotiana benthamiana
DOI:10.1128/JVI.79.24.15209-15217.2005
URL
[本文引用: 2]

\n SDE1/SGS2/RDR6, a putative RNA-dependent RNA polymerase (RdRP) from\n Arabidopsis thaliana\n, has previously been found to be indispensable for maintaining the posttranscriptional silencing of transgenes, but it is seemingly redundant for antiviral defense. To elucidate the antiviral role of this RdRP in a different host plant and to evaluate whether plant growth conditions affect its role, we down-regulated expression of the\n Nicotiana benthamiana\n homolog, NbRDR6, and examined the plants for altered susceptibility to various viruses at different growth temperatures. The results we describe here clearly show that plants with reduced expression of NbRDR6 were more susceptible to all viruses tested and that this effect was more pronounced at higher growth temperatures. Diminished expression of NbRDR6 also permitted efficient multiplication of tobacco mosaic virus in the shoot apices, leading to serious disruption with microRNA-mediated developmental regulation. Based on these results, we propose that NbRDR6 participates in the antiviral RNA silencing pathway that is stimulated by rising temperatures but suppressed by virus-encoded silencing suppressors. The relative strengths of these two factors, along with other plant defense components, critically influence the outcome of virus infections.\n
Elevated ambient temperature differentially affects virus resistance in two tobacco species
DOI:10.1094/PHYTO-11-14-0300-R
PMID:26474332
[本文引用: 1]

Antiviral defense of plants is usually enhanced by an elevated temperature under natural conditions. In order to better understand this phenomenon, we carried out temperature shift experiments with Nicotiana glutinosa plants that were infected with Potato virus X (PVX) or the necrotic strain of Potato virus Y (PVY(N)). The virus titer of the plants was found to be much lower when they were maintained at 30°C compared with 22°C, particularly in the upper leaves. PVX resistance at 30°C persisted for a short period even when temperature was shifted back to 22°C. In contrast, N. benthamiana lost the virus resistance immediately after the temperature dropped to 22°C. Expression analysis of two RNA-dependent RNA polymerases in N. glutinosa (NgRDR) showed that a 12-day treatment at 30°C increased the expression of NgRDR1, while NgRDR6 was not affected. In addition, the NgRDR6 mRNA level correlated with the PVX titer but was unaffected by PVY(N) infection. These observations indicate that PVX and PVY(N), although they are both RNA viruses, might trigger different defense responses at elevated temperatures. Our study provides valuable data for a better understanding of the temperature-regulated host virus interaction.
Chinese wheat mosaic virus- induced gene silencing in monocots and dicots at low temperature
DOI:10.3389/fpls.2018.01627 URL [本文引用: 1]
Geminivirus-mediated gene silencing from cotton leaf crumple virus is enhanced by low temperature in cotton
DOI:10.1104/pp.108.123869
PMID:18621976
[本文引用: 1]

A silencing vector for cotton (Gossypium hirsutum) was developed from the geminivirus Cotton leaf crumple virus (CLCrV). The CLCrV coat protein gene was replaced by up to 500 bp of DNA homologous to one of two endogenous genes, the magnesium chelatase subunit I gene (ChlI) or the phytoene desaturase gene (PDS). Cotyledons of cotton cultivar 'Deltapine 5415' bombarded with the modified viral vectors manifested chlorosis due to silencing of either ChlI or PDS in approximately 70% of inoculated plants after 2 to 3 weeks. Use of the green fluorescence protein gene showed that replication of viral DNA was restricted to vascular tissue and that the viral vector could transmit to leaves, roots, and the ovule integument from which fibers originate. Temperature had profound effects on vector DNA accumulation and the spread of endogenous gene silencing. Consistent with reports that silencing against viruses increases at higher temperatures, plants grown at a 30 degrees C/26 degrees C day/night cycle had a greater than 10-fold reduction in viral DNA accumulation compared to plants grown at 22 degrees C/18 degrees C. However, endogenous gene silencing decreased at 30 degrees C/26 degrees C. There was an approximately 7 d delay in the onset of gene silencing at 22 degrees C/18 degrees C, but silencing was extensive and persisted throughout the life of the plant. The extent of silencing in new growth could be increased or decreased by changing temperature regimes at various times following the onset of silencing. Our experiments establish the use of the CLCrV silencing vector to study gene function in cotton and show that temperature can have a major impact on the extent of geminivirus-induced gene silencing.