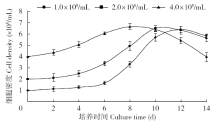
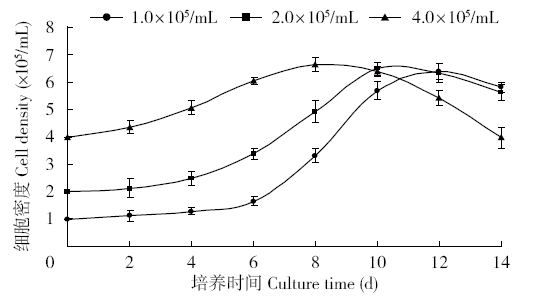
Crops ›› 2018, Vol. 34 ›› Issue (1): 35-40.doi: 10.16035/j.issn.1001-7283.2018.01.005
Previous Articles Next Articles
Establishment of Sorghum Cell Suspensions with Young Leaves
Hao Yanfang1,2,Wang Liangqun1,2,Liu Yong1,Zhang Wei1,2,Yang Wei1,Bai Hongyan1,Wu Bo1
- 1 Sorghum Institute, Shanxi Academy of Agricultural Sciences, Jinzhong 030600, Shanxi, China
2 Shanxi Key Laboratory of Sorghum Genetic and Germplasm Innovation, Jinzhong 030600, Shanxi, China
| [1] | 刘庆昌, 吴国良 . 植物细胞组织培养. 北京: 中国农业大学出版社, 2003: 139-162. |
| [2] | 杜晓映 . 葡萄细胞的悬浮培养及诱导子对悬浮细胞多酚产量影响的研究. 杨凌:西北农林科技大学, 2008. |
| [3] | 葛台明, 章荣德, 秦发兰 , 等. 冬小麦原生质体培养的胚状体直接发生. 生物工程学报, 2000,16(5):609-613. |
| [4] | 蔺忠龙, 白现广, 吕广磊 , 等. 疣粒野生稻胚性悬浮细胞系的建立及其原生质体的培养和植株再生. 植物生理学通讯, 2008,44(6):1181-1184. |
| [5] |
Xiang F N, Xia G M, Chen H M . Asymmetric somatic hybridization between wheat (Triticum aestivum) and Avena sativa L. Science in China Series C-Life Sciences, 2003,46(3):243-252.
doi: 10.1360/03yc9026 pmid: 18763139 |
| [6] |
Xiao W, Huang X, Gong Q , et al. Somatic hybrids obtained by asymmetric protoplast fusion between Musa Silk cv. Guoshanxiang (AAB) and Musa acuminata cv. Mas (AA). Plant Cell, Tissue and Organ Culture, 2009,97:313-321.
doi: 10.1007/s11240-009-9530-1 |
| [7] | Wagiran A, Ismail I, Zain C R , et al. Agrobacterium tumefaciens-mediated transformation of the isopentenyltransferase gene in japonica rice suspension cell culture. Australian Journal of Crop Science, 2010,4(6):421-429. |
| [8] |
Masteller V J, Holden D J . The growth of and organ formation from callus tissue of sorghum. Plant Physiology, 1970,45:362-364.
doi: 10.1104/pp.45.3.362 pmid: 16657320 |
| [9] |
Wernicke W, Brettel R I S . Morphogenesis from cultured leaf tissue of Sorghum bicolor-culture initiation. Protoplasma, 1982,111:19-27.
doi: 10.1007/BF01287643 |
| [10] |
Kumaravadivel N, Rangasamy S R S . Plant regeneration from sorghum anther cultures and field evaluation of progeny. Plant Cell Reports, 1994,13:286-290.
doi: 10.1007/BF00233321 pmid: 24193766 |
| [11] | Jogeswar G, Ranadheer D, Anjaiah V , et al. High frequency somatic embryogenesis and regeneration in different genotypes of Sorghum bicolor (L. ) Moench from immature inflorescence explants. In Vitro Cellular and Developmental Biology-Plant, 2007,43:159-166. |
| [12] |
Oldach C H, Morgenstern A, Rother S , et al. Efficient in vitro plant regeneration from immature zygotic embryos of pearl millet [Pennisetum glaucum (L.) R. Br. ] and Sorghum bicolor (L.) Moench. Plant Cell Reports, 2001,20:416-421.
doi: 10.1007/s002990100335 |
| [13] |
Kishore N S, Visarada K B R S,Lakshmi Y A , et al. In vitro culture methods in sorghum with shoot tip as the explant material. Plant Cell Reports, 2006,25:174-182.
doi: 10.1007/s00299-005-0044-y pmid: 16402251 |
| [14] |
Mythili P K, Seetharama N, Reddy V D . Plant regeneration from embryogenic cell suspension cultures of wild sorghum (Sorghum dimidiatum Stapf. ). Plant Cell Reports, 1999,18:424-428.
doi: 10.1007/s002990050597 |
| [15] |
王良群, 刘勇, 郝艳芳 , 等. 高粱茎尖和幼穗细胞悬浮系的建立. 农学学报, 2013,3(6):1-3.
doi: 10.3969/j.issn.1007-7774.2013.06.001 |
| [16] | 韩福光, 张颖 . 高粱不同外植体愈伤组织诱导的研究. 辽宁农业科学, 1993(1):45-48. |
| [17] |
王良群, 白志良, 王呈祥 , 等. 高粱不同外植体再生苗分化培养的研究. 杂粮作物, 2004,24(5):262-263.
doi: 10.3969/j.issn.2095-0896.2004.05.005 |
| [18] |
Baker C J, Mock N M . An improved method for monitoring cell death in cell suspension and leaf disc assays using evans blue. Plant Cell, Tissue and Organ Culture, 1994,39:7-12.
doi: 10.1007/BF00037585 |
| [19] |
赖钟雄, 陈振光 . 龙眼胚性细胞悬浮培养再生植株. 应用与环境生物学报, 2002,8(5):485-491.
doi: 10.3321/j.issn:1006-687X.2002.05.008 |
| [20] | 袁文娅, 关淑艳, 马红丹 , 等. 玉米自交系H99与齐319幼胚愈伤组织诱导条件的优化. 作物杂志, 2013(4):48-51. |
| [21] |
蔡汉权, 赖钟雄, 林珊珊, 等 . 罗勒 ( Ocimum basilicum)悬浮细胞系的建立与保持. 热带作物学报, 2006,27(1):44-48.
doi: 10.3969/j.issn.1000-2561.2006.01.009 |
| [22] | 欧巧明, 厚毅清, 包梅年 , 等. 中药半夏单细胞悬浮培养、胚胎发生及植株再生. 中国生物工程杂志, 2012,32(10):39-49. |
| [23] |
戴雪梅, 华玉伟, 李哲 , 等. 植物悬浮细胞培养的关键技术及存在问题. 热带生物学报, 2013,4(4):381-385.
doi: 10.3969/j.issn.1009-1823.2013.04.016 |
| [24] | 芦笛, 杨清, 陆巍 . 影响禾谷类作物胚性细胞悬浮系建立的一些因素. 植物生理学通讯, 2007,43(3):399-406. |
| [25] |
陈琰 . 不同抗叶锈小麦品种悬浮细胞系的建立. 保定:河北农业大学, 2005.
doi: 10.7666/d.y740357 |
| [26] |
乔妹 . 小麦“5389”悬浮细胞系的建立和植株再生. 保定:河北农业大学, 2011.
doi: 10.7666/d.y1897526 |
| [27] |
梁军, 魏刚, 吕全 , 等. 印楝细胞悬浮培养系的建立及悬浮培养. 林业科学研究, 2003,16(5):568-574.
doi: 10.3321/j.issn:1001-1498.2003.05.008 |
| [28] | 朱至清 . 植物细胞工程. 北京: 化学工业出版社, 2003: 25-26. |
| [29] | 方文娟, 韩烈保, 曾会明 . 植物细胞悬浮培养影响因子研究. 生物技术通报, 2005(5):11-15. |
| No related articles found! |
|