Crops ›› 2025, Vol. 41 ›› Issue (6): 100-111.doi: 10.16035/j.issn.1001-7283.2025.06.013
Previous Articles Next Articles
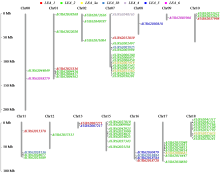
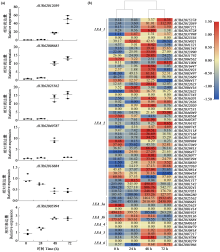
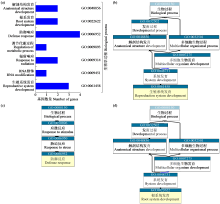
Identification and Expression Analysis of LEA Gene Family in Chenopodium quinoa Willd. under Salt Stress
Zhang Yemeng1,2( ), Lu Kexin1, Mao Wenyu1, Zhang Xiaoyan1, Li Wenjing1, Yang Xiu1
), Lu Kexin1, Mao Wenyu1, Zhang Xiaoyan1, Li Wenjing1, Yang Xiu1
- 1
School of Life Science and Bioengineering ,Jining University Qufu 273155, Shandong, China
2Northwest Institute of Plateau Biology ,Chinese Academy of Sciences Xining 810000, Qinghai, China
| [1] |
Miyazaki K, Ohkubo Y, Yasui H, et al. Overexpression of rice OsLEA5 relieves the deterioration in seed quality caused by high- temperature stress. Plant Biotechnology, 2021, 38(3):367-371.
doi: 10.5511/plantbiotechnology.21.0603a pmid: 34782824 |
| [2] |
Vidović M, Battisti I, Pantelić A, et al. Desiccation tolerance in Ramonda serbica panc.: an integrative transcriptomic, proteomic, metabolite and photosynthetic study. Plants, 2022, 11(9):1199.
doi: 10.3390/plants11091199 |
| [3] |
Dure L Ⅲ, Greenway S C, Galau G A. Developmental biochemistry of cottonseed embryogenesis and germination: changing messenger ribonucleic acid populations as shown by in vitro and in vivo protein synthesis. Biochemistry, 1981, 20(14):4162-4168.
doi: 10.1021/bi00517a033 pmid: 7284317 |
| [4] |
Huang R L, Xiao D, Wang X, et al. Genome-wide identification,evolutionary and expression analyses of LEA gene family in peanut (Arachis hypogaea L.). BMC Plant Biology, 2022, 22(1):155.
doi: 10.1186/s12870-022-03462-7 |
| [5] |
Jia C P, Guo B, Wang B K, et al. The LEA gene family in tomato and its wild relatives: genome-wide identification, structural characterization, expression profiling, and role of SlLEA6 in drought stress. BMC Plant Biology, 2022, 22(1):596.
doi: 10.1186/s12870-022-03953-7 |
| [6] |
Singh K K, Graether S P. The in vitro structure and functions of the disordered late embryogenesis abundant three proteins. Protein Science, 2021, 30(3):678-692.
doi: 10.1002/pro.4028 pmid: 33474748 |
| [7] | Furuki T, Sakurai M. Group 3 LEA protein model peptides protect enzymes against desiccation stress. Acta Biochimica et Biophysica Sinica, 2016, 1864(9):1237-1243. |
| [8] |
Nadarajah K K. ROS homeostasis in abiotic stress tolerance in plants. International Journal of Molecular Sciences. 2020, 21(15):5208.
doi: 10.3390/ijms21155208 |
| [9] |
Zhou C C, Niu S H, El-Kassaby Y A, et al. Genome-wide identification of late embryogenesis abundant protein family and their key regulatory network in Pinus tabuliformis cold acclimation. Tree Physiology, 2023, 43(11):1964-1985.
doi: 10.1093/treephys/tpad095 |
| [10] |
Yang Z F, Mu Y H, Wang Y Q, et al. Characterization of a novel TtLEA2 gene from Tritipyrum and its transformation in wheat to enhance salt tolerance. Frontiers in Plant Science, 2022, 13:830848.
doi: 10.3389/fpls.2022.830848 |
| [11] |
Cheng Z H, Zhang X M., Yao W J. et al. Genome-wide search and structural and functional analyses for late embryogenesis- abundant (LEA) gene family in poplar. BMC Plant Biology, 2021, 21(1):110.
doi: 10.1186/s12870-021-02872-3 |
| [12] |
Hinojosa L, González J, Barrios-Masias F, et al. Quinoa abiotic stress responses: a review. Plants, 2018, 7(4):106.
doi: 10.3390/plants7040106 |
| [13] |
Younis I Y, Sedeek M S, Essa A F, et al. Exploring geographic variations in quinoa grains: unveiling anti-alzheimer activity via GC-MS, LC-QTOF-MS/MS, molecular networking, and chemometric analysis. Food Chemistry, 2025, 465:141918.
doi: 10.1016/j.foodchem.2024.141918 |
| [14] |
Jarvis D E, Ho Y S, Lightfoot D J, et al. The genome of Chenopodium quinoa. Nature, 2017, 542:307-312.
doi: 10.1038/nature21370 |
| [15] |
Zhao Y Y, Hao Y P, Dong Z Y, et al. Identification and expression analysis of LEA gene family members in pepper (Capsicum annuum L.). FEBS Open Bio, 2023, 13(12):2246-2262.
doi: 10.1002/feb4.v13.12 |
| [16] | 贾冰晨, 王宇, 张东亮, 等. 藜麦内参基因筛选及盐胁迫相关基因表达分析. 烟台大学学报(自然科学与工程版), 2020, 33(3):283-288. |
| [17] |
Zhao X X, Wang S Y, Guo F G, et al. Genome-wide identification of polyamine metabolism and ethylene synthesis genes in Chenopodium quinoa Willd. and their responses to low-temperature stress. BMC Genomics, 2024, 25(1):370.
doi: 10.1186/s12864-024-10265-7 |
| [18] |
Wang Q, Lei X J, Wang Y H, et al. Genome-wide identification of the LEA gene family in Panax ginseng: evidence for the role of PgLEA2-50 in plant abiotic stress response. Plant Physiology and Biochemistry, 2024, 212:108742.
doi: 10.1016/j.plaphy.2024.108742 |
| [19] |
Chen Y K, Li C H, Zhang B, et al. The role of the late embryogenesis-abundant (LEA) protein family in development and the abiotic stress response: a comprehensive expression analysis of potato (Solanum tuberosum). Genes, 2019, 10(2):148.
doi: 10.3390/genes10020148 |
| [20] |
Li Y F, Xu Y H, Ma Z W. Comparative analysis of the exon- intron structure in eukaryotic genomes. Yangtze Medicine, 2017, 1(1):50-64.
doi: 10.4236/ym.2017.11006 |
| [21] |
Hibshman J D, Goldstein B. LEA motifs promote desiccation tolerance in vivo. BMC Biology, 2021, 19(1):263.
doi: 10.1186/s12915-021-01176-0 pmid: 34903234 |
| [22] |
Chen Y, Shen J, Zhang L, et al. Nuclear translocation of OsMFT 1 that is impeded by OsFTIP1 promotes drought tolerance in rice. Molecular Plant, 2021, 14(8):1297-1311.
doi: 10.1016/j.molp.2021.05.001 |
| [23] |
Lu Y, Sun J, Yang Z M, et al. Genome-wide identification and expression analysis of glycine-rich RNA-binding protein family in sweet potato wild relative Ipomoea trifida. Gene, 2019, 686:177-186.
doi: S0378-1119(18)31187-9 pmid: 30453066 |
| [24] |
Hu M Q, Li Z Q, Lin X J, et al. Comparative analysis of the LEA gene family in seven Ipomoea species, focuses on sweet potato (Ipomoea batatas L.). BMC Plant Biology, 2024, 24:1256.
doi: 10.1186/s12870-024-05981-x |
| [25] |
Jia J S, Ge N, Wang Q Y, et al. Genome-wide identification and characterization of members of the LEA gene family in Panax notoginseng and their transcriptional responses to dehydration of recalcitrant seeds. BMC Genomics, 2023, 24(1):126.
doi: 10.1186/s12864-023-09229-0 |
| [26] |
Zhang Y J, Fan N N, Wen W W, et al. Genome-wide identification and analysis of LEA_2 gene family in alfalfa (Medicago sativa L.) under aluminum stress. Frontiers in Plant Science, 2022, 13:976160.
doi: 10.3389/fpls.2022.976160 |
| [27] | Liu S Y, Zhang L, Sang Y P, et al. Demographic history and natural selection shape patterns of deleterious mutation load and barriers to introgression across Populus genome. Molecular Biology and Evolution, 2022, 39(2):msac008. |
| [1] | Chen Yi, Chen Xiao, Zhang Maoxing, Li Jing, Chang Jingjing, Li Jiawei, Lin Haiqing, Chen Xingping, Deng Xiaoliang, Xie Dasen, Guo Shaolong, Shen Zhongdeng, Zhang Baige. Response of Different Wax Gourd Varieties to Low-Salt Stress and Comprehensive Evaluation of Their Salt Tolerance [J]. Crops, 2025, 41(6): 58-66. |
| [2] | Teng Wen, Ye Fan, Zhou Zhou, Wang Yule, Liu Lijun. Effects of Wheat and Rapeseed Straw Returning on Yield and Quality of Rice under Salt Stress [J]. Crops, 2025, 41(5): 11-18. |
| [3] | Yan Jingrong, Pang Chunhua, Zhang Yongqing, Wu Yueyue, Hou Yuchen, Wang Jiaqi, Qiao Man. Effects of Desulfurized Gypsum and Humic Acid Interaction on Soil and Quinoa Growth in Saline-Alkali Land [J]. Crops, 2025, 41(5): 47-53. |
| [4] | Li Xiaoyu, Huang Jie, Yang Zhao, Chai Jikuan, Yang Farong, Wei Yuming, Liu Wenyu, Bai Weijun. Establishment and Application of a Comprehensive Evaluation System for Ornamental Quinoa Based on Analytic Hierarchy Process [J]. Crops, 2025, 41(3): 70-77. |
| [5] | Zhang Shengchang, Wei Yuming, Ma Lina, Yang Zhao, Liu Wenyu, Huang Jie, Liu Huan, Yang Farong. Effects of Planting Density and Fertilization on Growth Characteristics of Forage Quinoa [J]. Crops, 2025, 41(2): 128-134. |
| [6] | Jing Maoya, Zhang Ziyu, Zhang Meng, He Jiamin, Yan Fanfan, Gao Yanmei, Zhang Yongqing. Effects of Seed Soaking with Salicylic Acid on Seed Germination and Seedling Growth of Quinoa under Salt Stress [J]. Crops, 2025, 41(1): 194-201. |
| [7] | Li Feng, Gao Hongyun, Zhang Chong, Zhang Baoying, Ma Jianfu, Guo Na, Bai Wei, Fang Aiguo, Yang Zhimin, Li Yuan. Effects of Salt Stress on Growth and Physiological Indexes of Oat [J]. Crops, 2024, 40(6): 140-146. |
| [8] | E Lifeng, Xu Jinchong, Chen Xiubin, Quan Jianhua, Hua Jun, Yin Lijuan, Wang Shunqi, Zhao Wenqin. Effects of Exogenous Silicon on Seed Germination and Physiological Characteristics of Brassica pekinensis under Salt Stress [J]. Crops, 2024, 40(6): 212-217. |
| [9] | Ma Lina, Wei Yuming, Wen Lifang, Zhang Xuejian, Yang Zhao, Huang Jie, Zhang Shengchang, Li Xiaoyu, Liu Huan, Yang Farong. Analysis of Agronomic Traits and Nutritional Quality of 22 Quinoa Germplasms in Yuanmou Area of Yunnan Province [J]. Crops, 2024, 40(6): 47-54. |
| [10] | Hou Yuchen, Pang Chunhua, Zhang Yongqing, Kang Shuyu, Wu Yueyue, Yan Jingrong, Wang Jiaqi. Effects of Biochar and Nitrogen Fertilizer on the Physiological Growth Characteristics of Quinoa Seedlings under Saline Alkali Stress [J]. Crops, 2024, 40(4): 240-246. |
| [11] | Gu Huaiying, Hu Shiqin, Zhao Qing, Liu Changhua, Meng Lijun. The Progress on Enhancing Salt Tolerance of Rice by Rhizosphere Microorganisms [J]. Crops, 2024, 40(4): 8-13. |
| [12] | Liu Jianxia, Wang Wenqing, Xue Naiwen, Guo Xuhu, Ma Saiya, Zhu Guofang, Wen Riyu. Chromosome Karyotype Analysis of 14 Quinoa Germplasms from Different Habitats [J]. Crops, 2024, 40(3): 82-89. |
| [13] | He Jiamin, Zhang Yongqing, Zhang Meng, Liang Ping, Wang Dan, Yan Fanfan. Effects of Seed Soaking with Uniconazole on Agronomic and Physiological Characteristics of Quinoa under Saline-Alkali Stress [J]. Crops, 2024, 40(2): 234-241. |
| [14] | Lü Baolian, Yang Yuxin, Cui Licao, Shi Feng, Ma Liang, Kong Xiuying, Zhang Lichao, Ni Zhiyong. Identification of bHLH Family Transcription Factors of Wheat and Expression Analysis under Salt Stress [J]. Crops, 2024, 40(1): 65-72. |
| [15] | Yang Enze, Wang Shuyan, Liu Ruixiang, Shi Fengyuan, Zhang Jinhao, Li Jiana, Li Zhiwei, Guo Zhanbin. Genetic Diversity Analysis of Quinoa Germplasm Resources Based on SRAP [J]. Crops, 2023, 39(6): 79-85. |
|
||