作物杂志,2020, 第4期: 114–120 doi: 10.16035/j.issn.1001-7283.2020.04.016
玉米早期籽粒中强表达启动子的筛选
王莉1,2( ), 王作平2(
), 王作平2( ), 张中保2, 白玲1(
), 张中保2, 白玲1( ), 吴忠义2(
), 吴忠义2( )
)
- 1河南大学省部共建作物逆境适应与改良国家重点实验室,475004,河南开封
2北京市农林科学院北京农业生物技术研究中心/北京市农业基因资源和生物技术重点实验室,100097,北京
Screening of Strongly Expressed Promoters in Immature Maize Kernels
Wang Li1,2( ), Wang Zuoping2(
), Wang Zuoping2( ), Zhang Zhongbao2, Bai Ling1(
), Zhang Zhongbao2, Bai Ling1( ), Wu Zhongyi2(
), Wu Zhongyi2( )
)
- 1State Key Laboratory of Crop Stress Adaption and Improvement, Henan University, Kaifeng 475004, Henan, China
2Beijing Agro-Biotechnology Research Center, Beijing Academy of Agriculture and Forestry Sciences/Beijing Key Laboratory of Agricultural Gene Resources and Biotechnology, Beijing 100097, China
摘要:
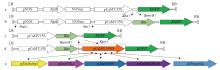
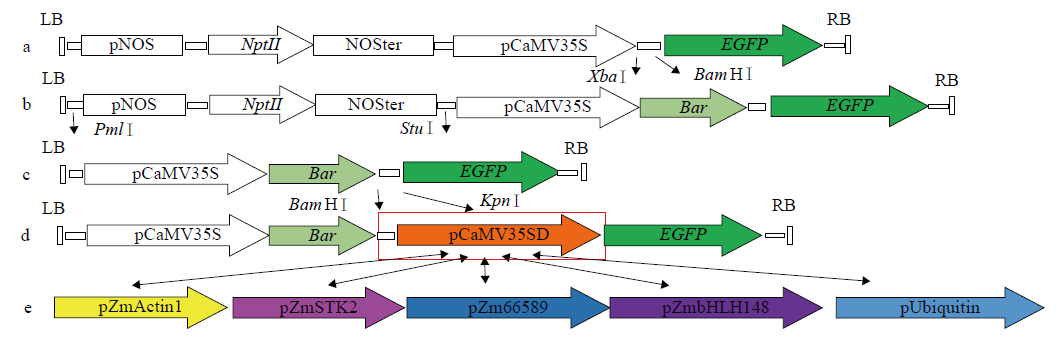
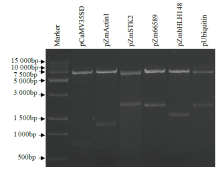
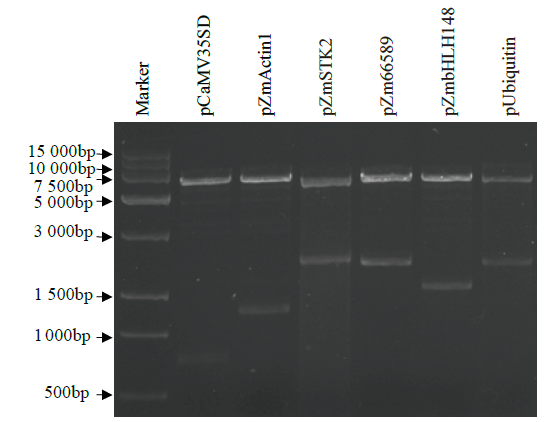
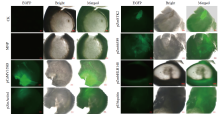
在植物基因表达调控的过程中,启动子作为调控基因表达的顺式元件起着重要作用。为了筛选在玉米早期籽粒中强表达的启动子,选取6个启动子pCaMV35SD、pUbiquitin、pZmActin1、pZmSTK2、pZm66589和pZmbHLH148,分别构建EGFP表达载体并转染玉米原生质体,快速验证载体功能构建的正确性;同时采用花粉磁转染法将6个表达载体导入玉米自交系郑58中,并对不同启动子驱动的EGFP载体在授粉后48h玉米籽粒中的荧光强度和荧光检出率进行观察和统计分析。结果表明,6个启动子驱动EGFP表达载体构建正确;pCaMV35SD启动子驱动EGFP表达载体的荧光最强,6个启动子驱动EGFP表达载体由强到弱依次为pCaMV35SD>pZmSTK2>pZm66589>pZmbHLH148>pUbiquitin>pZmActin1,其荧光检出率分别为27.17%、27.17%、29.83%、23.84%、13.40%和30.57%。
| [1] | 戴景瑞, 鄂立柱 . 我国玉米育种科技创新问题的几点思考. 玉米科学, 2010,18(1):1-5. |
| [2] | Nuccio M L. Maize. New York: Humana Press, 2018. |
| [3] | 夏江东, 夏平 . 高等植物启动子功能和结构研究进展. 楚雄师范学院学报, 2005,20(3):41-48. |
| [4] | 朱丽萍, 于壮, 邹翠霞 , 等. 植物逆境相关启动子及功能. 遗传, 2010,32(3):229-234. |
| [5] | Odell J T, Nagy F, Chua N H . Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature, 1985,313(28):810-812. |
| [6] | Mc Elroy D, Zhang W G, Cao J , et al. Isolation of an efficient actin promoter for use in rice transformation. The Plant Cell, 1990,2(2):163-171. |
| [7] | Toki S, Takamatsu S, Nojiri C , et al. Expression of a maize ubiquitin gene promoter-bar chimeric gene in transgenic rice plants. Plant Physiology, 1992,100(3):1503-1507. |
| [8] | Bhullar S, Chakravarthy S, Advani S , et al. Strategies for development of functionally equivalent promoters with minimum sequence homology for transgene expression in plants: cis-elements in a novel DNA context versus domain swapping. Plant Physiology, 2003,132(2):988-998. |
| [9] | Bénédicte C, Scollan C, Ross S , et al. Co-silencing of homologous transgenes in tobacco. Molecular Breeding, 2000,6(4):407-419. |
| [10] | Xu L, Ye R J, Zheng Y S , et al. Isolation of the endosperm-specific LPAAT gene promoter from coconut (Cocos nucifera L.) and its functional analysis in transgenic rice plants. Plant Cell Reports, 2010,29(9):1061-1068. |
| [11] | Ye R J, Zhou F, Lin Y J . Two novel positive cis-regulatory elements involved in green tissue-specific promoter activity in rice (Oryza sativa L. ssp.). Plant Cell Reports, 2012,31(7):1159-1172. |
| [12] | Chen L, Jiang B J, Wu C X , et al. GmPRP2 promoter drives root-preferential expression in transgenic Arabidopsis and soybean hairy roots. BMC Plant Biology, 2014,14(1):245-257. |
| [13] | Mande X, Yan L, Zhao Z Q , et al. Isolation and characterization of a green-tissue promoter from common wild rice (Oryza rufipogon Griff.). International Journal of Molecular Sciences, 2018,19(7):2009-2021. |
| [14] | Wang H, Fan M X, Wang G H , et al. Isolation and characterization of a novel pollen-specific promoter in maize (Zea mays L.). Genome, 2017,60(6):485-495. |
| [15] | Xu W Z, Liu W S, Ye R J , et al. A profilin gene promoter from switchgrass (Panicum virgatum L.) directs strong and specific transgene expression to vascular bundles in rice. Plant Cell Reports, 2018,37(4):1-11. |
| [16] | Komarnytsky S, Borisjuk N . Functional analysis of promoter elements in plants. Genetic Engineering, 2003,25:113-141. |
| [17] | Roy S, Choudhury S R, Singh S K , et al. Functional analysis of light-regulated promoter region of AtPolλ gene. Planta, 2012,235(2):411-432. |
| [18] | Lee S C, Kim S H, Kim S R . Drought inducible OsDhn1 promoter is activated by OsDREB1A and OsDREB1D. Journal of Plant Biology, 2013,56(2):115-121. |
| [19] | Tao Y, Wang F T, Jia D M , et al. Cloning and functional analysis of the promoter of a stress-inducible gene (ZmRXO1) in Maize. Plant Molecular Biology Reporter, 2015,33(2):200-208. |
| [20] | Liu X Q, Tian J, Zhou X J , et al. Identification and characterization of promoters specifically and strongly expressed in maize embryos. Plant Biotechnology Journal, 2014,12(9):1286-1296. |
| [21] | Lu X D, Chen D J, Shu D F , et al. The differential transcription network between embryo and endosperm in the early developing maize seed. Plant Physiology, 2013,162(1):440-455. |
| [22] | Yoo S D, Cho Y H, Sheen J . Arabidopsis mesophyll protoplasts:a versatile cell system for transient gene expression analysis. Nature Protocols, 2007,2(7):1565-1572. |
| [23] | Zhao X, Meng Z G, Wang Y , et al. Pollen magnetofection for genetic modification with magnetic nanoparticles as gene carriers. Nature Plants, 2017,3(12):956-964. |
| [24] | Kausch A P, Owen T P, Zachwieja S J , et al. Mesophyll-specific,light and metabolic regulation of the C4 PPCZm1 promoter in transgenic maize. Plant Molecular Biology, 2001,45(1):1-15. |
| [25] | José-Estanyol M, Pérez P, Puigdomènech P . Expression of the promoter of HyPRP,an embryo-specific gene from Zea mays in maize and tobacco transgenic plants. Gene, 2005,356(1):146-152. |
| [26] | Chen X P, Wang Z Y, Gu R L , et al. Isolation of the maize Zpu1 gene promoter and its functional analysis in transgenic tobacco plants. Plant Cell Reports, 2007,26(9):1555-1565. |
| [27] | Srilunchang K O, Krohn N G, Dresselhaus T . DiSUMO-like DSUL is required for nuclei positioning,cell specification and viability during female gametophyte maturation in maize. Development, 2010,137(2):333-345. |
| [28] | 王昌涛, 梁粤, 王欢 , 等. 玉米Ubiquitin启动子的克隆及功能鉴定. 沈阳农业大学学报, 2006,37(1):9-12. |
| [29] | 焦勇, 柳小庆, 江海洋 , 等. 植物组织特异性启动子研究进展. 中国农业科技导报, 2019,21(1):24-34. |
| [1] | 闫丽,杨强,邵宇鹏,李丹丹,王志坤,李文滨. 大豆GmWRI1a基因启动子克隆及序列分析[J]. 作物杂志, 2017, (2): 51–58 |
| [2] | 刘建伟, 陈晓峰, 刘广富, 郭宗端, 李新柱, 胡兆平, 张亮. 大豆CYP78A5基因组织特异性启动子的克隆及表达分析[J]. 作物杂志, 2014, (1): 54–58 |
| [3] | 张红梅, 王国英, 张中东, 等. 农杆菌介导的玉米遗传转化进展[J]. 作物杂志, 2000, (6): 1–4 |
| [4] | 马兴林, 林治安, 许建新, 等. 密度对玉米籽粒及秸秆产量的影响[J]. 作物杂志, 1998, (6): 12–13 |
| [5] | 张效梅, 穆志新, 刘金玉. 黑玉米籽粒的营养成分分析[J]. 作物杂志, 1998, (1): 16–17 |
| [6] | 张泽民, 于正坦, 苗亚琦. 不同年代玉米杂交种籽粒营养成分的分析[J]. 作物杂志, 1997, (4): 32–33 |
| [7] | 杨斌, 陈泽辉. CIMMYT优质蛋白玉米研究概况[J]. 作物杂志, 1995, (4): 8–10 |
| [8] | 刘仁东, 杨秀海, 徐家舜. 我国高油玉米的发展前景展望[J]. 作物杂志, 1995, (3): 1–5 |
| [9] | 石德权. 提高优质蛋白玉米含油量的研究[J]. 作物杂志, 1994, (5): 13–13 |
| [10] | 刘瑞征. 玉米湿储法[J]. 作物杂志, 1992, (2): 39–40 |
|
||