作物杂志,2016, 第1期: 69–75 doi: 10.16035/j.issn.1001-7283.2016.01.013
转G2-EPSPS基因玉米D-3侧翼序列分析与转化体特异性检测方法
郭翠1,2,张维2,余桂容3,周正富2,李亮2,冯帅1,2,陈明2,王劲1,2
- 1 西南科技大学生命科学与工程学院,621010,四川绵阳
2 中国农业科学院生物技术研究所,100081,北京
3 四川省农业科学院生物技术核技术研究所,610066,四川成都
Analysis of the Flanking Sequence and Event-Specific Detection of Transgenic G2-EPSPS Line of Maize
Guo Cui1,2,Zhang Wei2,Yu Guirong3,Zhou Zhengfu2,Li Liang2,Feng Shuai1,2,Chen Ming2,Wang Jin1,2
- 1 Life Science and Engineering College,Southwest University of Science and Technology,Mianyang 621010,Sichuan,China
2 Biotechnology Research Institute,Chinese Academy of Agriculture Sciences,Beijing 100081,China
3 Institute of Biological & Nuclear Technology,Sichuan Academy of Agricultural Sciences,Chengdu 610066,Sichuan,China;
摘要:
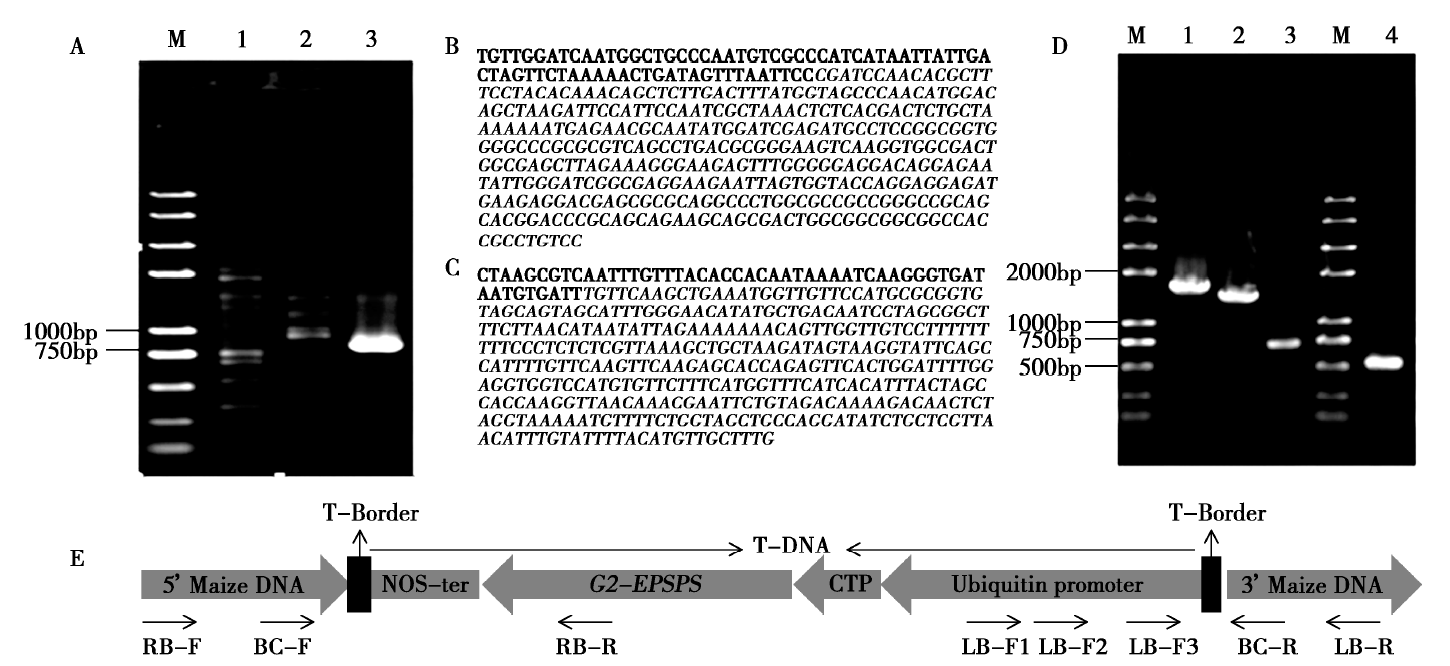
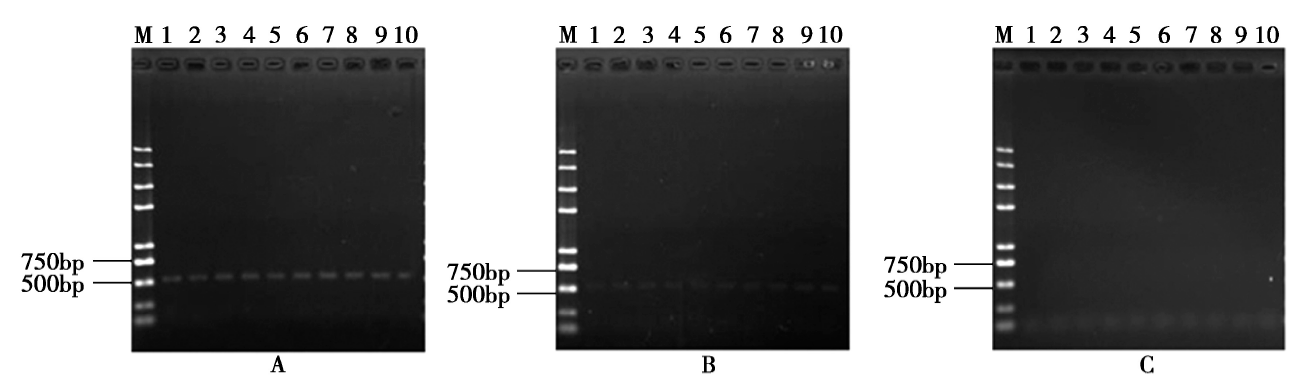
通过一次3'端高效热不对称交互PCR(hiTAIL-PCR)和一次长链PCR扩增方法获得了转抗草甘膦基因(G2-EPSPS)玉米品系D-3的外源DNA插入片段的全DNA序列(T-DNA)及两端侧翼序列,并建立了转化体特异性PCR检测方法。结果显示:T-DNA插入片段全长4 318bp,由一个G2-EPSPS基因的表达盒构成。根据T-DNA 5'、3'端侧翼序列设计引物,建立了转化体特异性检测方法,并分析了该方法的灵敏度以及检出限,研究结果表明,3'端定性PCR检测方法特异性强,灵敏度高,检出极限为每100ng模板量的0.05%。本研究结果对转抗草甘膦外源基因检测和生物安全评价及监管具有重要意义。
| [1] | 申爱娟, 陈松, 周晓盈 , 等. 转基因油菜W-4 T-DNA旁侧序列分析与事件特异性检测. 江苏农业学报, 2014,30(1):10-20. |
| [2] | 许文涛, 杨蓉, 陆姣 , 等. 转基因玉米59122品系的特异性检测. 食品科学, 2011,32(4):139-142. |
| [3] |
张广远, 孙红炜, 李凡 , 等. 转基因玉米MIR162转化事件特异性检测方法及其标准化. 作物学报, 2013,39(7):1141-1147.
doi: 10.3724/SP.J.1006.2013.01141 |
| [4] | 魏俊杰 . 转基因产品的检测方法. 河北农业科学, 2011,15(11):48-49,79. |
| [5] |
Made D, Degner C, Grohmann L . Detection of genetically modified rice:a construct-specific real-time PCR method based on DNA sequences from transgenic Bt rice. European Food Research and Technology, 2006,224(2):271-278.
doi: 10.1007/s00217-006-0467-x |
| [6] | 金芜军, 郝旸, 程红梅 , 等. 用复合PCR方法对6种转基因玉米中的外源DNA进行特异性检测. 农业生物技术, 2005,13(5):562-567. |
| [7] |
Yang L T, Pan A H, Zhang K W , et al. Qualitative and quantitative PCR methods for event-specific detection of genetically modified cotton Mon1445 and Mon531. Transgenic Research, 2005,14:817-831.
doi: 10.1007/s11248-005-0010-z |
| [8] |
Wu G, Wu Y H, Xiao L , et al. Event-specific qualitative and quantitative PCR methods for the detection of genetically modified rapeseed Oxy-235. Transgenic Research, 2008,17:851-862.
doi: 10.1007/s11248-008-9168-5 |
| [9] |
Pan A H, Yang L T, Xu S C , et al. Event-specific qualitative and quantitative PCR detection of MON863 maize based upon the 3'-transgene integration sequence. Cereal Science, 2006,43:250-257.
doi: 10.1016/j.jcs.2005.10.003 |
| [10] |
Yang L T, Xu S C, Pan A H , et al. Event specific qualitative and quantitative polymerase chain reaction detection of genetically modified MON863 maize based on the 5'-transgene integration sequence. Agricultural Food Chemistry, 2005,53:9312-9318.
doi: 10.1021/jf051782o |
| [11] |
Katarina C, Valérie C A, Marie-Noelle F , et al. Detection of nonauthorized genetically modified organisms using differential quantitative polymerase chain reaction:application to 35S in maize. Analytical Biochemistry, 2008,376:189-199.
doi: 10.1016/j.ab.2008.02.013 |
| [12] |
Yang L T, Guo J C, Pan A H , et al. Event-specific quantitative detection of nine genetically modified maizes using one novel standard reference molecule. Agricultural Food Chemistry, 2007,55:15-24.
doi: 10.1021/jf0615754 |
| [13] |
Sissel B R, Marc V, Knut G B , et al. Event specific real-time quantitative PCR for genetically modified Bt11 maize (Zea mays). European Food Research and Technology, 2003,216:347-354.
doi: 10.1007/s00217-002-0653-4 |
| [14] |
Hari K S, Kae-Kang H, Wang S J , et al. Simultaneous detection of eight genetically modified maize lines using a combination of event and construct specific Multiplex-PCR technique. Agricultural Food Chemistry, 2008,56:8962-8968.
doi: 10.1021/jf800501z |
| [15] |
Tigst D, Indira R . Multiplex qualitative PCR assay for identification of genetically modified canola events and real-time event-specific PCR assay for quantification of the GT73 canola event. Food Control, 2008,19:893-897.
doi: 10.1016/j.foodcont.2007.08.020 |
| [16] | 颜静宛, 林琳, 王峰 . 获得基因侧翼序列位点信息的几种扩增方法. 福建农业学报, 2005,20(S1):125-129. |
| [17] | 杨坤, 吴学龙, 朗春秀 , 等. 优化反向PCR法分离转基因油菜外源T-DNA侧翼序列的研究. 安徽农业科学, 2010,38(10):5002-5005. |
| [18] |
Liu Y G, Mitsukawa N, Oosumi T , et al. Efficient isolation and mapping of Arabidopsis thaliana T-DNA insert junctions by thermal asymmetric interlaced PCR. Plant Journal, 1995,8:457-463.
doi: 10.1046/j.1365-313X.1995.08030457.x |
| [19] |
Liu Y G, Whittier R F . Thermal asymmetric interlaced PCR:Automatable amplification and sequencing of insert end fragments from P1 and YAC clones for chromosome walking. Genomics, 1995,25:674-681.
doi: 10.1016/0888-7543(95)80010-J |
| [20] |
Sessions A, Burke E, Presting G , et al. A High-through put reverse genetics system. Plant Cell, 2002,14:2985-2994.
doi: 10.1105/tpc.004630 |
| [21] | 郭金英, 张天真 . TAIL-PCR方法研究花粉管通道法转化机理粗探. 河北农业大学学报, 2010,33(4):5-9. |
| [22] |
Liu Y G, Chen Y L . High-efficiency thermal asymmetric interlaced PCR for amplification of unknown flanking sequences. Biotechniques, 2007,43(5):649-654.
doi: 10.2144/000112601 |
| [23] | 徐荣旗, 汪佳妮, 陈捷胤 , 等. 棉花黄萎病菌T-DNA 插入突变体表型特征和侧翼序列分析. 中国农业科学, 2010,43(3):489-496. |
| [24] | 侯娜, 贺辉群, 董美 , 等. 转基因抗虫棉外源DNA插入整合结构分析和转化体特异性检测方法的建立. 分子植物育种, 2012,10(3):317-323. |
| [25] |
Afolabi A S, Worland B, Snape J W , et al. A large-scale study of rice plant stransformed with different T-DNAs provides new insights into locus composition and T-DNA linkage configurations. Theoretical and Applied Genetics, 2004,109:815-826.
doi: 10.1007/s00122-004-1692-y |
| [26] | Tinland B . The integration of T-DNA into plant genomes.Trends in Plant Science, 1996(1):178-184. |
| [27] |
Krizkova L, Hrouda M . Direct repeats of T-DNA integratedin tobacco chromosome:characterization of junction regions. Plant Journal, 1998,16:673-680.
doi: 10.1046/j.1365-313x.1998.00330.x |
| [28] | 李飞武, 李葱葱, 邢珍娟 , 等. 第二代抗草甘膦大豆PCR检测方法研究. 大豆科学, 2009,28(2):296-300. |
| [29] |
Berdal K G, Holst-Jensen A . Roundup ready soybean event-specific rea1-time quantitative PCR assay and estimation of the practical detection and quantification limits in GMO analyses. European Food Research and Technology, 2001,213(6):432-438.
doi: 10.1007/s002170100403 |
| [30] | Christer R N, Knut G B, Arne H J . Characterisation of the 5'-integration site and development of an event-specific real-time PCR assay for NK603 maize from a low starting copy number. European Food Research and Technology, 2004,219(4):421-427. |
| [31] |
袁磊, 孙红炜, 杨崇良 , 等. 转基因玉米MON88017侧翼序列分析及定性 PCR检测. 作物学报, 2010,36(2):361-364.
doi: 10.3724/SP.J.1006.2010.00361 |
| [32] | 潘爱虎, 张大兵, 潘良文 , 等. 转基因抗草甘膦油菜的实用PCR检测方法. 中国农业科学, 2003,36(7):856-860. |
| [1] | 李丽娜,金龙国,谢传晓,刘昌林. 转基因玉米和转基因大豆盲样检测方法[J]. 作物杂志, 2017, (6): 37–44 |
| [2] | 刘新颖,王柏凤,周琳,冯树丹,宋新元. 转cry1Ie基因抗虫玉米IE09S034种植对田间大型土壤动物多样性的影响[J]. 作物杂志, 2016, (1): 62–68 |
| [3] | 韩猛, 王霄汉, 张中保, 等. BADH基因转人玉米自交系的研究[J]. 作物杂志, 2015, (2): 64–69 |
| [4] | 李杰, 郭欣慰, 张中保, 等. 将拟南芥ATNCED3基因导人玉米自交系的研究[J]. 作物杂志, 2014, (1): 58–62 |
| [5] | 赫福霞, 郎志宏, 张杰, 陆伟, 何康来, 黄大昉. 转Bt cry1Ah/2mG2-epsps双价基因抗虫/耐除草剂玉米研究[J]. 作物杂志, 2012, (6): 29–33 |
| [6] | 陈丽娟, 黄从林, 张秀海, 等. CspB基因植物表达载体的构建及转化玉米自交系的研究[J]. 作物杂志, 2011, (3): 70–74 |
| [7] | 杨朝国, 杨方方, 张登峰. 抗除草剂草丁膦转基因玉米后代筛选方法的研究[J]. 作物杂志, 2010, (6): 32–35 |
|