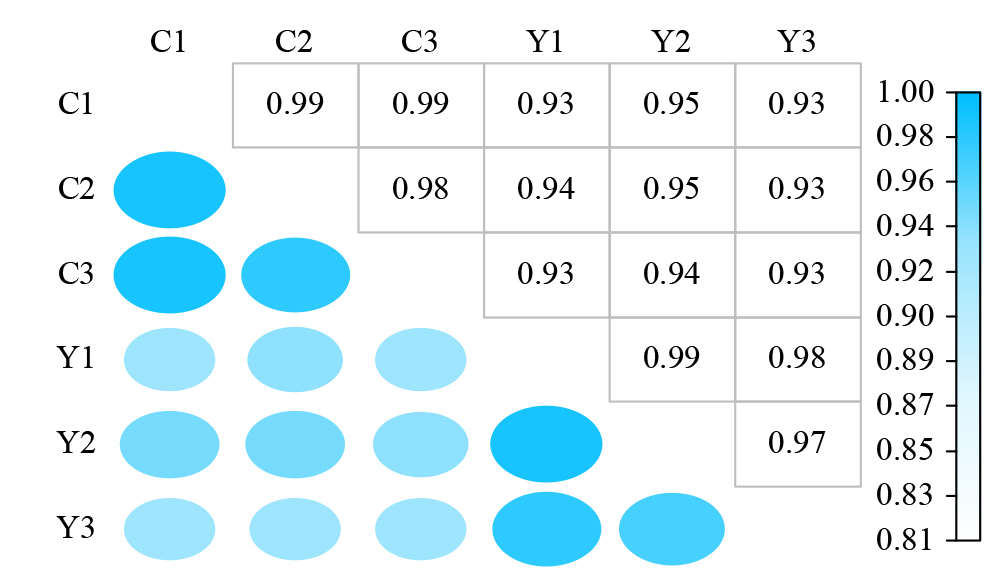
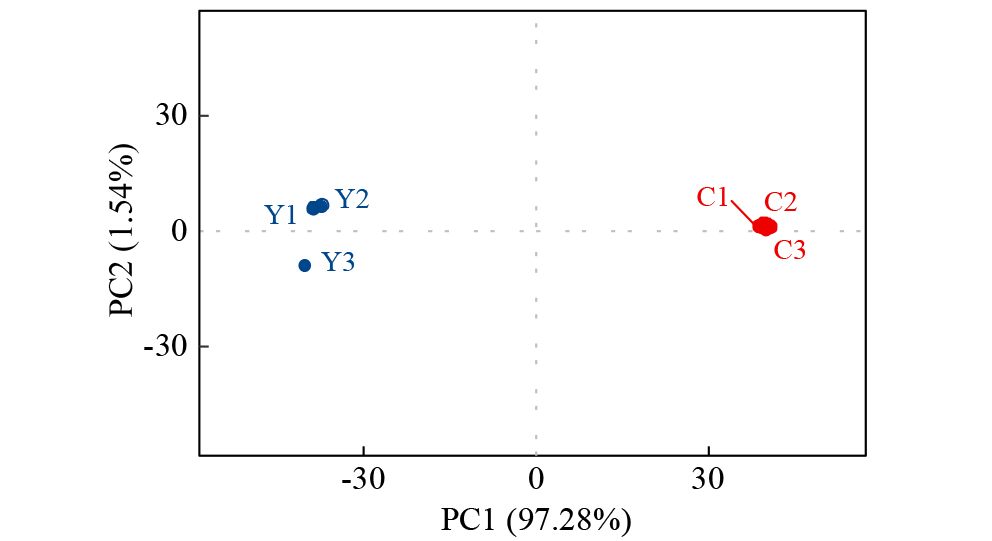
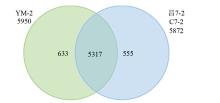
Crops ›› 2025, Vol. 41 ›› Issue (5): 102-112.doi: 10.16035/j.issn.1001-7283.2025.05.014
Previous Articles Next Articles
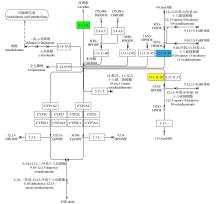
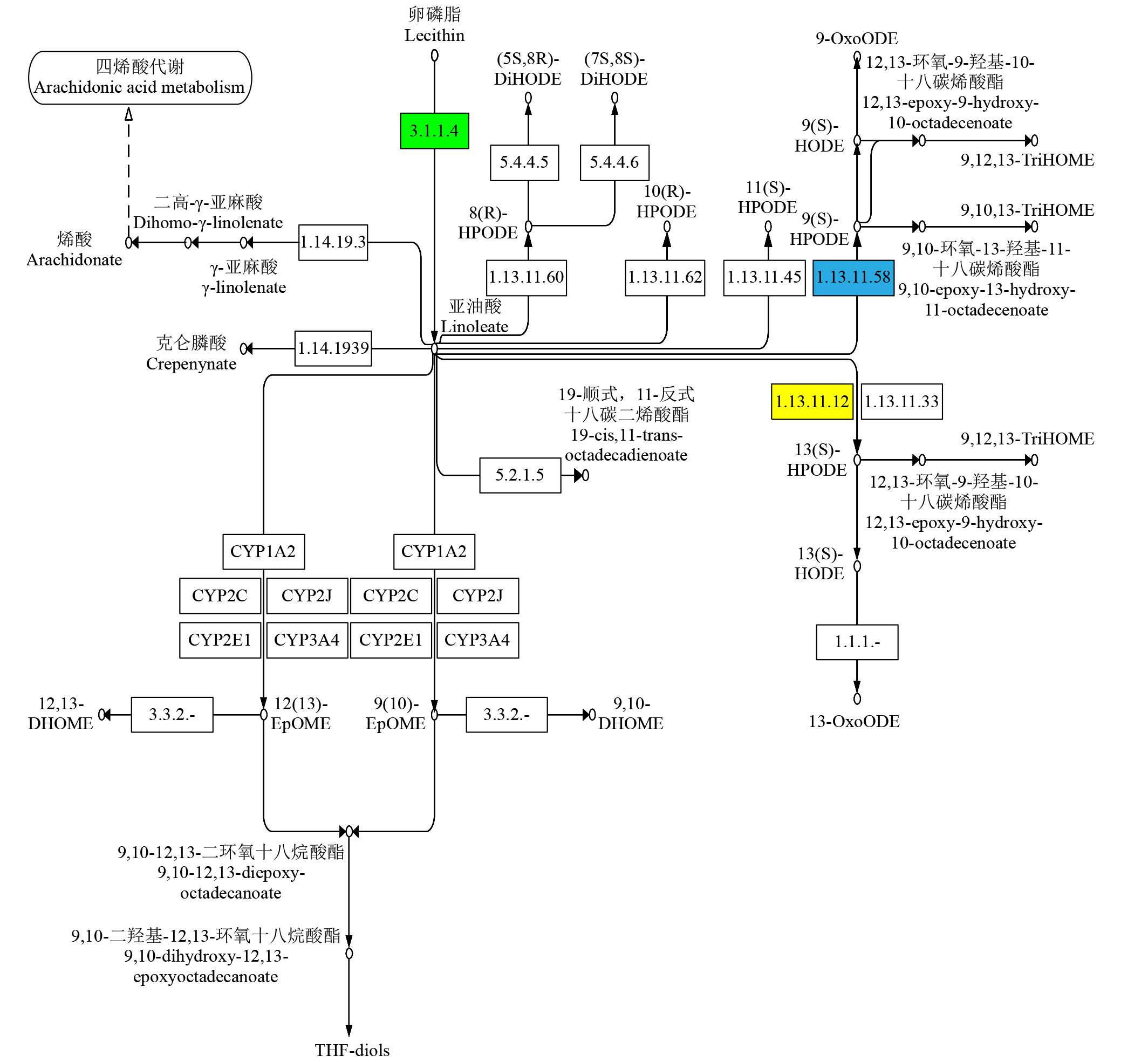
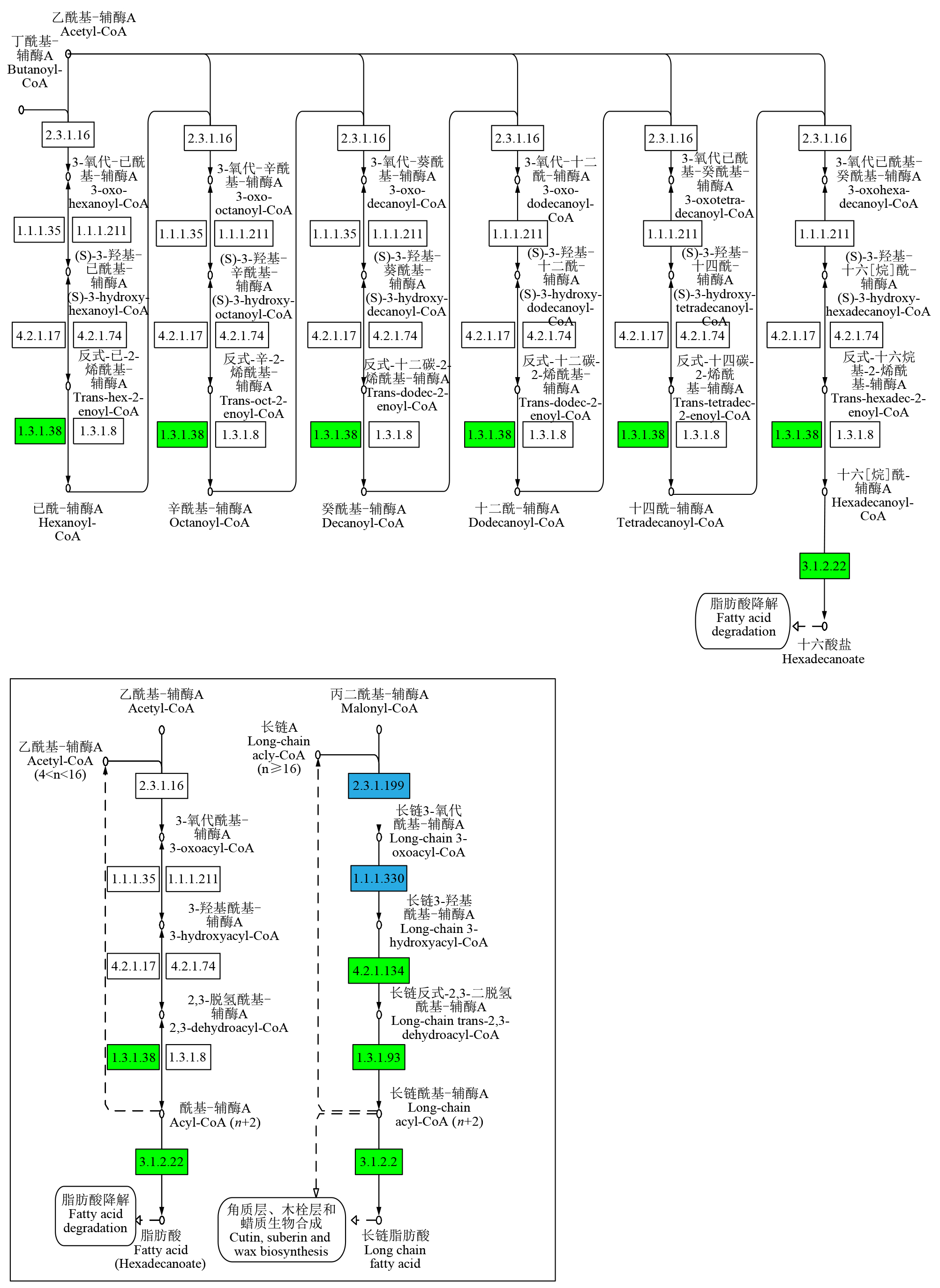
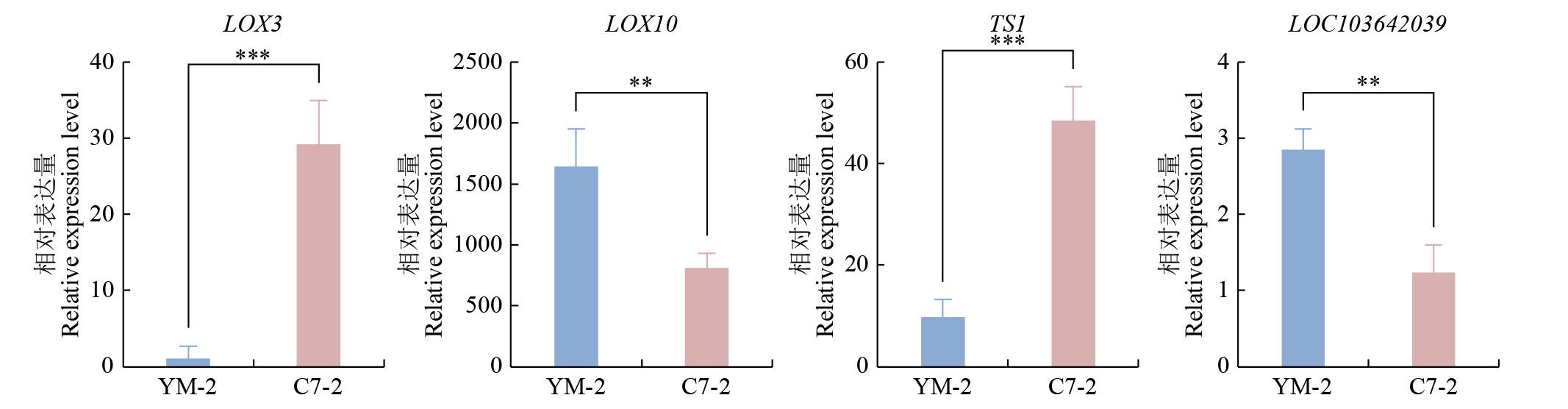
Study on the Mining and Transcriptional Regulation Mechanism of Genes Related to Linoleic Acid Synthesis in Maize
Yang Ke( ), Lai Shangkun, Jin Qian, Wang Weijun, Ding Yidong, Liu Xiaofei(
), Lai Shangkun, Jin Qian, Wang Weijun, Ding Yidong, Liu Xiaofei( )
)
Suqian Institute of Agricultural Sciences ,Jiangsu Academy of Agricultural Sciences Suqian 223800, Jiangsu, China
| [1] | 倪雪梅, 胡玉兰, 李旭, 等. 大胚高油玉米籽粒蛋白质含量相关QTLs定位研究. 分子植物育种,(2024-03-07)[2025-04-18]. https://link.cnki.net/urlid/46.1068.S.20240305.1751.018. |
| [2] | 刘晓飞, 崔小平, 孙宝林, 等. 高亚油酸玉米新组合生长发育特性及亚油酸含量. 江西农业学报, 2019, 31(11):11-16. |
| [3] |
Lacombe R J S. Chouinard-Watkins R, Bazinet R P. Brain docosahexaenoic acid uptake and metabolism. Molecular Aspects of Medicine, 2018, 64:109-134.
doi: S0098-2997(17)30135-8 pmid: 29305120 |
| [4] | 戴永军. 日粮亚油酸对中华绒螯蟹生长、脂氧合酶代谢和风味品质的影响. 南京:南京农业大学, 2022. |
| [5] | 姚池蓓. 不同比例的α-亚麻酸/亚油酸对生长后期草鱼生产性能和肉质的影响及其作用机制. 雅安:四川农业大学, 2023. |
| [6] |
Li H, Peng Z Y, Yang X H, et al. Genome-wide association study dissects the genetic architecture of oil biosynthesis in maize kernels. Nature Genetics, 2013, 45(1):43-50.
doi: 10.1038/ng.2484 pmid: 23242369 |
| [7] | 杨凤萍, 李向龙, 张立全, 等. 双功能去饱和酶AcD12基因改良玉米不饱和脂肪酸成分. 分子植物育种, 2017, 15(12):4805-4812. |
| [8] |
Shen B, Allen W B, Zheng P Z, et al. Expression of ZmLEC1 and ZmWRI1 increases seed oil production in maize. Plant Physiology, 2010, 153(3):980-987.
doi: 10.1104/pp.110.157537 pmid: 20488892 |
| [9] | Wang J B, Liu L, Zhang Q, et al. Genome-wide association analysis-based mining of quality genes related to linoleic and linolenic acids in soybean. Agriculture, 2023, 13:2250. |
| [10] | Di Q, Piersanti A, Zhang Q, et al. Genome-wide association study identifies candidate genes related to the linoleic acid content in soybean seeds. International Journal of Molecular Sciences, 2021, 23(1):454. |
| [11] | Wang F L, Zheng T, et al. Overexpression of miR319a altered oil body morphogenesis and lipid content in Arabidopsis seeds. Plant Molecular Biology Reporter, 2020, 38:531-537. |
| [12] | Wang T, Wang F H, Deng S H, et al. Single-cell transcriptomes reveal spatiotemporal heat stress response in maize roots. Nature Communications, 2025, 16:177. |
| [13] |
Lin Y N, et al. Utilizing transcriptomics and metabolomics to unravel key genes and metabolites of maize seedlings in response to drought stress. BMC Plant Biology, 2024, 24:34.
doi: 10.1186/s12870-023-04712-y pmid: 38185653 |
| [14] |
Song G S, et al. Gene expression and expression quantitative trait loci analyses uncover natural variations underlying the improvement of important agronomic traits during modern maize breeding. The Plant Journal, 2023, 115(3):772-787.
doi: 10.1111/tpj.16260 pmid: 37186341 |
| [15] | 郭新月, 张岚, 张晴, 等. 转录组测序解析油菜素甾醇调控番茄根发育的机制. 河北师范大学学报, 2022, 46(5):512-519. |
| [16] | 王继玥, 余庭跃, 张采波.玉米转录组学研究进展. 华北农学报 , 2014,29(增):10-15. |
| [17] | Luo M J, Shi Y X, et al. Genetic basis of the oil biosynthesis in ultra-high-oil maize grains with an oil content exceeding 20%. Front Plant Science, 2023, 14:1168216. |
| [18] | Pouvreau B, Baud S, Vernoud V, et al. Duplicate maize Wrinkled1 transcription factors activate target genes involved in seed oil biosynthesis. Plant Physiology, 2011, 156(2):674-686. |
| [19] | Kanehisa M, Araki M, Goto S, et al. KEGG for linking genomes to life and the environment. Nucleic Acids Research,2008,36:D480-D484. |
| [20] | Ogunola O F, Hawkins L K, Mylroie E, et al. Characterization of the maize lipoxygenase gene family in relation to aflatoxin accumulation resistance. PLoS ONE, 2017, 12(7):e0181265. |
| [21] |
Acosta I F, Laparra H, Romero S P, et al. tasselseed1 is a lipoxygenase affecting jasmonic acid signaling in sex determination of maize. Science, 2009, 323(5911):262-265.
doi: 10.1126/science.1164645 pmid: 19131630 |
| [22] | Xu Y, Yan F, Liu Y J, et al. Quantitative proteomic and lipidomics analyses of high oil contentGmDGAT1-2 transgenic soybean illustrate the regulatory mechanism of lipoxygenase and oleosin. Plant Cell Report, 2021, 40(12):2303-2323. |
| [23] |
Pan R H, Liu J, Wang S S, et al. Peroxisomes: versatile organelles with diverse roles in plants. New Phytologist, 2020, 225(4):1410-1427.
doi: 10.1111/nph.16134 pmid: 31442305 |
| [24] |
Braun D M. Phloem loading and unloading of sucrose: what a long, strange trip from source to sink. Annual Review of Plant Biology, 2022, 73:553-584.
doi: 10.1146/annurev-arplant-070721-083240 pmid: 35171647 |
| [25] |
Feussner I, Wasternack C. The lipoxygenase pathway. Annual Review of Plant Biology, 2002, 53:275-297.
pmid: 12221977 |
| [26] | Gao X Q, Starr J, Göbel C, et al. Maize 9-lipoxygenase ZmLOX3 controls development, root-specific expression of defense genes, and resistance to root-knot nematodes. Molecular Plant-Microbe Interactions, 2008, 21(1):98-109. |
| [27] | Gao X Q, Brodhagen M, Isakeit T, et al. Inactivation of the lipoxygenase ZmLOX3 increases susceptibility of maize to Aspergillus spp.. Molecular Plant-Microbe Interactions, 2009, 22(2):222-231. |
| [1] | Zhou Tingfang, Li Ran, Liu Qianqian, Zhang Ze, Wang Zhenhua, Ma Baoxin, Lu Ming, Zhang Lin, Han Yehui, Yang Bo, Li Mingshun, Zhang Degui, Weng Jianfeng, Yong Hongjun, Xu Jingyu, Han Jienan, Li Xinhai. Analysis of Salt Tolerance at Germination Stage of 118 Maize Hybrids in Northeast China [J]. Crops, 2025, 41(5): 1-10. |
| [2] | Wang Changliang, Yan Lihui, Chang Jianzhi, Zhang Guohe, Wang Jing, Wang Fenxia, Huang Lihua. Breeding and Innovative Idea of Maize New Variety Xundan 168 [J]. Crops, 2025, 41(5): 142-146. |
| [3] | Li Zhehao, Ji Miyuan, Lü Meng, Ming Bo, Li Shaokun, Zhang Haiyan, Xie Ruizhi. Effects of Different Nitrogen Fertilizer Management Strategies on the Root and Shoot Development of Spring Maize under Long-Term Positioning Conditions [J]. Crops, 2025, 41(4): 135-141. |
| [4] | Wang Xingya, Chen Yuhan, Zhang Mengwen, Sun Linlin, Chen Lirong, Guo Yuqiu, Gong Kuijie. The Effects of ABA Application at Different Stages on Maize Grain Filling and Dehydration [J]. Crops, 2025, 41(4): 173-180. |
| [5] | He Xinchun, Du Hewei, Huang Min. Cloning and Expression Analysis of Maize ZmCaM1 Gene [J]. Crops, 2025, 41(4): 19-28. |
| [6] | Shi Yaxing, Liu Junling, Zhu Guichuan, He Zhongyou, Liu Hui, Fan Yanli, Xu Li, Lu Baishan, Zhao Jiuran, Luo Meijie. Cloning of the du1 Novel Allelic Variant and the Development of Its Molecular Markers [J]. Crops, 2025, 41(4): 41-48. |
| [7] | Chen Minghui, Xu Yu, Huang Zhiqiang, Wang Junqing, Zhang Baoqing. Analysis of the Transcriptome and Jasmonic Acid Signal Transduction in Chewing Cane Leaves under Cold Stress [J]. Crops, 2025, 41(4): 95-103. |
| [8] | Hou Yue, Wang Hongliang, Li Jie, Li Chunjie, Chen Fanjun. Research Advances on the Effects of Cereal/Legume Forage Intercropping on Forage Quality and Nitrogen Uptake [J]. Crops, 2025, 41(3): 1-10. |
| [9] | Jin Zihao, Zhao Wenqing, Wang Fang, Wang Wei, Peng Yunling, Chang Fangguo. Effects of Three Exogenous Plant Growth Regulators on the Cold Tolerance of Maize Seedlings [J]. Crops, 2025, 41(3): 125-132. |
| [10] | Wang Yi, Ren Yongfu, Zhang Zhengpeng, Ding Defang, Zhang Jing, Liu Yihong, Sun Duoxin, Chen Guangrong. The Effects of Different Covering Materials on Soil Environment and Maize Yield in Hexi Irrigation Area [J]. Crops, 2025, 41(3): 149-155. |
| [11] | Hou Nan, Wu Fengjie, Qi Xiangkun, Wang Yufeng, Yang Kejun, Fu Jian. Effects of Different Nitrogen Application Levels on Carbon Metabolism of Waxy Maize during Filling Period in Black Soil Area [J]. Crops, 2025, 41(3): 178-184. |
| [12] | Zhang Zhengjie, Yang Guohua, Guo Ruihong, Cheng Kaihua, Mi Xingwang, Liu Fei. Comprehensive Evaluation of Regional Trials for the Spring Maize in Northwest China Based on DTOPSIS Method and Membership Function Method [J]. Crops, 2025, 41(3): 78-84. |
| [13] | Ren Yongfu, Li Jiayi, Chen Guopeng, Pu Tian, Chen Hong, Wang Xiaochun. Effects of Different Planting Patterns on the Yield and Efficiency of Maize in Strip Intercropping System [J]. Crops, 2025, 41(2): 101-108. |
| [14] | Chang Hongbing, Wang Chen, He Meijing, Cao Ximin, Yu Fengfang, Cao Xiaoliang, Song Wei, Lü Aizhi. Genetic Diversity Analysis of 69 Maize Germplasm Resources Based on SSR Markers [J]. Crops, 2025, 41(2): 47-53. |
| [15] | Li Wenyue, Yu Tao, Cao Shiliang, Ma Xuena, Tang Gui, Gao Li, Yang Gengbin. Genetic Diversity Analysis of 96 Waxy Maize Inbred Lines Based on SNP Chip [J]. Crops, 2025, 41(2): 74-78. |
|
||