Crops ›› 2025, Vol. 41 ›› Issue (4): 126-134.doi: 10.16035/j.issn.1001-7283.2025.04.016
Previous Articles Next Articles
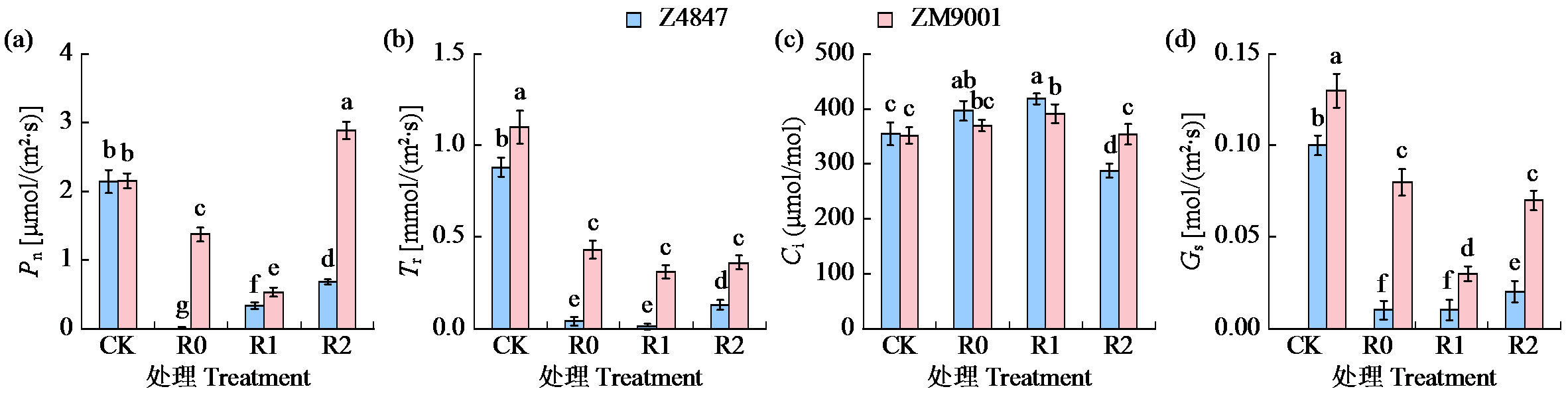
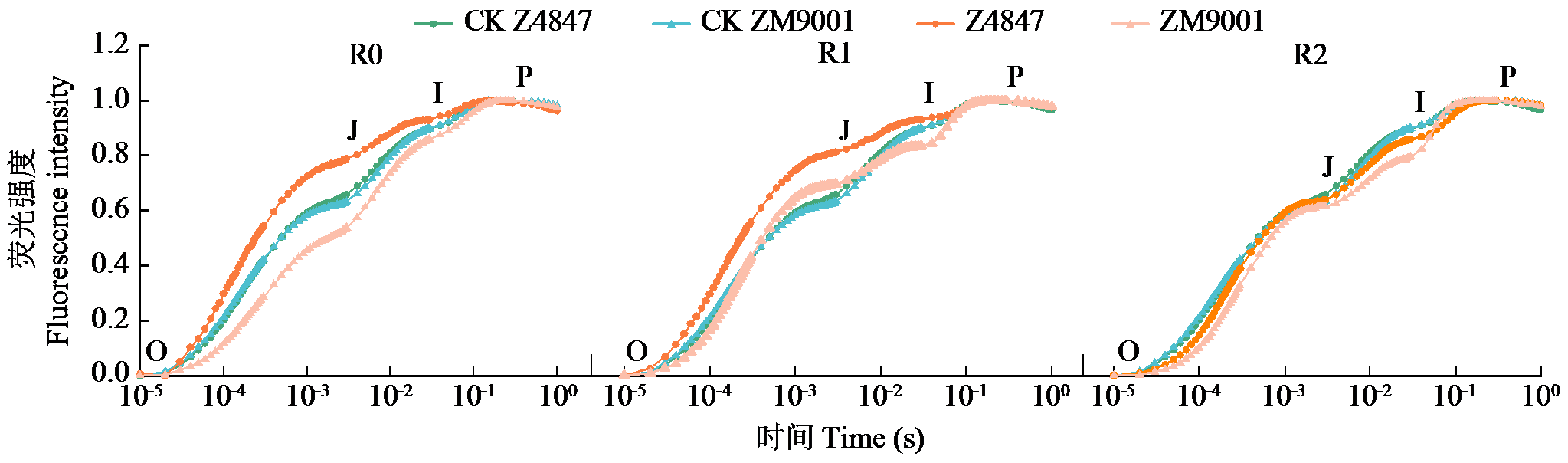
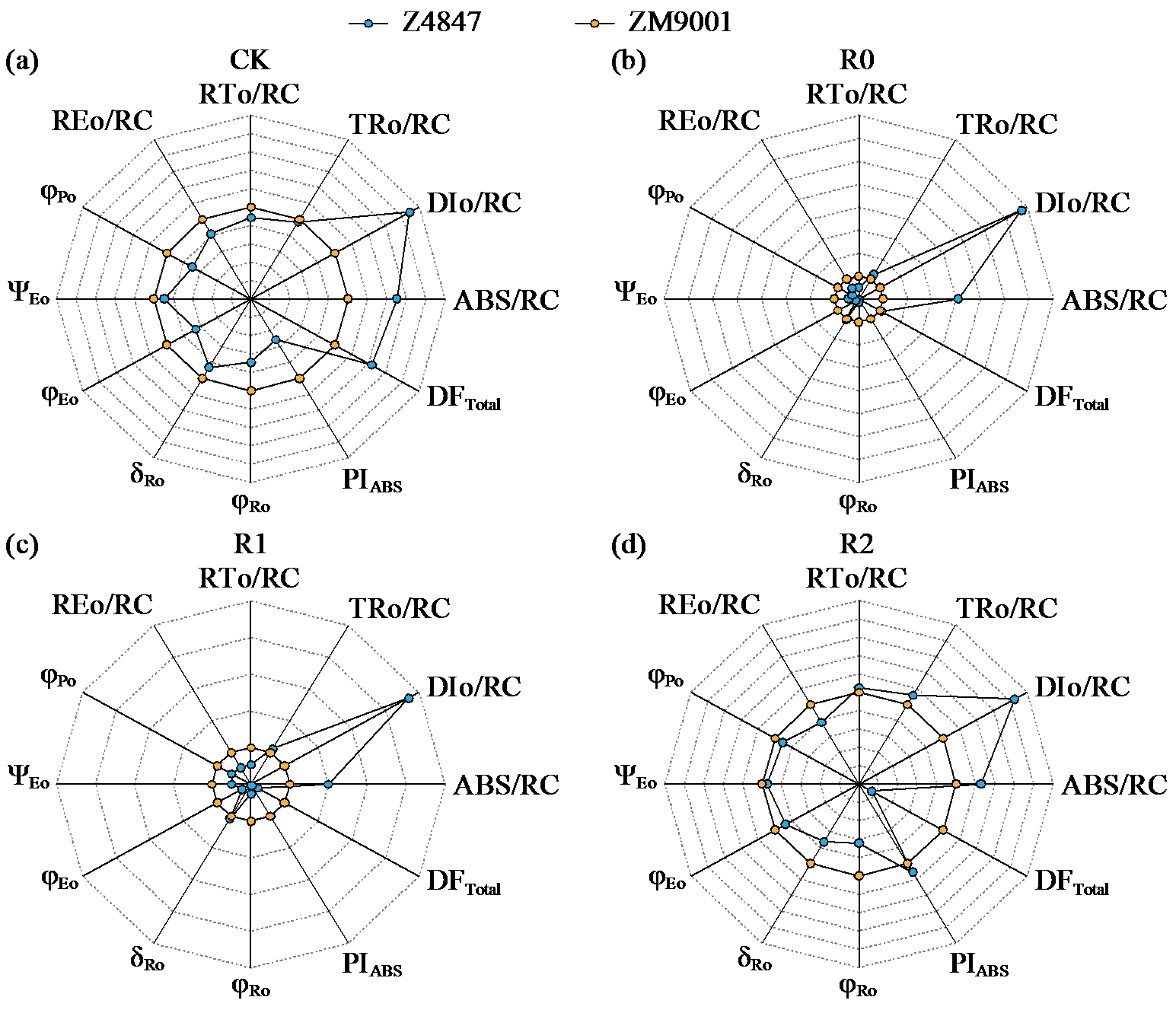
Preliminary Study on the Waterlogging Tolerance Mechanisms in Two Cotton Varieties at Seedling Stage during Waterlogging Stress and Recovery Period
Liu Xuanxuan1( ), Guo Ruishi2, Dong Mengmeng1, Zhu Keying1, Zhu Xiaopin1, Wang Li1, Wang Ning2(
), Guo Ruishi2, Dong Mengmeng1, Zhu Keying1, Zhu Xiaopin1, Wang Li1, Wang Ning2( )
)
- 1College of Life Sciences, Henan Normal University, Xinxiang 453007, Henan, China
2Institute of Cotton Research, Chinese Academy of Agricultural Sciences / State Key Laboratory of Cotton Bio-Breeding and Integrated Utilization, Anyang 455000, Henan, China
| [1] | Zhang Y J, Liu G Y, Dong H Z, et al. Waterlogging stress in cotton: damage, adaptability, alleviation strategies, and mechanisms. The Crop Journal, 2021, 9(2):257-270. |
| [2] | Tian L X, Zhang Y C, Chen P L, et al. How does the waterlogging regime affect crop yield? a global meta-analysis. Frontiers in Plant Science, 2021, 12:634898. |
| [3] | 何小伟, 童金林, 刘怡鑫, 等. 中国洪涝灾害保险:现状、困境与优化. 中国应急管理科学, 2023(11):15-30. |
| [4] | Huang X, Shabala L, Zhang X C, et al. Cation transporters in cell fate determination and plant adaptive responses to a low-oxygen environment. Journal of Experimental Botany, 2022, 73(3):636-645. |
| [5] | 赵可夫. 植物对水涝胁迫的适应. 生物学通报, 2003, 38(12):11-14. |
| [6] | Cao G, Wang X G, Liu Y, et al. Effect of water logging stress on cotton leaf area index and yield. Procedia Engineering, 2012, 28:202-209. |
| [7] | Jiang Z Z, Zhu L, Wang Q Y, et al. Autophagy-related 2 regulates chlorophyll degradation under abiotic stress conditions in Arabidopsis. International Journal of Molecular Sciences, 2020, 21(12):4515. |
| [8] |
Wang P, Liu W C, Han C, et al. Reactive oxygen species: multidimensional regulators of plant adaptation to abiotic stress and development. Journal of Integrative Plant Biology, 2024, 66(3):330-367.
doi: 10.1111/jipb.13601 |
| [9] |
Wang G Y, Ahmad S, Wang Y, et al. Multivariate analysis compares and evaluates drought and flooding tolerances of maize germplasm. Plant Physiology, 2023, 193(1):339-355.
doi: 10.1093/plphys/kiad317 pmid: 37249039 |
| [10] | Dias M C, Pinto D C, Silva A M. Plant flavonoids: chemical characteristics and biological activity. Molecules, 2021, 26(17):5377. |
| [11] | 孙小艳, 邹华文. 外源脱落酸对涝渍胁迫下棉花产量的影响. 湖北农业科学, 2015, 54(14):3341-3342. |
| [12] |
Cisse E H M, Zhang J, Li D D, et al. Exogenous ABA and IAA modulate physiological and hormonal adaptation strategies in Cleistocalyx operculatus and Syzygium jambos under long-term waterlogging conditions. BMC Plant Biology, 2022, 22(1):523.
doi: 10.1186/s12870-022-03888-z pmid: 36357840 |
| [13] |
Yeung E, Bailey-Serres J, Sasidharan R. After the deluge: plant revival post-flooding. Trends in Plant Science, 2019, 24(5):443-454.
doi: S1360-1385(19)30046-9 pmid: 30857921 |
| [14] | Yuan L B, Chen M X, Wang L N, et al. Multi-stress resilience in plants recovering from submergence. Plant Biotechnology Journal, 2022, 21(3):466-481. |
| [15] | León J, Castillo M C, Gayubas B. The hypoxia-reoxygenation stress in plants. Journal of Experimental Botany, 2021, 72(16):5841-5856. |
| [16] | Bange M, Milroy S, Thongbai P. Growth and yield of cotton in response to waterlogging. Field Crops Research, 2004, 88(2/3):129-142. |
| [17] |
Wang H M, Liu X Y, Yang P, et al. Potassium application promote cotton acclimation to soil waterlogging stress by regulating endogenous protective enzymes activities and hormones contents. Plant Physiology and Biochemistry, 2022, 185:336-343.
doi: 10.1016/j.plaphy.2022.06.019 pmid: 35750001 |
| [18] | Pan J W, Sharif R, Xu X W, et al. Mechanisms of waterlogging tolerance in plants: research progress and prospects. Frontiers in Plant Science, 2021, 11:627331. |
| [19] | Niu L F, Jiang F L, Yin J, et al. ROS-mediated waterlogging memory, induced by priming, mitigates photosynthesis inhibition in tomato under waterlogging stress. Frontiers in Plant Science, 2023, 14:1238108. |
| [20] | Ge Q, Zhang Y, Wu J R, et al. Exogenous strigolactone alleviates post-waterlogging stress in grapevine. Plant Physiology and Biochemistry, 2024, 216:109124. |
| [21] |
Flexas J, Medrano H. Drought‐inhibition of photosynthesis in C3 plants: stomatal and non‐stomatal limitations revisited. Annals of Botany, 2002, 89(2):183-189.
doi: 10.1093/aob/mcf027 pmid: 12099349 |
| [22] | Barickman T C, Simpson C R, Sams C E. Waterlogging causes early modification in the physiological performance, carotenoids, chlorophylls, proline, and soluble sugars of cucumber plants. Plants, 2019, 8(6):160. |
| [23] | Kashtoh H, Baek K H. Structural and functional insights into the role of guard cell ion channels in abiotic stress-induced stomatal closure. Plants, 2021, 10(12):2774. |
| [24] | Ahmed S, Nawata E, Sakuratani T. Changes of endogenous ABA and ACC, and their correlations to photosynthesis and water relations in mungbean (Vigna radiata (L.) Wilczak cv. KPS1) during waterlogging. Environmental and Experimental Botany, 2006, 57(3):278-284. |
| [25] | He J X, Wang J, Liang H G. Effects of water stress on photochemical function and protein metabolism of photosystem II in wheat leaves. Physiologia Plantarum, 1995, 93(4):771-777. |
| [26] | Chen H, Li K F, Xue C L, et al. A novel method for non-invasive estimation of primary productivity in aquatic ecosystems using a chlorophyll fluorescence-induced dynamic curve. Frontiers in Microbiology, 2021, 12:682250. |
| [27] | Yeung E, van Veen H, Vashisht D, et al. A stress recovery signaling network for enhanced flooding tolerance in Arabidopsis thaliana. Proceedings of the National Academy of Sciences of the United States of America, 2018, 115(26):6085-6094. |
| [28] |
Małecka A, Konkolewska A, Hanć A, et al. Activation of antioxidative and detoxificative systems in Brassica juncea L. plants against the toxicity of heavy metals. Scientific Reports, 2021, 11(1):22345.
doi: 10.1038/s41598-021-01827-w pmid: 34785730 |
| [29] | Zhang F Q, Wang Y S, Lou Z P, et al. Effect of heavy metal stress on antioxidative enzymes and lipid peroxidation in leaves and roots of two mangrove plant seedlings (Kandelia candel and Bruguiera gymnorrhiza). Chemosphere, 2007, 67(1):44-50. |
| [30] | Singh S, Dubey N K, Tripathi D K, et al. Nitric oxide and hydrogen peroxide mediated regulation of chromium (VI) toxicity in wheat seedlings involves alterations in antioxidants and high affinity sulfate transporter. Plant Science, 2023, 332:111697. |
| [31] | Zhang X M, Duan S G, Xia Y, et al. Transcriptomic, physiological, and metabolomic response of an alpine plant, Rhododendron delavayi, to waterlogging stress and post- waterlogging recovery. International Journal of Molecular Sciences, 2023, 24(13):10509. |
| [32] | Tamang B G, Magliozzi J O, Maroof M S, et al. Physiological and transcriptomic characterization of submergence and reoxygenation responses in soybean seedlings. Plant, Cell & Environment, 2014, 37(10):2350-2365. |
| [33] | Dorion S, Ouellet J C, Rivoal J. Glutathione metabolism in plants under stress: beyond reactive oxygen species detoxification. Metabolites, 2021, 11(9):641. |
| [34] | Hasan M M, Ali M A, Soliman M H, et al. Insights into 28-homobrassinolide (HBR)-mediated redox homeostasis, AsA- GSH cycle, and methylglyoxal detoxification in soybean under drought-induced oxidative stress. Journal of Plant Interactions, 2020, 15(1):371-385. |
| [35] | Umićević S, Kukavica B, Maksimović I, et al. Stress response in tomato as influenced by repeated waterlogging. Frontiers in Plant Science, 2024, 15:1331281. |
| [36] | Zhao N, Li C W, Yan Y J, et al. Comparative transcriptome analysis of waterlogging-sensitive and waterlogging-tolerant Chrysanthemum morifolium cultivars under waterlogging stress and reoxygenation conditions. International Journal of Molecular Sciences, 2018, 19(5):1455. |
| [37] | Xiong X W, Tian H, Fan G, et al. Characterization and transformation of a novel ABI3/VP1-1 gene from hot pepper to enhance waterlogging tolerance. Environmental and Experimental Botany, 2024, 220:105708. |
| [1] | Qu Hongyue, Zhu Linxin, He Yejun, Shao Mingyu, Han Xinran, Jie Yucheng, Xing Hucheng. The Effect of Ramie Extract on Waterlogging Stress of Rapeseed Seed Germination and Pod Stage [J]. Crops, 2025, 41(4): 142-149. |
| [2] | Peng Yuqing, Xiang Shipeng, Chen Zhifeng, Liu Jie, Hu Junjie, Li Yangyang, Zhou Shimin, Zhou Zhi, Li Qiang. Effects of Transplanting Period on Cigar Growth, Photosynthetic Parameters and Activities of Key Enzymes in Carbon and Nitrogen Metabolism [J]. Crops, 2025, 41(4): 259-266. |
| [3] | Wang Li, Zhang Chengjie, Hu Haoran, Ning Liyun, Wu Yifan, Guo Rensong, Zhang Jusong. Effects of Nitrogen Application Rate and Planting Density on Canopy Structure and Photosynthetic Characteristics of Sea Island Cotton [J]. Crops, 2025, 41(3): 116-124. |
| [4] | Hou Xiaomin, Shen Huibo, Dong Shoukun, Yan Feng, Dong Yang, Zhao Fuyang, Li Qingquan, Zuo Yuetao. Physiological Effects of Mepiquat Chloride on Alleviating Drought Stress in Soybean Seedling Leaves [J]. Crops, 2025, 41(3): 133-140. |
| [5] | Zhu Jindi, Zhu Xuegang, Du Wenqing, Qiu Tuoyu, Zhao Xinbin. Effects of Chemical Fertilizer Reduction Combined with Organic Fertilizer Application on Photosynthetic Characteristics, Quality and Yield of Tomatoes Cultivated in Facilities [J]. Crops, 2025, 41(3): 185-189. |
| [6] | Di Na, Zheng Xiqing, Wang Jing, Han Haijun, Li Na. Study on the Difference of Physiological Response of Sunflower to Broomrape Parasitism [J]. Crops, 2025, 41(2): 123-127. |
| [7] | Hou Xiaomin, Yan Feng, Dong Yang, Zhao Fuyang, Li Qingquan, Ji Shengdong, Liu Yue, Lan Ying. Effects of Exogenous Betaine on Germination and Seedling Physiological Characteristics of Foxtail Millet under Drought Stress [J]. Crops, 2025, 41(2): 228-233. |
| [8] | Liu Peiyao, Ran Liping, Yang Jiaqing, Wang Haibo, Xiong Fei, Yu Xurun. Research Progress on Morphogenesis, Physiological Characteristics, and Its External Influencing Factors in Wheat Spike [J]. Crops, 2025, 41(1): 1-9. |
| [9] | Ma Junmei, Dou Min, Liu Di, Yang Xiuhua, Yang Yong, Nian Fuzhao, Liu Yating, Li Yongzhong. Effects of Intercropping Flue-Cured Tobacco and Maize on Rhizosphere Soil Nutrients and Crop Growth [J]. Crops, 2025, 41(1): 227-234. |
| [10] | Li Feng, Gao Hongyun, Zhang Chong, Zhang Baoying, Ma Jianfu, Guo Na, Bai Wei, Fang Aiguo, Yang Zhimin, Li Yuan. Effects of Salt Stress on Growth and Physiological Indexes of Oat [J]. Crops, 2024, 40(6): 140-146. |
| [11] | Wang Yifan, Lin Tao, Wang Dong, Wang Xincui, Zhang Hao, Liu Haijun, Chen Maoguang, Tang Qiuxiang. Effects of Biodegradable Film and Irrigation Quota on Soil Hydrothermal Characteristics in Cotton Field [J]. Crops, 2024, 40(5): 86-95. |
| [12] | Zhang Wei, Wang Qi, Yan Peng, Xu Yanli, Yan Hongdong, Li Guiying, Chen Disu, Jiao Xiaoyan, Lu Lin, Dong Zhiqiang. Effects of PASP-KT-NAA on Leaf Senescence and Yield of Sorghum Populations with Different Densities in Northeast China [J]. Crops, 2024, 40(5): 96-104. |
| [13] | Wang Wenxia, Chang Bokai, Xia Qing, Zhi Hui, Du Jie. Effects of Foliar Spraying Selenium on Physiological Characteristics, Yield and Quality of Flax [J]. Crops, 2024, 40(4): 130-137. |
| [14] | Zhang Lijuan, Qin Yukun, Chen Junying. Effects of Nitrogen Application Rate on Cotton Yield Formation and Nitrogen Utilization Efficiency under Rape-Cotton Double Cropping Straw Returning Condition [J]. Crops, 2024, 40(4): 158-163. |
| [15] | Lü Bo, Ding Liang, Guo Cong, Chen Feng, Zhou Haiping, Wang Xuesong, Dong Xiaolin, Xiang Fayun. Effects of Compound Microbial Fertilizer on Soil Nutrients and Rhizosphere Bacterial Community in Cotton Field [J]. Crops, 2024, 40(4): 209-215. |
|
||