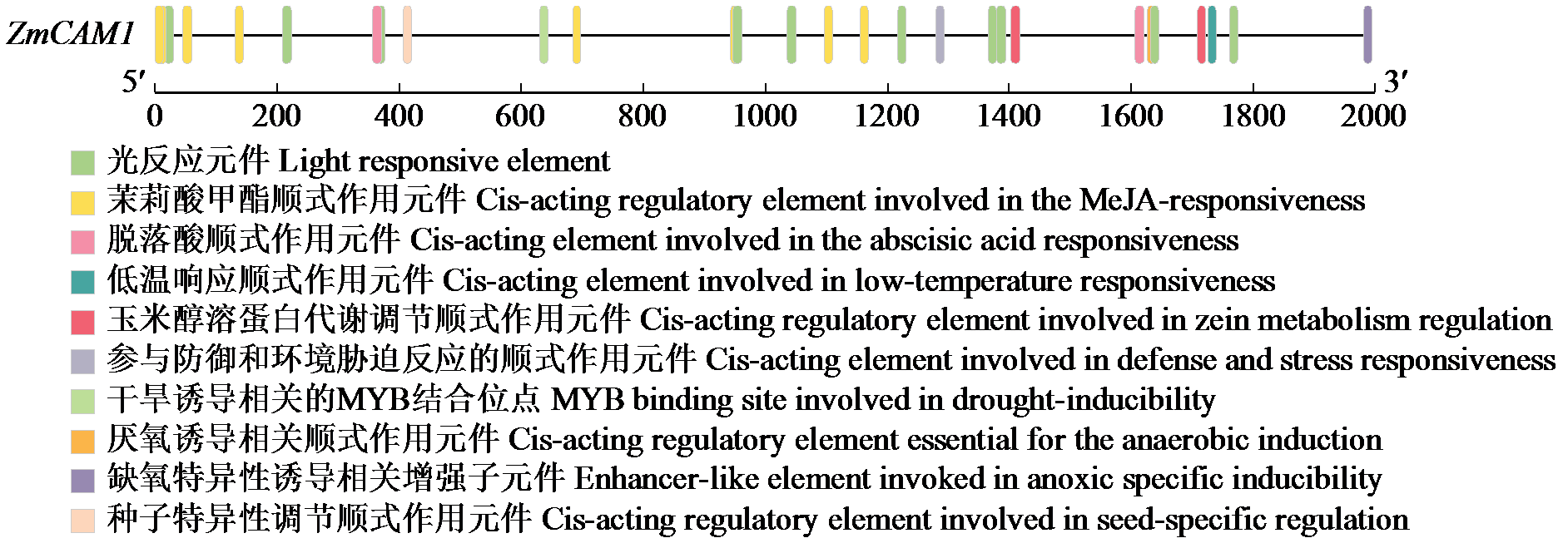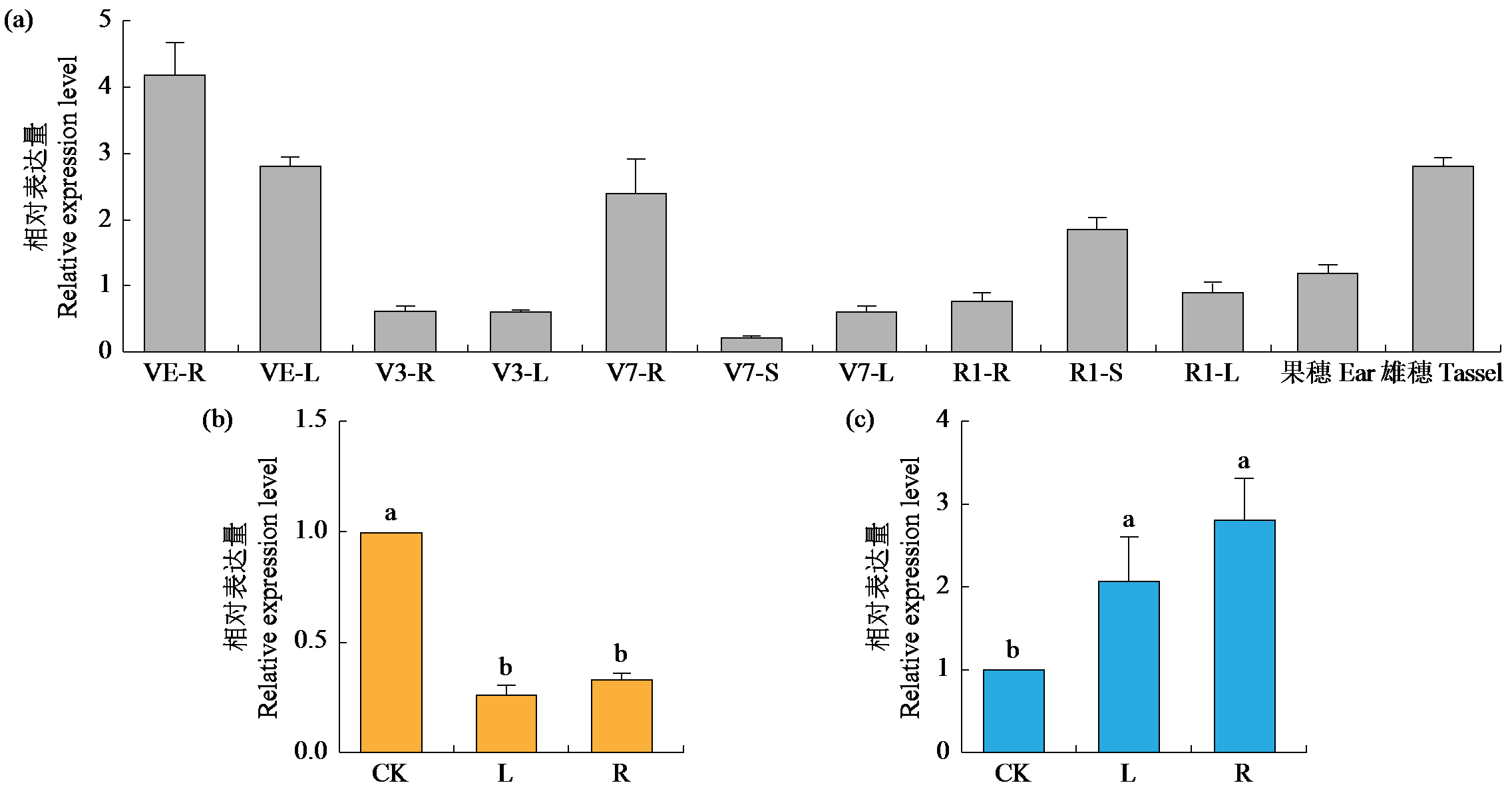Crops ›› 2025, Vol. 41 ›› Issue (4): 19-28.doi: 10.16035/j.issn.1001-7283.2025.04.003
Previous Articles Next Articles
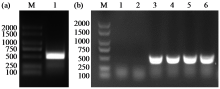
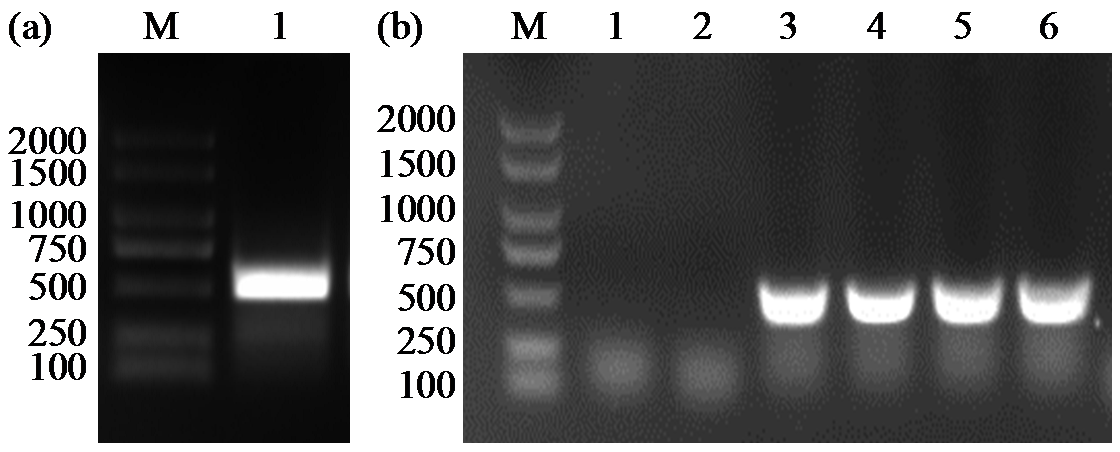
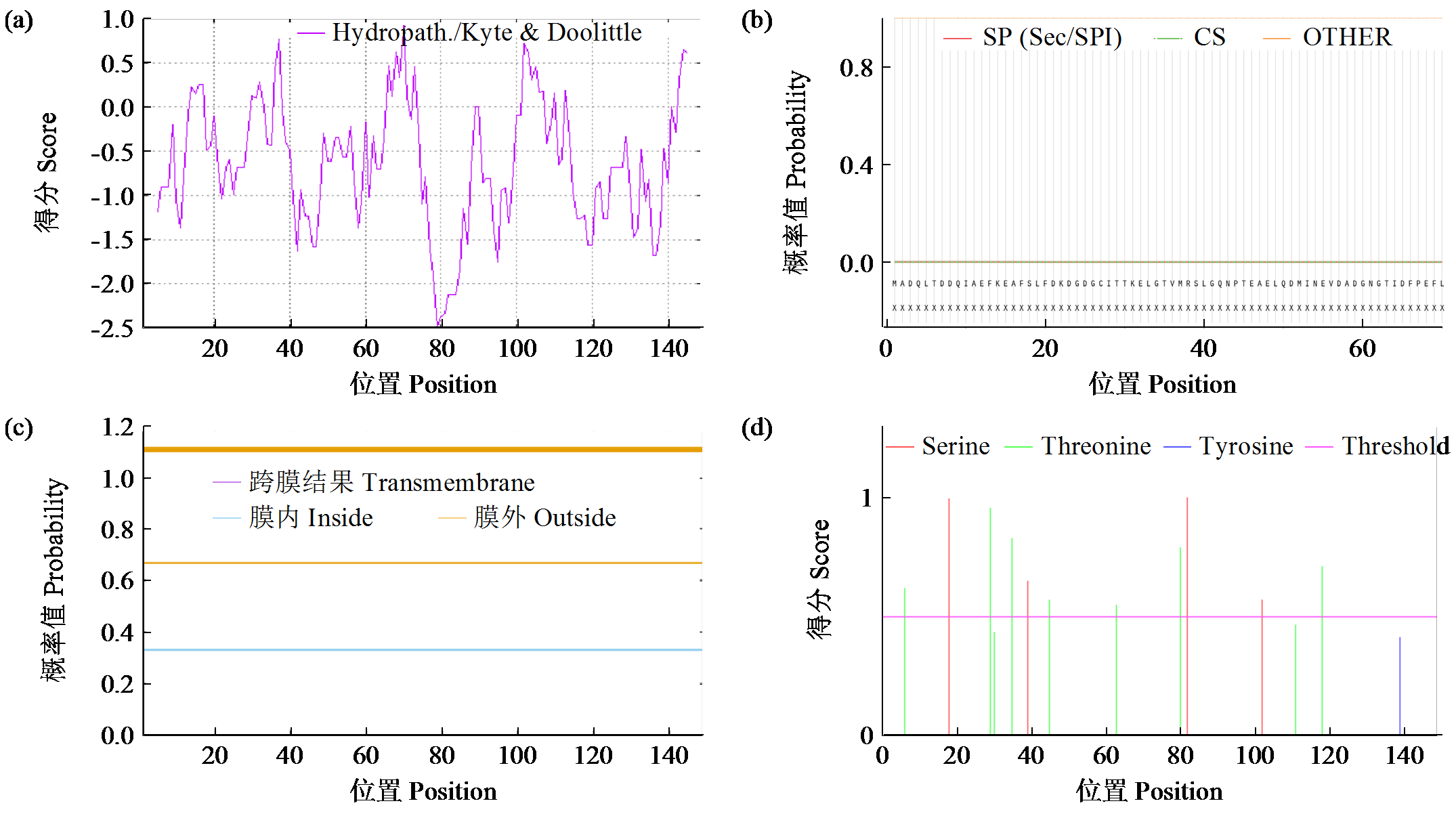
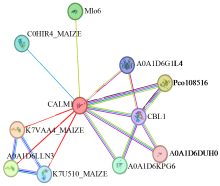
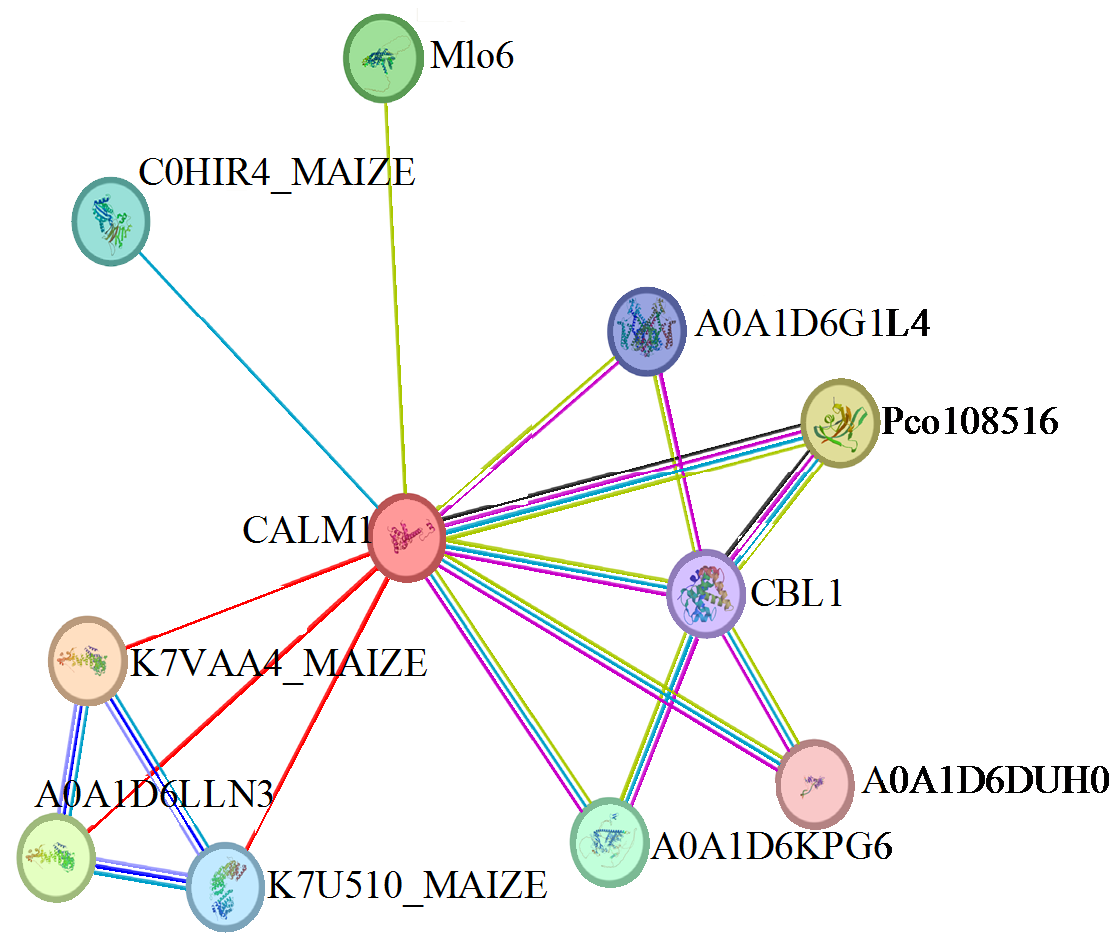
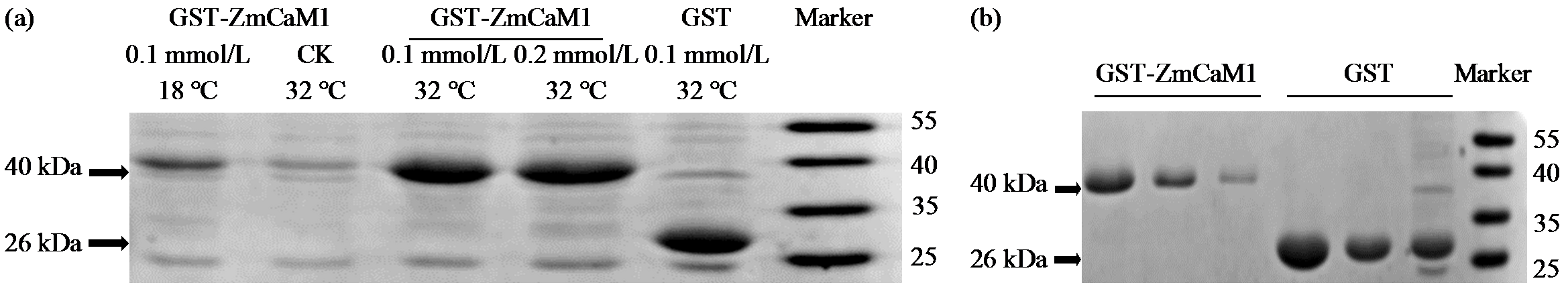
Cloning and Expression Analysis of Maize ZmCaM1 Gene
He Xinchun( ), Du Hewei, Huang Min(
), Du Hewei, Huang Min( )
)
- School of Life Sciences, Yangtze University, Jingzhou 434025, Hubei, China
| [1] | Bothwell J H, Ng C K. The evolution of Ca2+ signalling in photosynthetic eukaryotes. New Phytologist, 2005, 1166(1):21-38. |
| [2] | Azimzadeh J, Nacry P, Christodoulidou A, et al. Arabidopsis TONNEAU1 proteins are essential for preprophase band formation and interact with centrin. The Plant Cell, 2008, 20(8):2146-2159. |
| [3] | 段红沙, 汪春桃, 魏凤菊. 玉米CaM基因家族鉴定及特性分析. 玉米科学, 2008, 31(2):30-38. |
| [4] | 孟凡力, 关珊, 刘晓进. CFP荧光蛋白文库构建及FRET技术筛选钙调素结合蛋白. 中国细胞生物学学报, 2013, 35(12):12-13. |
| [5] |
Vandelle E, Vannozzi A, Wong D, et al. Identification,characterization, and expression analysis of calmodulin and calmodulin-like genes in grapevine (Vitis vinifera) reveal likely roles in stress responses. Plant Physiology and Biochemistry, 2018, 129:221-237.
doi: S0981-9428(18)30254-7 pmid: 29908490 |
| [6] |
Yang T, Poovaiah B W. Molecular and biochemical evidence for the involvement of calcium/calmodulin in auxin action. Journal of Biological Chemistry, 2000, 275(5):3137-3143.
doi: 10.1074/jbc.275.5.3137 pmid: 10652297 |
| [7] |
Xuan Y, Zhou S, Wang L, et al. Nitric oxide functions as a signal and acts upstream of AtCaM3 in thermotolerance in Arabidopsis seedlings. Plant Physiology, 2010, 153(4):1895-1906.
doi: 10.1104/pp.110.160424 pmid: 20576787 |
| [8] | Ranty B, Aldon D, Galaud J P. Plant calmodulins and calmodulin- related proteins: multifaceted relays to decode calcium signals. Plant Signaling & Behavior, 2006, 1(3):96-104. |
| [9] | Weinl S, Kudla J. The CBL-CIPK Ca2+-decoding signaling network: function and perspectives. New Phytologist, 2009, 184(3):517-528. |
| [10] | Boonburapong B, Buaboocha T. Genome-wide identification and analyses of the rice calmodulin and related potential calcium sensor proteins. BMC Plant Biology, 2007, 30:4-7. |
| [11] | Tsai Y C, Koo Y, Delk N A, et al. Calmodulin-related CML24 interacts with ATG4b and affects autophagy progression in Arabidopsis. Plant Journal, 2013, 73(2):325-335. |
| [12] | Mohanta T K, Kumar P, Bae H. Genomics and evolutionary aspect of calcium signaling event in calmodulin and calmodulin- like proteins in plants. BMC Plant Biology, 2003, 17(1):38-39. |
| [13] | Shimada T L, Takano Y, Shimada T, et al. Leaf oil body functions as a subcellular factory for the production of a phytoalexin in Arabidopsis. Plant Physiology, 2014, 164(1):105-118. |
| [14] | 徐渴, 王伟伟, 武宁静, 等. 小麦OFP基因家族鉴定及其低温胁迫的表达分析. 农业生物技术学报, 2005, 29(9):1665-1677. |
| [15] | Yoo J H, Park C Y, Kim J C, et al. Direct interaction of a divergent CaM isoform and the transcription factor, MYB2, enhances salt tolerance in Arabidopsis. Journal of Biological Chemistry, 2005, 280(5):3697-3706. |
| [16] |
DeFalco T A, Chiasson D, Munro K, et al. Characterization of GmCaMK1, a member of a soybean calmodulin-binding receptor- like kinase family. FEBS Letters, 2010, 584(23):4717-4724.
doi: 10.1016/j.febslet.2010.10.059 pmid: 21056039 |
| [17] | Yamaguchi T, Aharon G S, Sottosanto J B, et al. Vacuolar Na+/H+ antiporter cation selectivity is regulated by calmodulin from within the vacuole in a Ca2+- and pH-dependent manner. Proceedings of the National Academy of Sciences of the United States of America, 2005, 102(44):16107-16112. |
| [18] |
Levy M, Wang Q, Kaspi R, et al. Arabidopsis IQD1, a novel calmodulin-binding nuclear protein, stimulates glucosinolate accumulation and plant defense. Plant Journal, 2005, 43(1):79-96.
doi: 10.1111/j.1365-313X.2005.02435.x pmid: 15960618 |
| [19] | Neill S, Barros R, Bright J, et al. Nitric oxide, stomatal closure, and abiotic stress. Journal of Experimental Botany, 2008, 59(2):176-165. |
| [20] | McCormack E, Tsai Y C, Braam J. Handling calcium signaling: Arabidopsis CaMs and CMLs. Trends in Plant Science, 2005, 10(8):383-389. |
| [21] | McCormack E, Braam J. Calmodulins and related potential calcium sensors of Arabidopsis. New Phytologist, 2003, 159(3):585-598. |
| [1] | Li Zhehao, Ji Miyuan, Lü Meng, Ming Bo, Li Shaokun, Zhang Haiyan, Xie Ruizhi. Effects of Different Nitrogen Fertilizer Management Strategies on the Root and Shoot Development of Spring Maize under Long-Term Positioning Conditions [J]. Crops, 2025, 41(4): 135-141. |
| [2] | Wang Xingya, Chen Yuhan, Zhang Mengwen, Sun Linlin, Chen Lirong, Guo Yuqiu, Gong Kuijie. The Effects of ABA Application at Different Stages on Maize Grain Filling and Dehydration [J]. Crops, 2025, 41(4): 173-180. |
| [3] | Shi Yaxing, Liu Junling, Zhu Guichuan, He Zhongyou, Liu Hui, Fan Yanli, Xu Li, Lu Baishan, Zhao Jiuran, Luo Meijie. Cloning of the du1 Novel Allelic Variant and the Development of Its Molecular Markers [J]. Crops, 2025, 41(4): 41-48. |
| [4] | Bu Qing, Yang Kaiqiang, Wang Tian, Shi Si, Xin Guochen, Chen Yong, Dong Zhaoxia, An Jing. Research Progress on the Use of Nanomaterials and the Alleviation of Abiotic Stresses in Crops [J]. Crops, 2025, 41(4): 9-18. |
| [5] | Hou Yue, Wang Hongliang, Li Jie, Li Chunjie, Chen Fanjun. Research Advances on the Effects of Cereal/Legume Forage Intercropping on Forage Quality and Nitrogen Uptake [J]. Crops, 2025, 41(3): 1-10. |
| [6] | Jin Zihao, Zhao Wenqing, Wang Fang, Wang Wei, Peng Yunling, Chang Fangguo. Effects of Three Exogenous Plant Growth Regulators on the Cold Tolerance of Maize Seedlings [J]. Crops, 2025, 41(3): 125-132. |
| [7] | Wang Yi, Ren Yongfu, Zhang Zhengpeng, Ding Defang, Zhang Jing, Liu Yihong, Sun Duoxin, Chen Guangrong. The Effects of Different Covering Materials on Soil Environment and Maize Yield in Hexi Irrigation Area [J]. Crops, 2025, 41(3): 149-155. |
| [8] | Hou Nan, Wu Fengjie, Qi Xiangkun, Wang Yufeng, Yang Kejun, Fu Jian. Effects of Different Nitrogen Application Levels on Carbon Metabolism of Waxy Maize during Filling Period in Black Soil Area [J]. Crops, 2025, 41(3): 178-184. |
| [9] | Zhang Zhengjie, Yang Guohua, Guo Ruihong, Cheng Kaihua, Mi Xingwang, Liu Fei. Comprehensive Evaluation of Regional Trials for the Spring Maize in Northwest China Based on DTOPSIS Method and Membership Function Method [J]. Crops, 2025, 41(3): 78-84. |
| [10] | Ren Yongfu, Li Jiayi, Chen Guopeng, Pu Tian, Chen Hong, Wang Xiaochun. Effects of Different Planting Patterns on the Yield and Efficiency of Maize in Strip Intercropping System [J]. Crops, 2025, 41(2): 101-108. |
| [11] | Chang Hongbing, Wang Chen, He Meijing, Cao Ximin, Yu Fengfang, Cao Xiaoliang, Song Wei, Lü Aizhi. Genetic Diversity Analysis of 69 Maize Germplasm Resources Based on SSR Markers [J]. Crops, 2025, 41(2): 47-53. |
| [12] | Chen Hu, Gao Yuan, Sun Jiameng, Yu Peng, Xiao Hongwu, Zhang Haitao. Identification and Bioinformatics Analysis of the Typical Thioredoxin (TRX) Gene Family in Barley [J]. Crops, 2025, 41(2): 66-73. |
| [13] | Li Wenyue, Yu Tao, Cao Shiliang, Ma Xuena, Tang Gui, Gao Li, Yang Gengbin. Genetic Diversity Analysis of 96 Waxy Maize Inbred Lines Based on SNP Chip [J]. Crops, 2025, 41(2): 74-78. |
| [14] | Sun Yanjie, Wei Guocai, Wu Yuheng, Shi Yunqiang, Shao Yong, Liu Yingrui, Nan Yuantao, Zhang Weiyao. Genetic Diversity Analysis of 100 Maize Germplasm Resources by SNP Markers [J]. Crops, 2025, 41(2): 14-19. |
| [15] | Chu Zhaokang, Wang Shiji, Bi Jianjian, Zhang Lin, Peng Chen, Chen Xiang, Wu Wenming. Effects of Sowing Dates on Yield and Filling Characteristics of Summer Maize in the Central Yangtze-Huaihe Region [J]. Crops, 2025, 41(1): 117-122. |
|
||