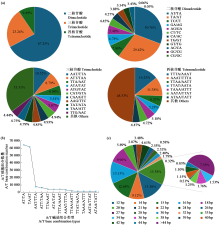
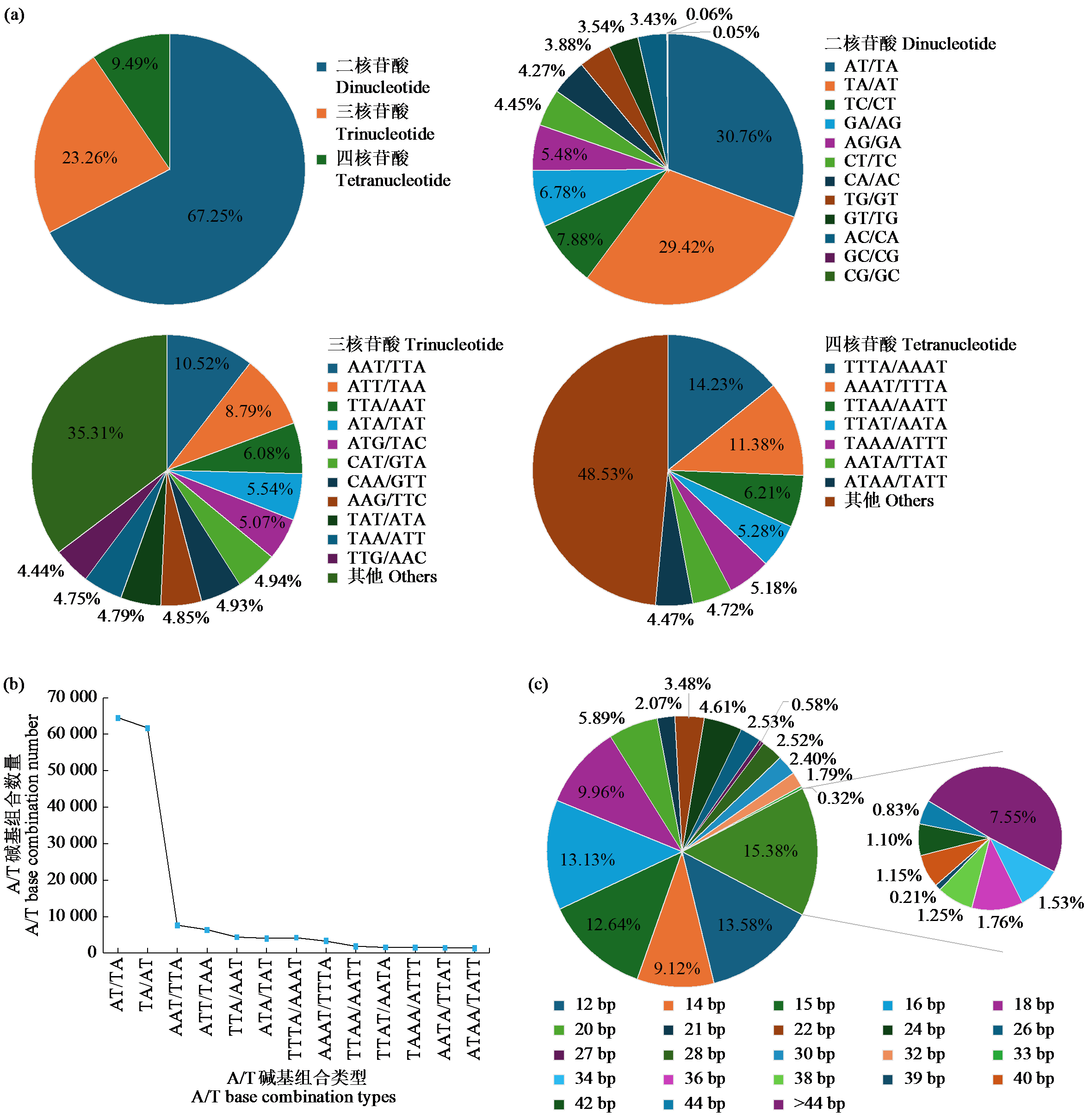
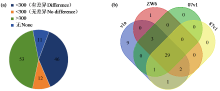
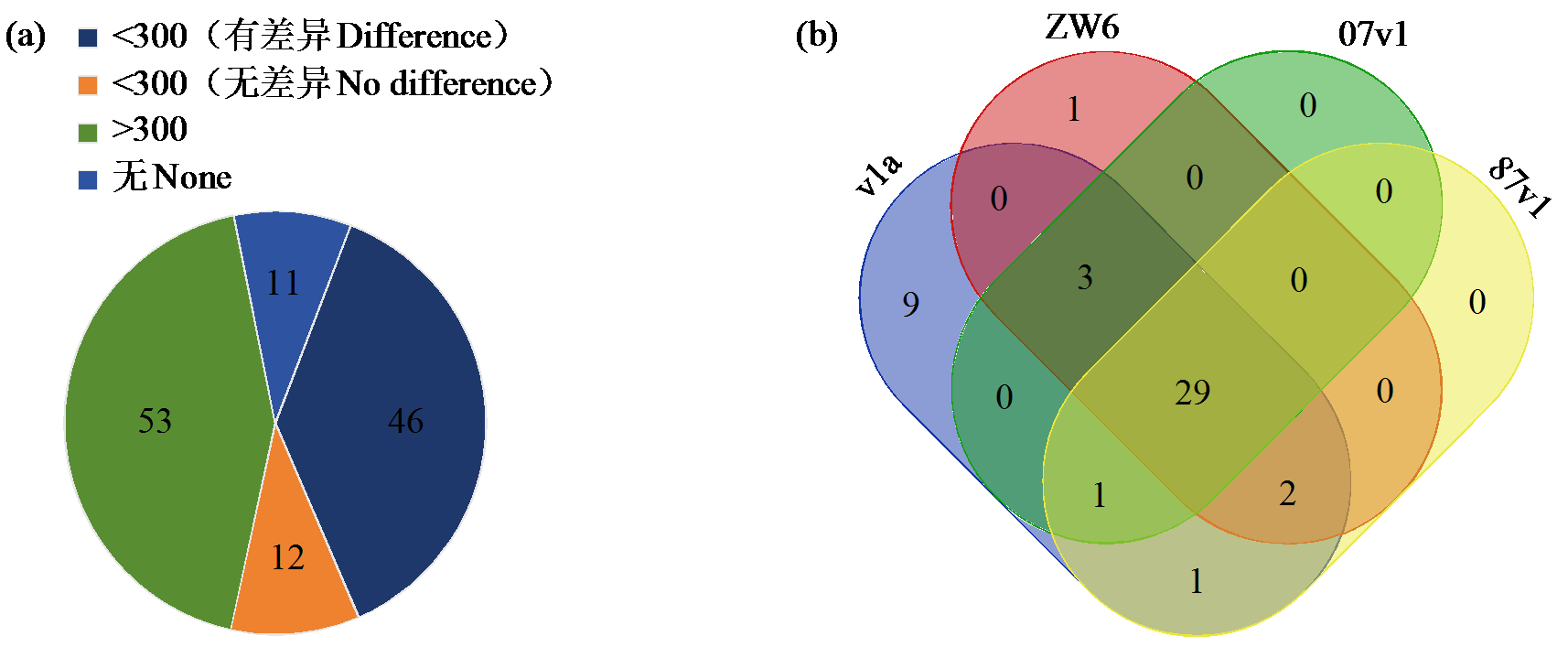
Crops ›› 2025, Vol. 41 ›› Issue (6): 19-27.doi: 10.16035/j.issn.1001-7283.2025.06.003
Previous Articles Next Articles
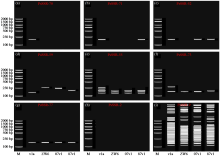
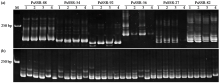
Screening and Fingerprint Construction of SSR Molecular Markers in Pea
Chen Haonan1( ), Zhao Hongyan2,3, Zhang Wenjing2,3, Zhang Qi2,3, Du Jidao2,3, Wang Qirui2,3, Ren Ruoran2,3, Han Yiqiang1,3(
), Zhao Hongyan2,3, Zhang Wenjing2,3, Zhang Qi2,3, Du Jidao2,3, Wang Qirui2,3, Ren Ruoran2,3, Han Yiqiang1,3( )
)
- 1
College of Life Science and Technology ,Heilongjiang Bayi Agricultural University Daqing 163711, Heilongjiang, China
2College of Agriculture ,Heilongjiang Bayi Agricultural University Daqing 163711, Heilongjiang, China
3National Coarse Cereals Engineering Technology Research Center Daqing 163319, Heilongjiang, China
| [1] | 郑卓杰. 中国食用豆类学. 北京: 中国农业出版社, 1997. |
| [2] | 吴星波. 豌豆核心种质资源遗传多样性研究. 重庆:西南大学, 2014. |
| [3] | 曾艳, 郝学财, 董婷, 等. 植物蛋白肉的原料开发、加工工艺与质构营养特性研究进展. 食品工业科技, 2021, 42(3):338-345,350. |
| [4] | 冯晓云, 鲍根生, 刘文辉. 150份豌豆(Pisum sativum L.)种质资源开花期关键性状评价. 中国草地学报, 2023, 45(9):30-37. |
| [5] | 韩毅强, 陈浩楠, 张琦, 等. 一种用于鉴别豌豆品种的分子标记组合、引物组合及其应用:CN202311257283.3. 2024-05-10. |
| [6] | 洪文娟. 石榴种质资源SSR分子标记遗传多样性分析及指纹图谱构建. 北京: 北京林业大学, 2021. |
| [7] | 王红梅, 张正英, 陈玉梁. SSR标记技术及其在植物遗传学中的应用. 西北师范大学学报(自然科学版), 2003, 39(1):113-116. |
| [8] |
Wang Z, Weber J L, Zhong G, et al. Survey of plant short tandem DNA repeats. Theoretical and Applied Genetics, 1994, 88(1):1-6.
doi: 10.1007/BF00222386 pmid: 24185874 |
| [9] |
Bhargava A, Fuentes F F. Mutational dynamics of microsatellites. Molecular Biotechnology, 2010, 44(3):250-266.
doi: 10.1007/s12033-009-9230-4 pmid: 20012711 |
| [10] |
Mathema V B, Nakeesathit S, White N J, et al. Genome-wide microsatellite characteristics of five human Plasmodium species, focusing on Plasmodium malariae and P.ovale curtisi. Parasite, 2020, 27:34.
doi: 10.1051/parasite/2020034 pmid: 32410726 |
| [11] |
Dharajiya D T, Shah A, Galvadiya B P, et al. Genome-wide microsatellite markers in castor (Ricinus communis L.): identification, development, characterization, and transferability in Euphorbiaceae. Industrial Crops and Products, 2020, 151:112461.
doi: 10.1016/j.indcrop.2020.112461 |
| [12] |
Manee M M, Algarni A T, Alharbi S N, et al. Genome-wide characterization and analysis of microsatellite sequences in camelid species. Mammal Research, 2019, 65(16):359-373.
doi: 10.1007/s13364-019-00458-x |
| [13] |
Gupta P K, Varshney R K. The development and use of microsatellite markers for genetic analysis and plant breeding with emphasis on bread wheat. Euphytica, 2000, 113(3):163-185.
doi: 10.1023/A:1003910819967 |
| [14] | 熊登坤, 鲍大鹏, 边银丙. 利用电子PCR分析草菇基因组SSR标记多态性. 微生物学通报, 2014, 41(10):2070-2075. |
| [15] | Jo I H, Han S, Shim D, et al. Complete chloroplast genome of the inverted repeat-lacking species Vicia bungei and development of polymorphic simple sequence repeat markers. Frontiers in Plant Science, 2022, 5(13):891783. |
| [16] |
闵学阳, 韦兴燚, 刘文献, 等. 箭筈豌豆品种间遗传差异的SSR分析及指纹图谱构建. 草业学报, 2019, 28(4):116-128.
doi: 10.11686/cyxb2018226 |
| [17] | 侯万伟, 严清彪, 张小田, 等. 西北地区主栽豌豆品种SSR指纹图谱构建. 西南农业学报, 2012, 25(1):240-242. |
| [18] | 公丹, 王素华, 程须珍, 等. 普通豇豆应用核心种质的SSR指纹图谱构建及多样性分析. 作物杂志, 2020(4):79-83. |
| [19] | 陈其福, 李艳美, 李佳荫, 等. 基于SSR标记的食荚菜豆指纹图谱构建. 北方园艺, 2019(9):1-7. |
| [20] | 张洁, 陆海峰, 李有志. 电子PCR. 分子植物育种, 2004(1):138-144. |
| [21] | 陆景标. 高丹草SSR引物电子PCR及遗传多态性分析. 合肥:安徽农业大学, 2011. |
| [22] | 管培丽, 任静, 张建昆, 等. 甘蓝全基因组锚定SSR标记分析. 分子植物育种,(2023-02-11) [2024-11-07]. https://link.cnki.net/urlid/46.1068.S.20230213.1502.003. |
| [23] |
李金璐, 王硕, 于婧, 等. 一种改良的植物DNA提取方法. 植物学报, 2013, 48(1):72-78.
doi: 10.3724/SP.J.1259.2013.00072 |
| [24] | 李彩华, 李玉环, 陈昌健, 等. 一种鉴别芦笋品种的SSR分子标记组合及其应用:CN202311698176.4. 2024-04-19. |
| [25] | 常媛飞, 刘博文, 刘万良, 等. 野豌豆属14个种牧草幼苗形态多样性与分类鉴定方法的研究. 中国草地学报, 2021, 43 (7):28-36,53. |
| [26] | 王志刚. 菜用豌豆种质资源形态性状遗传多样性分析. 北京: 中国农业科学院, 2009. |
| [27] | 杨会肖, 刘天颐, 罗锐, 等. 用SRAP标记构建松树良种指纹图谱方法的研究. 广东林业科技, 2012, 28(5):1-8. |
| [28] | 李群, 王栋, 张文兰, 等. 基于SSR标记的世界豌豆种质遗传多样性分析. 植物遗传资源学报, 2021, 22(3):684-691. |
| [29] |
童治军, 焦芳婵, 肖炳光. 普通烟草及其祖先种基因组SSR位点分析. 中国农业科学, 2015, 48(11):2108-2117.
doi: 10.3864/j.issn.0578-1752.2015.11.003 |
| [30] | Lawson M J, Zhang L Q. Distinct patterns of SSR distribution in the Arabidopsis thaliana and rice genomes. Genome Biology, 2006, 7(2):R14. |
| [31] |
Kantety R V, Rota M L, Matthews D E, et al. Data mining for simple sequence repeats in expressed sequence tags from barley, maize, rice, sorghum and wheat. Plant Molecular Biology, 2002, 48(5/6):501-510.
doi: 10.1023/A:1014875206165 |
| [32] |
Tang J F, Baldwin S J, Jacobs J M, et al. Large-scale identification of polymorphic microsatellites using an in silico approach. BMC Bioinformatics, 2008, 9(1):374-386.
doi: 10.1186/1471-2105-9-374 |
| [33] |
王玉龙, 黄冰艳, 王思雨, 等. 四倍体野生种花生A.monticola全基因组SSR的开发与特征分析. 中国农业科学, 2019, 52 (15):2567-2585.
doi: 10.3864/j.issn.0578-1752.2019.15.002 |
| [1] | Chen Lei, Tang Maoyan, Zhang Zhanying, Zhong Xiaoyuan, Gao Guoqing, Zhang Xiaoli, Liang Tianfeng, Pan Yinghua. Analysis and Evaluation of Grain Appearance Quality Traits in Rice Germplasm Resources under Heat Stress during Flowering Stage [J]. Crops, 2025, 41(6): 132-139. |
| [2] | Yang Shuo, Ma Xiaojuan, Zheng Xuan, Hou Yi, Ye Lin, Cheng Guoxin. Genetic Diversity Analysis and Disease Resistance Evaluation of 100 Pepper Germplasm Resources [J]. Crops, 2025, 41(6): 73-82. |
| [3] | Zhao Caixia, Baima Yangzhen, Yang Guanghuan, Tang Lin. Identification and Cluster Analysis of Agronomic Traits in Different Types of Rapeseed Resources [J]. Crops, 2025, 41(5): 120-127. |
| [4] | Jin Lulu, Xu Min, Wang Zisheng, Wu Xiaodong. Comparative Study on Canopy Temperature, Light and Water Characteristics and Dry Matter Production Characteristics of Organs under Cotton/Peanut Intercropping [J]. Crops, 2025, 41(5): 177-183. |
| [5] | Wang Liang, Wang Rui, Zhu Jincheng, Sang Yuwei, Shi Biao, Guo Jiashuai, Jiao Huimin, He Zongling, Shui Yong. Analysis of Salt Tolerance in 50 Peanut Varieties (Lines) at Seedling Stage [J]. Crops, 2025, 41(5): 35-41. |
| [6] | Yin Junhua, Deng Li, Guo Minjie, Miao Jianli, Hu Junping, Li Shaowei, Ren Li. Comprehensive Evaluation of Small-Seeded Peanut Varieties Based on BLUP Values and GGE Biplot [J]. Crops, 2025, 41(4): 118-125. |
| [7] | Wang Dan, Zhang Zhicheng, Han Haixia, Han Na, Wang Chunyong, Wang Zheng, Pan Mengyang, Li Qiang, Wang Yiqian, Liu Yufei, Zhang Dan. Genetic Diversity Analysis of Potato Germplasm Resources Based on SSR Technology [J]. Crops, 2025, 41(4): 29-40. |
| [8] | Gao Shuguang, Xu Dongyang, Hu Minjie, Wang Ruixia, Zhang Chunhua, Xu Bohan, Li Weifeng, Zhang Liuping. Preliminary Identification and Evaluation of Quality Traits of Sesame Germplasm Resources [J]. Crops, 2025, 41(4): 58-64. |
| [9] | Lü Rongzhen, Maihemuti∙Rouzi , Zhang Yong, Maihemuti∙Remutula , Yaermaimaiti∙Alimu , Zhang Jiancheng, Yu Tianyi. Effects of Exogenous Hormones and Inhibitors on Hormone Content, Growth and Development of Peanut in Acidified Soil [J]. Crops, 2025, 41(3): 218-224. |
| [10] | Zhao Yajie, Wen Rui, Jia Yiming, Jin Xiaolei, Zhang Yonghu, Zhang Lijun, Zhang Biao, Zhang Hui, Yu Lixia. Analysis of Genetic Diversity of Phenotypic Traits of Foxtail Millet Germplasm Resources [J]. Crops, 2025, 41(3): 61-69. |
| [11] | Chang Hongbing, Wang Chen, He Meijing, Cao Ximin, Yu Fengfang, Cao Xiaoliang, Song Wei, Lü Aizhi. Genetic Diversity Analysis of 69 Maize Germplasm Resources Based on SSR Markers [J]. Crops, 2025, 41(2): 47-53. |
| [12] | Jiang Hui, Zhong Qiaofang, Yin Fuyou, Li Jinlu, Liu Li, Zhang Yun, Wang Bo, Jiang Cong, Cheng Zaiquan, Zhang Hui, Xiao Suqin. Research Progress on Germplasm Resources and Multi-Omics of Oryza officinalis [J]. Crops, 2025, 41(2): 1-8. |
| [13] | Lu Jing, Yu Bo, Jiang Mi, Peng Lianxin, Ren Yuanhang, Wu Qi. Assessment of Genetic Diversity in 58 Germplasm Resources of Highland Barley [J]. Crops, 2025, 41(2): 20-28. |
| [14] | Yan Qunxiang, Pang Yuhui, Hong Zhuangzhuang, Bi Junge, Wang Chunping. Genetic Diversity Analysis and Specificity Evaluation of Main Traits of 141 Wheat Germplasm Resources at Domestic and Foreign [J]. Crops, 2025, 41(1): 26-34. |
| [15] | Yao Luming, Yuan Juan, Ma Xiaohong, Wang Biao. Genetic Diversity Analysis of Lablab purpureus Germplasm Resources Based on Morphological Trait and SSR Markers [J]. Crops, 2025, 41(1): 35-45. |
|
||