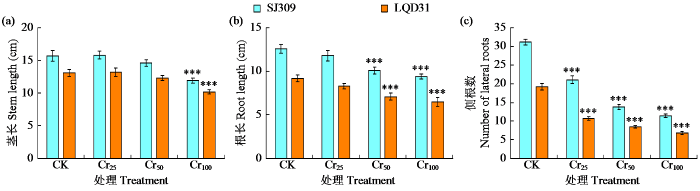重金属进入植物根表皮层主要是通过共质体与质外体途径进行的短程运输,以及木质部与韧皮部进行装卸的长程运输。植物对重金属的防御机制主要包括诱导产生金属硫蛋白、胁迫形成植物螯合肽及利用液泡对重金属进行区域化隔离[5]。植物通过细胞质膜吸收Cr6+,此过程涉及磷酸盐和硫酸盐转运体能量依赖性的活性转运[3,5]。研究[6-
叶片生理性状与形态特征变化是植物对重金属胁迫作出的信息反馈。铬的植物毒性会减少实质细胞数量、改变叶绿体超微结构、抑制电子传递过程、缩小细胞间空间,并导致叶片气孔导度异常,进而使光合速率降低[12-13]。此外,铬还会干扰细胞的矿物质吸收,破坏水分平衡[6]。气孔的保卫细胞能对包括重金属胁迫在内的非生物胁迫作出快速响应,通过调节气孔的孔径和面积来适应环境变化。在非生物胁迫条件下,保卫细胞会积累更多脱落酸(abscisic acid,ABA),促使气孔关闭。尽管ABA和水杨酸(salicylic acid,SA)可能通过改变气孔的协同作用来调节植物对非生物胁迫的耐受性[14-15],但研究[16]指出,ABA依赖基因NCED1和NCED2过表达会导致ABA积累,进而使气孔关闭、蒸腾速率下降。土壤中重金属浓度过高还会影响植物毛状体的结构完整性与分布。相关研究[17]显示,当砷浓度较高时,绿豆叶片正面和背面的毛状体密度均会降低。水稻叶片背面和正面的叶脉与表皮之间分布着腺毛状体和非腺毛状体,不过目前尚无研究报道水稻毛状体对土壤中较高浓度铬胁迫的反应情况。
1 材料与方法
1.1 试验材料
供试材料为2个水稻品种绥粳309(SJ309)和龙庆稻31(LQD31),由黑龙江省农业科学院绥化分院提供。2个水稻品种均属于常规水稻,种植普及率较高,且具有高产、优质、多抗和耐旱等特点。
1.2 试验方法
选取外观健康且大小均匀的种子,于实验室可控环境生长室中繁育发芽,将发芽种子在18 h光照/6 h黑暗、30 °C/22 °C、相对湿度50%~60%以及光合有效辐射500±25 μmol/(m2·s)的环境条件下进行生长发育。2周后,分别用含有0(CK)、25(Cr25)、50(Cr50)和100 μmol/L(Cr100)Cr6+的重铬酸钾溶液处理2种供试水稻。由于经150 μmol/L铬胁迫处理20 d后的LQD31大部分已死亡,因此,最终确定试验所需的铬胁迫浓度范围在0~100 μmol/L。对照组和各浓度处理组均进行5次重复试验。在第3周至第5周,按上述相同条件每3 d更换1次处理液,共处理21 d。
1.3 测定指标与方法
1.3.1 铬(Cr6+)含量
经铬胁迫处理3周后,收获2个水稻品种的根和叶。于60 °C下干燥24 h后制样,称取0.5 g待测样品,使用7 mL HNO3进行微波消解,用水定容至50 mL。使用iCAPTM RQ ICP-MS电感耦合等离子体质谱仪(赛默飞世尔科技公司)测定Cr6+含量,绘制标准曲线,相关系数R2≥0.999。
1.3.2 生理性状与光合气体交换参数
使用CIRAS-3便携式光合作用测定系统(PP Systems,美国)分析CK和Cr100处理下2个水稻品种叶片的气孔导度(Gs)、蒸腾速率(Tr)、水分利用率(WUE)、胞间CO2浓度(Ci)、蒸气压亏缺和叶片温度。
1.3.3 叶片组织形态与生长指标
经铬胁迫处理3周后,在真空室中,用甲醛―乙醇―乙酸(formaldehyde-alcohol-acetic acid,FAA)固定叶片30 min,在初固定的15 min内需抽放真空数次。用碘化丙啶(10 μg/mL,磷酸盐缓冲液)对洗净的叶片染色15 min,随后放入甘油中。利用TCS SP8激光扫描共聚焦显微系统(徕卡,德国)在DMI600倒置显微镜(徕卡,德国)上运行,配备63×NA1.3甘油浸没式透镜用于成像,在采集透射光图像的同时使用488 nm激发波长采集荧光图像。利用ImageJ软件测定水稻茎长、根长和侧根数。定量分析时进行50次重复试验。
1.3.4 光合色素
分别称取CK与Cr100处理的2个水稻品种叶片样品(25 mg),用1.5 mL 80%(v/v)丙酮在4 ℃避光处理48 h。随后,将样品在13 000 g下离心10 min,将上清液稀释10倍后在663、645和480 nm波长下测定吸光度[22]。计算叶绿素和类胡萝卜素含量:叶绿素含量=(7.93×A663+ 19.53×A645)×f×V/FW,类胡萝卜素含量=(A480+ 0.114×A663-0.638×A645)×f×V/FW× 112.5,式中,f表示稀释因子,V表示样品溶液体积,FW表示收获待测样品的鲜重(mg)。
1.3.5 抗氧化酶活性与丙二醛(MDA)含量
采用硫代巴比妥酸(thiobarbituric acid,TBA)法测定MDA含量。称取0.2 g鲜叶样品,用5 mL含10%三氯乙酸的0.25% TBA溶液进行均质。在95 ℃下煮沸30 min,然后在10 000 g下离心10 min,于532 nm波长处测定吸光度,然后减去在600 nm波长处测得的吸光度(非特异性)。
参照Larkindale等[23]的方法,从100 mg叶片样品中提取抗氧化酶。用2 mL含有1%聚乙烯吡咯烷酮(PVP)和1 mmol/L乙二胺四乙酸(EDTA)的100 mmol/L磷酸盐缓冲液(pH=7.0)冷冻并研磨叶片样品。在4 ℃、13 000 g下离心20 min,将上清液分离并备用。
采用紫外分光光度法测定过氧化氢酶(CAT)活性,将0.1 mL酶提取物与1 mL 50 mmol/L磷酸钠缓冲液(pH=7.0)混匀,加入0.1 mL 100 mmol/L H2O2引发反应,每隔30s记录240 nm处的吸光度变化,持续2 min。采用愈创木酚法[24]测定过氧化物酶(POD)活性,将100 μL酶提取物与0.9 mL含有1 mmol/L EDTA的磷酸二氢钾缓冲液(pH=7.0)和10 mmol/L愈创木酚混合,加入0.1 mL 100 mmol/L H2O2引发反应,室温孵育5 min,于470 nm处记录吸光度变化。采用氮蓝四唑(NBT)光还原法测定超氧化物歧化酶(SOD)活性,将0.1 mL酶提取物加入189 μL 1 mmol/L硝基蓝四氮唑和39 μL 1 mmol/L甲硫氨酸中,再用50 mmol/L磷酸盐缓冲液(pH=7.8)定容,加入3.9 μL 1 mmol/L核黄素后,将反应混合物在15 W荧光灯下孵育10 min,于560 nm处测量吸光度。CAT、POD和SOD活性单位为U/(mg prot∙min)。
1.3.6 实时荧光定量PCR(qRT-PCR)
将CK和Cr100处理的2个水稻品种叶片用液氮速冻,并在-80°C保存备用。使用RNeasy Plant Mini Kit试剂盒(Qiagen,德国)提取总RNA。基于1 mg总RNA采用Reverse Transcription Supermix for RT-qPCR逆转录预混液(Bio-Rad,美国)获得cDNA。对于相对转录水平,使用CFX384 Touch™实时检测系统(Bio-Rad,美国),按照说明和iTaq Universal SYBR Green超混合液(Bio-Rad,美国),进行qRT-PCR的定量分析[25]。使用Primer Quest软件设计qRT- PCR中使用的基因特异性引物(表1),所有反应均在384孔PCR板中进行。使用肌动蛋白(内参基因)将转录水平归一化。每个qRT-PCR试验均进行3个重复。
表1 qRT-PCR引物信息
Table 1
| 编号Code | 基因Gene | 正向引物Forward primer (5′→3′) | 反向引物Reverse primer (3′→5′) |
|---|---|---|---|
| 1 | NCED1 | GCTCGGTCACTCACTCACTC | GCGTTCTTCTTCCTGCCATAG |
| 2 | NCED2 | CATGCTCCACTCCCTTCTCA | GAAGCCAGCGAAGAAGTTTGG |
1.3.7 脱落酸(ABA)和水杨酸(SA)含量
参考Vadassery等[26]的方法,采用超高效液相色谱法对防御性植物激素(ABA和SA)进行测定与分析。将CK和Cr100处理的2个水稻品种叶片用液氮速冻,并在-80 ℃保存备用。叶片部分使用Tissuelyser-II组织研磨仪(上海净信实业发展有限公司)进行均质,于冻干机中保存过夜。取20 mg冻干样品用1 mL甲醇提取,甲醇中用40 ng/mL D6-ABA和40 ng/mL D4-SA作内标。将均质样品在振动器中混合30 min,然后在4 ℃下以13 000 g离心20 min。收集上清液,再次用500 μL甲醇提取匀浆并离心。合并提取物进行氮吹,并重新悬浮于500 μL甲醇中上机检测。
1.4 数据处理
采用方差分析(ANOVA)进行统计检验,使用Tukey检验进行事后多重比较,使用Origin 2021软件进行统计分析,显著性水平P<0.05,图表中的数据用平均值±标准差表示。
2 结果与分析
2.1 铬胁迫下水稻根和叶中的铬含量
水稻植株根和叶片中铬(Cr6+)含量的测定结果如表2所示,2个水稻品种根部铬积累量是叶的11.1~26.6倍,且随铬胁迫浓度升高,根和叶的铬积累量呈递增趋势。比较不同水稻品种根和叶中铬的富集量发现,LQD31的富集能力强于SJ309,在Cr100处理下差值最大。然而,CK处理下SJ309和LQD31的根和叶中均未检出铬,说明铬不易在水稻植株体内向茎部和叶片转移,且SJ309比LQD31能更有效地抑制植株对铬的吸收与转移。
表2 不同铬胁迫水平对水稻品种根和叶中Cr6+含量的影响
Table 2
| 品种Variety | 处理Treatment | 根Root | 叶Leaf |
|---|---|---|---|
| SJ309 | CK | 0.000±0.000e | 0.000±0.000e |
| Cr25 | 0.238±0.003d | 0.012±0.001d | |
| Cr50 | 0.337±0.006c | 0.013±0.004d | |
| Cr100 | 0.452±0.009b | 0.017±0.003c | |
| LQD31 | CK | 0.000±0.000e | 0.000±0.000e |
| Cr25 | 0.255±0.004d | 0.023±0.002b | |
| Cr50 | 0.346±0.005c | 0.025±0.003b | |
| Cr100 | 0.550±0.011a | 0.034±0.005a |
不同小写字母表示具有显著性差异(P < 0.05)。
Different lowercase letters indicate significant differences (P < 0.05).
2.2 铬胁迫下水稻幼苗的生长指标
图1
图1
不同铬胁迫水平对水稻品种幼苗生长指标的影响
“***”表示处理间差异达极显著水平(P < 0.001),下同。
Fig.1
Effects of different chromium stress levels on seedling growth index of rice varieties
“***”indicates extremely significant difference among treatments at P < 0.001 level, the same below.
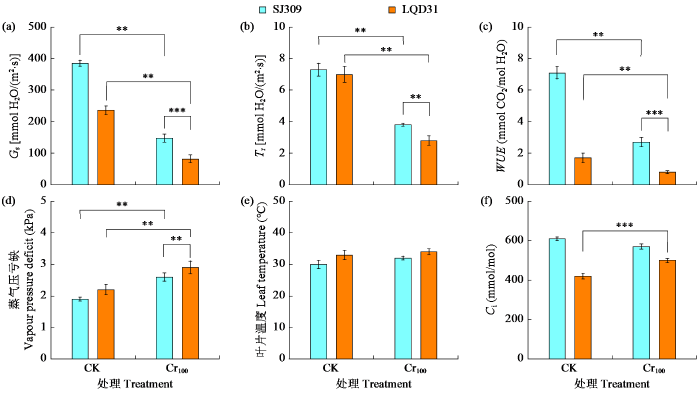
2.3 铬胁迫下水稻叶片的光合气体交换参数和生理性状
图2
图2
铬胁迫对不同水稻品种光合气体交换参数和生理性状的影响
“**”表示处理间差异达极显著水平(P < 0.01),下同。
Fig.2
Effects of chromium stress on photosynthetic gas exchange parameters and physiological traits of different rice varieties
“**”indicates extremely significant difference among treatments (P < 0.01), the same below.
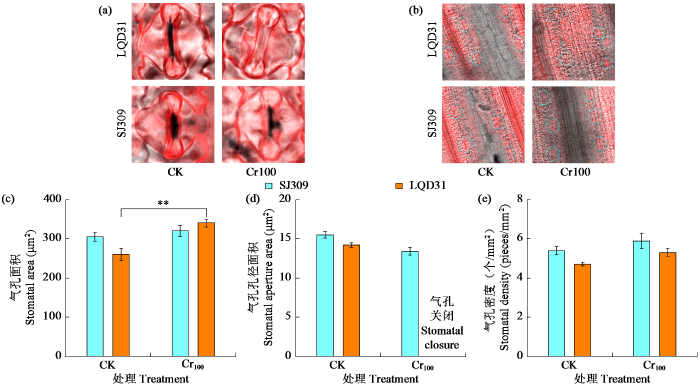
2.4 铬胁迫下水稻叶片的组织形态
图3
图3
铬胁迫对不同水稻品种叶片气孔形态的影响
Fig.3
Effects of chromium stress on leaf stomatal morphology of different rice varieties
图4
图4
铬胁迫对不同水稻品种叶片毛状体形态的影响
Fig.4
Effects of chromium stress on leaf trichome morphology of different rice varieties
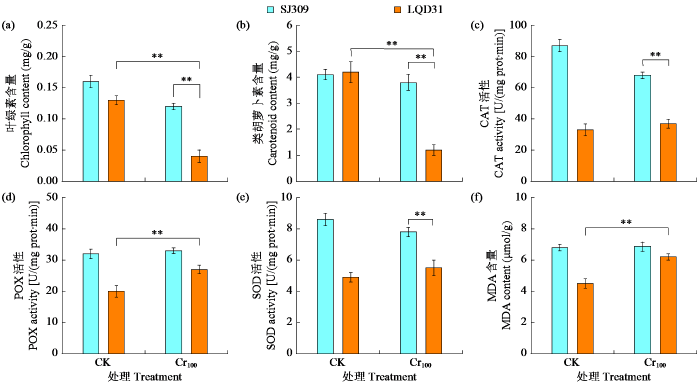
2.5 铬胁迫下水稻的光合色素与抗氧化酶活性
水稻的光合色素含量受铬胁迫的影响(图5a~b),Cr100处理下SJ309和LQD31叶片中叶绿素含量分别减少了22.0%和65.8%,类胡萝卜素含量分别减少了8.1%和65.2%,表明LQD31叶片受铬胁迫的影响更为显著。Cr100处理下,SJ309的CAT活性降低了21.0%,LQD31则提高了16.3%(图5c);SJ309和LQD31的POD活性分别提高了3.0%和34.9%(图5d);SJ309的SOD活性降低了9.1%,LQD31则提高了7.0%(图5e);SJ309和LQD31的MDA含量分别提高了1.0%和34.2%,LQD31材料Cr100与CK处理相比有显著差异(图5f)。
图5
图5
铬胁迫对不同水稻品种光合色素与抗氧化酶活性的影响
Fig.5
Effects of chromium stress on photosynthetic pigments and antioxidant enzyme activities in different rice varieties
2.6 铬胁迫下水稻植物激素的调控与表达
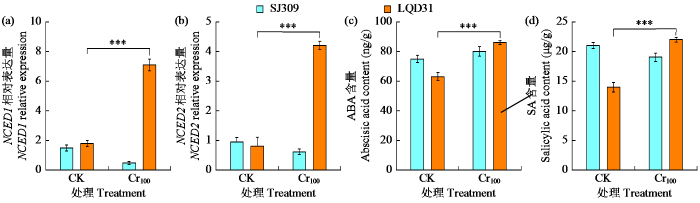
基于CK与Cr100处理植物激素调控与表达的差异分析(图6),探究不同水稻品种气孔保卫细胞孔径的分子调控作用。Cr100处理下,SJ309和LQD31叶片中NCED1基因的表达水平分别下调了72.3%和上调了315.1%,NCED2基因的表达水平分别下调了46.2%和上调了280.8%。SJ309和LQD31叶片中ABA含量分别增加了7.2%和30.7%,SA含量则分别为减少8.1%和增加43.0%。综上,铬胁迫下LQD31中ABA依赖的NCED1和NCED2基因水平上调,同时ABA和SA的协同作用增强,共同参与调控水稻叶片气孔孔径的关闭过程。
图6
图6
铬胁迫对不同水稻品种植物激素调控与表达的影响
Fig.6
Effects of chromium stress on regulation and expression of plant hormones in different rice varieties
3 讨论
由水稻LQD31根和叶中铬富集量较高以及各项生理参数的检测结果可知,LQD31因无法对铬毒性进行螯合与解离,相较于SJ309更易在根部积累Cr6+,并经茎部向叶片转移。Daud等[8]和Singh等[27]针对棉花和绿豆品种的研究均报道了类似对铬胁迫的反应。2个水稻品种SJ309和LQD31的根部均积累了较高浓度的Cr6+,这是由于Cr6+固定于根细胞的液泡中,此为植物应对毒性的自然反应。由于根系是重金属元素的首要接触部位,根部的重金属积累量高于茎和叶[28-29]。铬胁迫诱导的植物毒性效应表现为2个水稻品种生长参数下降。Jabeen等[30]研究表明,铬是毒性最强的金属,会影响农作物植株的根长和侧根数,本研究结果与其一致。根长缩短是由于根细胞壁的细胞分裂受到抑制,茎长缩短则归因于叶片细胞的超微结构变形。
较高浓度的CO2和蒸气压亏缺会促使气孔关闭,这可能与LQD31在气压不足、CO2浓度升高时气孔保卫细胞孔径关闭有关。经铬胁迫处理后,水稻叶片气孔面积增大,表明铬及其他重金属的同化与沉积会干扰气孔的功能形态,进而影响整体的光合气体交换参数,这与Chandra等[32]的结果一致。铬胁迫下LQD31的保卫细胞变形,Chen等[33]研究发现这可能是由于气孔保卫细胞细胞壁的含水量和机械性能改变,引发膨压增大所致,研究[34]中也观察到了类似的气孔保卫细胞变形现象。铬毒性导致水稻植株叶片缺水,引发SJ309和LQD31气孔密度增大。植物接触铬后,毛状体数量会发生变化,而毛状体可能是金属元素的储存区域。LQD31腺毛密度增加,表明该品种水稻是植物精油以及各类挥发性和非挥发性次级代谢产物的来源,这些代谢产物可为植物抵御食草动物提供保护[35]。重金属胁迫会影响水稻腺毛状体的形态和超微结构。此外,铬毒性还会导致SJ309非腺毛状体密度和长度增加,表明SJ309既能抵御紫外线造成的损伤、稳定温度,又能作为抵御食草动物的屏障,同时也说明SJ309比LQD31更能耐受多种环境胁迫。
经Cr6+胁迫处理后,SJ309水稻叶片中的SOD和CAT活性降低,这一结果与Tang等[36]的研究一致,证实氧化胁迫可能导致这2种抗氧化酶活性下降。然而,POD在叶片组织中的定位可能促使水稻POD活性增强[37]。MDA含量是反映ROS对植物膜造成损伤的重要指标之一。与SJ309相比,LQD31的MDA含量更高,表明其叶片细胞膜和超微结构可能因铬毒性而受损,Basit等[10]研究发现这可能是由铬毒性引发的氧化应激导致ROS活性增加所致。2个水稻品种中均发现ROS产生的氧化应激,但LQD31受影响更为显著,表明SJ309能更有效地消除ROS活性并抵御铬胁迫。Cr100处理下,2个品种的叶绿素和类胡萝卜素含量均下降,这与ROS的氧化应激有关,且LQD31的光合色素受影响程度更为显著。
本研究不仅探究了水稻应对Cr6+胁迫的生理性状与形态特征适应机制,还提出了一种依据多种生理及形态特征参数筛选抗Cr6+胁迫水稻品种的方法。基于在SJ309和LQD31中观察到的生理、形态及生化参数变化,不同水稻品种对Cr6+毒性的耐受能力存在差异。结果表明,SJ309已具备多种对Cr6+毒性的耐受机制,可被视为一种超积累水稻植株,可通过不同的基因表达将Cr6+毒性螯合并解离至液泡中[27,41]。深入探究SJ309和LQD31中参与Cr6+积累、转运和解毒机制的分子途径,将为作物育种者在铬污染土壤中选择和培育水稻栽培品种提供更优的方案。SJ309的超积累特性可用于铬污染土壤的植物修复,因此,作物育种者可采用轮作策略以提高这2个水稻品种在铬污染土壤中的产量。
4 结论
铬胁迫下,2个水稻品种植株的茎长、根长和侧根数均极显著减少,铬主要富集于根部,且SJ309比LQD31更有效减少铬的吸收与转移。二者气孔面积和密度均增加,LQD31变化更明显,其气孔保卫细胞变形、孔径闭合;铬胁迫对SJ309毛状体形态影响较大。高浓度铬胁迫(Cr100)下,2个品种的气孔导度、蒸腾速率、水分利用率、光合色素含量、CAT和SOD活性降低,蒸气压亏缺、胞间CO₂浓度、MDA含量和POD活性升高,LQD31变化幅度更大,对铬胁迫更敏感。ABA和SA含量增加促使NCED1和NCED2基因过度表达,导致LQD31叶片气孔导度降低、孔径关闭。水稻SJ309对铬胁迫耐受性更好,其超积累特性可用于铬污染土壤的植物修复。
参考文献
Chromium induced cytotoxicity in blackgram (Vigna mungo L.)
Cyto-histological and morpho-physiological responses of common duckweed (Lemna minor L.) to chromium
Chromium toxicity in plants: signaling, mitigation, and future perspectives
DOI:10.3390/plants12071502
URL
[本文引用: 2]

Plants are very often confronted by different heavy metal (HM) stressors that adversely impair their growth and productivity. Among HMs, chromium (Cr) is one of the most prevalent toxic trace metals found in agricultural soils because of anthropogenic activities, lack of efficient treatment, and unregulated disposal. It has a huge detrimental impact on the physiological, biochemical, and molecular traits of crops, in addition to being carcinogenic to humans. In soil, Cr exists in different forms, including Cr (III) “trivalent” and Cr (VI) “hexavalent”, but the most pervasive and severely hazardous form to the biota is Cr (VI). Despite extensive research on the effects of Cr stress, the exact molecular mechanisms of Cr sensing, uptake, translocation, phytotoxicity, transcript processing, translation, post-translational protein modifications, as well as plant defensive responses are still largely unknown. Even though plants lack a Cr transporter system, it is efficiently accumulated and transported by other essential ion transporters, hence posing a serious challenge to the development of Cr-tolerant cultivars. In this review, we discuss Cr toxicity in plants, signaling perception, and transduction. Further, we highlight various mitigation processes for Cr toxicity in plants, such as microbial, chemical, and nano-based priming. We also discuss the biotechnological advancements in mitigating Cr toxicity in plants using plant and microbiome engineering approaches. Additionally, we also highlight the role of molecular breeding in mitigating Cr toxicity in sustainable agriculture. Finally, some conclusions are drawn along with potential directions for future research in order to better comprehend Cr signaling pathways and its mitigation in sustainable agriculture.
Effect of chromium (VI) toxicity on morpho-physiological characteristics, yield, and yield components of two chickpea (Cicer arietinum L.)
Chromium- induced ultrastructural changes and oxidative stress in roots of Arabidopsis thaliana
DOI:10.3390/ijms160715852
URL
[本文引用: 2]

Chromium (Cr) is an abundant heavy metal in nature, toxic to living organisms. As it is widely used in industry and leather tanning, it may accumulate locally at high concentrations, raising concerns for human health hazards. Though Cr effects have extensively been investigated in animals and mammals, in plants they are poorly understood. The present study was then undertaken to determine the ultrastructural malformations induced by hexavalent chromium [Cr(VI)], the most toxic form provided as 100 μM potassium dichromate (K2Cr2O7), in the root tip cells of the model plant Arabidopsis thaliana. A concentration-dependent decrease of root growth and a time-dependent increase of dead cells, callose deposition, hydrogen peroxide (H2O2) production and peroxidase activity were found in Cr(VI)-treated seedlings, mostly at the transition root zone. In the same zone, nuclei remained ultrastructurally unaffected, but in the meristematic zone some nuclei displayed bulbous outgrowths or contained tubular structures. Endoplasmic reticulum (ER) was less affected under Cr(VI) stress, but Golgi bodies appeared severely disintegrated. Moreover, mitochondria and plastids became spherical and displayed translucent stroma with diminished internal membranes, but noteworthy is that their double-membrane envelopes remained structurally intact. Starch grains and electron dense deposits occurred in the plastids. Amorphous material was also deposited in the cell walls, the middle lamella and the vacuoles. Some vacuoles were collapsed, but the tonoplast appeared integral. The plasma membrane was structurally unaffected and the cytoplasm contained opaque lipid droplets and dense electron deposits. All electron dense deposits presumably consisted of Cr that is sequestered from sensitive sites, thus contributing to metal tolerance. It is concluded that the ultrastructural changes are reactive oxygen species (ROS)-correlated and the malformations observed are organelle specific.
Physiological and molecular analyses of black and yellow seeded Brassica napus regulated by 5-aminolivulinic acid under chromium stress
Physiological, ultrastructural, biochemical, and molecular responses of glandless cotton to hexavalent chromium (Cr6+
Chromium (VI)-induced leaf-based differential physiological, metabolic and microstructural changes in two transgenic cotton cultivars (J208,Z905) and their hybrid line (ZD14)
Photosynthesis performance, antioxidant enzymes, and ultrastructural analyses of rice seedlings under chromium stress
Seed priming with brassinosteroids alleviates chromium stress in rice cultivars via improving ROS metabolism and antioxidant defense response at biochemical and molecular levels
DOI:10.3390/antiox10071089
URL
[本文引用: 2]

This research was performed to explore the vital role of seed priming with a 0.01 µM concentration of brassinosteroids (EBL) to alleviate the adverse effects of Cr (100 µM) in two different rice cultivars. Seed priming with EBL significantly enhanced the germination attributes (germination percentage, germination energy, germination index, and vigor index, etc.), photosynthetic rate as well as plant growth (shoot and root length including the fresh and dry weight) under Cr toxicity as compared to the plants primed with water. Cr toxicity induced antioxidant enzyme activities (SOD, POD, CAT, and APX) and ROS level (MDA and H2O2 contents) in both rice cultivars; however, a larger increment was observed in YLY-689 (tolerant) than CY-927 (sensitive) cultivar. EBL application stimulatingly increased antioxidant enzyme activities to scavenge ROS production under Cr stress. The gene expression of SOD and POD in EBL-primed rice plants followed a similar increasing trend as observed in the case of enzymatic activities of SOD and POD compared to water-primed rice plants. Simultaneously, Cr uptake was observed to be significantly higher in the water-primed control compared to plants primed with EBL. Moreover, Cr uptake was significant in YLY-689 compared to CY-927. In ultra-structure studies, it was observed that EBL priming relieved the rice plants from sub-cellular damage. Conclusively, our research indicated that seed priming with EBL could be adopted as a promising strategy to enhance rice growth by copping the venomous effect of Cr.
Chromium stress in plants
DOI:10.1590/S1677-04202005000100008
URL
[本文引用: 1]

The article presents an overview of the mechanism of chromium stress in plants. Chromium is known to be a toxic metal that can cause severe damage to plants and animals. Chromium-induced oxidative stress involves induction of lipid peroxidation in plants that causes severe damage to cell membranes. Oxidative stress induced by chromium initiates the degradation of photosynthetic pigments causing decline in growth. High chromium concentration can disturb the chloroplast ultrastructure thereby disturbing the photosynthetic process. Like copper and iron, chromium is also a redox metal and its redox behaviour exceeds that of other metals like Co, Fe, Zn, Ni, etc. The redox behaviour can thus be attributed to the direct involvement of chromium in inducing oxidative stress in plants. Chromium can affect antioxidant metabolism in plants. Antioxidant enzymes like SOD, CAT, POX and GR are found to be susceptible to chromium resulting in a decline in their catalytic activities. This decline in antioxidant efficiency is an important factor in generating oxidative stress in plants under chromium stress. However, both metallothioneins and organic acids are important in plants as components of tolerance mechanisms and are also involved in detoxification of this toxic metal.
Water relations in plants subjected to heavy metal stresses
Interaction of bioaccumulation of heavy metal chromium with water relation, mineral nutrition and photosynthesis in developed leaves of Lolium perenne L.
DOI:10.1016/j.chemosphere.2007.02.052 URL [本文引用: 2]
Open or close the gate-stomata action under the control of phytohormones in drought stress conditions
Guard cell salicylic acid signaling is integrated into abscisic acid signaling via the Ca2+/CPK-dependent pathway
DOI:10.1104/pp.18.00321
PMID:30037808
[本文引用: 2]

The phenolic hormone salicylic acid (SA) induces stomatal closure. It has been suggested that SA signaling is integrated with abscisic acid (ABA) signaling in guard cells, but the integration mechanism remains unclear. The Ca-independent protein kinase Open Stomata1 (OST1) and Ca-dependent protein kinases (CPKs) are key for ABA-induced activation of the slow-type anion channel SLAC1 and stomatal closure. Here, we show that SA-induced stomatal closure and SA activation of slow-type anion channel are impaired in the CPK disruption mutant but not in the OST1 disruption mutant We also found that the key phosphorylation sites of SLAC1 in ABA signaling, serine-59 and serine-120, also are important for SA signaling. Chemiluminescence-based detection of superoxide anion revealed that SA did not require CPK3 and CPK6 for the induction of reactive oxygen species production. Taken together, our results suggest that SA activates peroxidase-mediated reactive oxygen species signal that is integrated into Ca/CPK-dependent ABA signaling branch but not the OST1-dependent signaling branch in Arabidopsis () guard cells.© 2018 American Society of Plant Biologists. All rights reserved.
ABA signal transduction at the crossroad of biotic and abiotic stress responses
DOI:10.1111/pce.2012.35.issue-1 URL [本文引用: 2]
Spatial distribution of arsenic in different leaf tissues and its effect on structure and development of stomata and trichomes in mung bean, Vigna radiata (L.) Wilczek
Assessment of cadmium and lead contamination in rice farming soils and rice (Oryza sativa L.)
Thermotolerance and antioxidant systems in Agrostis stolonifera: involvement of salicylic acid, abscisic acid, calcium, hydrogen peroxide, and ethylene
This study investigated whether pre-treating plants with specific putative signaling components and heat acclimation would induce tolerance of a cool-season grass, creeping bentgrass (Agrostis stolonifera var. palustris), to subsequent heat stress and whether thermotolerance induction of those pretreatments was associated with the regulation of antioxidant regenerating enzymes. The treatments included foliar application of salicylic acid (SA), abscisic acid (ABA), calcium chloride (CaCl2), hydrogen peroxide (H2O2), 1-aminocyclopropane-1-carboxylic acid (ACC, a precursor of ethylene prior to the exposure of plants to heat stress (35 degrees C) in a growth chamber. Physiological measurements including turf quality, leaf photosynthetic rate, and levels of oxidative damage demonstrated that all treatments increased heat tolerance. The better heat tolerance for pre-treated plants as compared to controls was related to the protection of oxidative damage under heat stress. APX activity increased over the first 2 days and 5 days of heating for ACC and CaCl2 respectively, but for only 12 h for H2O2. SA and ABA pre-treatments had no effects on APX activity earlier, but maintained APX activity at a significantly higher level than in controls after 24 h of heating. SA and ABA pre-treatments had no effects on POX activity. ACC treatment significantly increased POX activity. Pre-treatment with CaCl2, H2O2, and HA reduced POX activity, particularly during the later phase of heating. Plants treated with SA, CaCl2, H2O2 and HA had lower CAT activity than their control plants prior to heating and within 48 h of heat stress. ABA and ACC pre-treatments maintained higher CAT activity than the controls after 48 h of heating. ACC, CaCl2, or HA pre-treatments increased SOD activity only before 5 days of heat stress. SA and ABA pre-treatments had less effect on APX activity earlier under heat stress. These results suggest that specific groups of potential signaling molecules may induce tolerance of creeping bentgrass to heat stress by reducing oxidative damage.
Enhanced drought and salinity tolerance in transgenic mustard [Brassica juncea (L.) Czern & Coss.] overexpressing Arabidopsis group 4 late embryogenesis abundant gene (AtLEA4-1)
Optimization of soil parameters and cost effective way of growing Arabidopsis thaliana from an Indian perspective
CML42-mediated calcium signaling coordinates responses to Spodoptera herbivory and abiotic stresses in Arabidopsis
DOI:10.1104/pp.112.198150
URL
[本文引用: 1]

In the interaction between Arabidopsis (Arabidopsis thaliana) and the generalist herbivorous insect Spodoptera littoralis, little is known about early events in defense signaling and their link to downstream phytohormone pathways. S. littoralis oral secretions induced both Ca2+ and phytohormone elevation in Arabidopsis. Plant gene expression induced by oral secretions revealed up-regulation of a gene encoding a calmodulin-like protein, CML42. Functional analysis of cml42 plants revealed more resistance to herbivory than in the wild type, because caterpillars gain less weight on the mutant, indicating that CML42 negatively regulates plant defense; cml42 also showed increased aliphatic glucosinolate content and hyperactivated transcript accumulation of the jasmonic acid (JA)-responsive genes VSP2 and Thi2.1 upon herbivory, which might contribute to increased resistance. CML42 up-regulation is negatively regulated by the jasmonate receptor Coronatine Insensitive1 (COI1), as loss of functional COI1 resulted in prolonged CML42 activation. CML42 thus acts as a negative regulator of plant defense by decreasing COI1-mediated JA sensitivity and the expression of JA-responsive genes and is independent of herbivory-induced JA biosynthesis. JA-induced Ca2+ elevation and root growth inhibition were more sensitive in cml42, also indicating higher JA perception. Our results indicate that CML42 acts as a crucial signaling component connecting Ca2+ and JA signaling. CML42 is localized to cytosol and nucleus. CML42 is also involved in abiotic stress responses, as kaempferol glycosides were down-regulated in cml42, and impaired in ultraviolet B resistance. Under drought stress, the level of abscisic acid accumulation was higher in cml42 plants. Thus, CML42 might serve as a Ca2+ sensor having multiple functions in insect herbivory defense and abiotic stress responses.
Heavy metal tolerance in plants: role of transcriptomics, proteomics, metabolomics, and ionomics
Differential antioxidative response of ascorbate glutathione pathway enzymes and metabolites to chromium speciation stress in green gram (Vigna radiata (L.) R. Wilczek. cv CO 4) roots
DOI:10.1016/j.plantsci.2003.12.015 URL [本文引用: 1]
Hazard and effects of pollution by lead on vegetable crops
DOI:10.1007/s10806-012-9403-1 URL [本文引用: 1]
Glycinebetaine mediates chromium tolerance in mung bean through lowering of Cr uptake and improved antioxidant system
DOI:10.1080/03650340.2015.1082032 URL [本文引用: 1]
Assessing some emerging effects of hexavalent chromium on leaf physiological performance in sunflower (Helianthus annuus L.)
Mixed heavy metal stress on photosynthesis, transpiration rate, and chlorophyll content in poplar hybrids
DOI:10.1080/21580103.2015.1044024 URL [本文引用: 1]
PECTATE LYASE LIKE 12 patterns the guard cell wall to coordinate turgor pressure and wall mechanics for proper stomatal function in Arabidopsis
DOI:10.1093/plcell/koab161
URL
[本文引用: 1]

Plant cell deformations are driven by cell pressurization and mechanical constraints imposed by the nanoscale architecture of the cell wall, but how these factors are controlled at the genetic and molecular levels to achieve different types of cell deformation is unclear. Here, we used stomatal guard cells to investigate the influences of wall mechanics and turgor pressure on cell deformation and demonstrate that the expression of the pectin-modifying gene PECTATE LYASE LIKE12 (PLL12) is required for normal stomatal dynamics in Arabidopsis thaliana. Using nanoindentation and finite element modeling to simultaneously measure wall modulus and turgor pressure, we found that both values undergo dynamic changes during induced stomatal opening and closure. PLL12 is required for guard cells to maintain normal wall modulus and turgor pressure during stomatal responses to light and to tune the levels of calcium crosslinked pectin in guard cell walls. Guard cell-specific knockdown of PLL12 caused defects in stomatal responses and reduced leaf growth, which were associated with lower cell proliferation but normal cell expansion. Together, these results force us to revise our view of how wall-modifying genes modulate wall mechanics and cell pressurization to accomplish the dynamic cellular deformations that underlie stomatal function and tissue growth in plants.
Stress protective effect of Rhododendron arboreum leaves (MEL) on chromium-treated Vigna radiata plants
DOI:10.1007/s00344-020-10111-6 [本文引用: 1]
Mechanisms of trichomes and terpene compounds in indigenous and commercial Thai rice varieties against brown planthopper
DOI:10.3390/insects13050427
URL
[本文引用: 1]

Plant trichomes generally act as a physical defense against herbivore attacks and are present in a variety of plants, including rice plants. This research examined the physical and chemical defenses of rice plants against the brown planthopper (BPH), Nilaparvata lugens (Stål) (Hemiptera: Delphacidae). A total of 10 rice varieties were used in this study. An electron microscope was used to observe trichomes. Constitutive and induced volatile compound profiles were assessed using GC-MS analyses. The preference of BPH for volatiles from the 10 rice plants was tested using a two-choice arena olfactometer system. The density of prickle trichomes had a negative relationship with the BPH injury level. Without BPH infestation, the volatile of the most resistant rice variety (Rathu Heenati (RH)) was preferred by BPH than those of the other varieties, with the exception of Gled Plah Chawn. However, the relative BPH preference for volatiles from the RH variety decreased during BPH infestation. When rice plants were infested by BPH, the numbers of VOCs and these quantities decreased. In the RH variety, the emission of essentities found without BPH infestation ceased during infestation by BPH. During the BPH infestation, rice plants started to emit new VOCs that were not detected before the BPH infestation started. In conclusion, we discovered that rice plants defended against BPH by changing VOC components during BPH infestation and β-Sesquiphellandrene was likely the most effective component.
Temporal variation in nutrient requirements of tea (Camellia sinensis) in China based on QUEFTS analysis
DOI:10.1038/s41598-020-57809-x
PMID:32019970
[本文引用: 1]

Fertilisation datasets collected from field experiments (n = 21) in tea-producing areas from 2016 to 2018 were used to build a quantitative evaluation of the fertility of tropical soils (QUEFTS) model to estimate nutrient uptake of tea plants, and to investigate relationships between tea yield and nutrient accumulation. The production of 1000 kg spring tea (based on one bud with two young expanding leaves) required 12.2 kg nitrogen (N), 1.2 kg phosphorus (P), and 3.9 kg potassium (K), and the corresponding internal efficiencies (IEs) for N, P, and K were 82.0, 833.3, and 256.4 kg kg. To produce 1000 kg summer tea, 9.1 kg N, 0.8 kg P, and 3.1 kg K were required, and the corresponding IEs for N, P, and K were 109.9, 1250.0, and 322.6 kg kg. For autumn tea, 8.8 kg N, 1.0 kg P, and 3.2 kg K were required to produce 1000 kg tea, and the corresponding IEs for N, P, and K were 113.6, 1000.0, and 312.5 kg kg. Field validation experiments performed in 2019 suggested that the QUEFTS model can appropriately estimate nutrient uptake of tea plants at a certain yield and contribute to developing a fertiliser recommendation strategy for tea production.
Role of iron-lysine on morpho-physiological traits and combating chromium toxicity in rapeseed (Brassica napus L.) plants irrigated with different levels of tannery wastewater
NCED expression is related to increased ABA biosynthesis and stomatal closure under aluminum stress
Expression of ABA synthesis and metabolism genes under different irrigation strategies and atmospheric VPDs is associated with stomatal conductance in grapevine (Vitis vinifera L. cv Cabernet Sauvignon)
DOI:10.1093/jxb/ert052
PMID:23630325
[本文引用: 1]

The influence of different levels of irrigation and of variation in atmospheric vapour pressure deficit (VPD) on the synthesis, metabolism, and transport of abscisic acid (ABA) and the effects on stomatal conductance were examined in field-grown Cabernet Sauvignon grapevines. Xylem sap, leaf tissue, and root tissue were collected at regular intervals during two seasons in conjunction with measurements of leaf water potential (Ψleaf) and stomatal conductance (gs). The different irrigation levels significantly altered the Ψleaf and gs of the vines across both seasons. ABA abundance in the xylem sap was correlated with gs. The expression of genes associated with ABA synthesis, NCED1 and NCED2, was higher in the roots than in the leaves throughout and highest in the roots in mid January, a time when soil moisture declined and VPD was at its highest. Their expression in roots was also inversely related to the levels of irrigation and correlated with ABA abundance in the roots, xylem sap, and leaves. Three genes encoding ABA 8'-hydroxylases were isolated and their identities confirmed by expression in yeast cells. The expression of one of these, Hyd1, was elevated in leaves when VPD was below 2.0-2.5 kPa and minimal at higher VPD levels. The results provide evidence that ABA plays an important role in linking stomatal response to soil moisture status and that changes in ABA catabolism at or near its site of action allows optimization of gas exchange to current environmental conditions.
Plant stomata: a checkpoint of host immunity and pathogen virulence
DOI:10.1016/j.copbio.2010.05.006
PMID:20573499
[本文引用: 1]

Stomata are microscopic pores formed by pairs of guard cells in the epidermis of terrestrial plants; they are essential for gas exchange with the environment and controlling water loss. Accordingly, plants regulate stomatal aperture in response to environmental conditions, such as relative humidity, CO(2) concentration, and light intensity. Stomatal openings are also a major route of pathogen entry into the plant and plants have evolved mechanisms to regulate stomatal aperture as an immune response against bacterial invasion. In this review, we highlight studies that begin to elucidate signaling events involved in bacterium-triggered stomatal closure and discuss how pathogens may have exploited environmental conditions or, in some cases, have evolved virulence factors to actively counter stomatal closure to facilitate invasion.Copyright © 2010 Elsevier Ltd. All rights reserved.
Agronomic, breeding, and biotechnological interventions to mitigate heavy metal toxicity problems in agriculture