作物杂志,2021, 第3期: 8–18 doi: 10.16035/j.issn.1001-7283.2021.03.002
大豆百粒重相关基因的全基因组发掘分析
宿洋1( ), 杨静2, 郭勇2(
), 杨静2, 郭勇2( ), 杜维俊1(
), 杜维俊1( ), 邱丽娟2(
), 邱丽娟2( )
)
- 1山西农业大学农学院,030801,山西晋中
2中国农业科学院作物科学研究所/农作物基因资源与遗传改良国家重大科学工程/农业农村部北京大豆生物学重点实验室,100081,北京
Whole Genome Discovery and Analysis of Genes Related to 100-Seed Weight in Soybean
Su Yang1( ), Yang Jing2, Guo Yong2(
), Yang Jing2, Guo Yong2( ), Du Weijun1(
), Du Weijun1( ), Qiu Lijuan2(
), Qiu Lijuan2( )
)
- 1College of Agriculture, Shanxi Agricultural University, Jinzhong 030801, Shanxi, China
2Institute of Crop Sciences, Chinese Academy of Agricultural Sciences/The National Key Facility for Crop Gene Resources and Genetic Improvement/MOA Key Laboratory of Soybean Biology (Beijing), Beijing 100081, China
摘要:
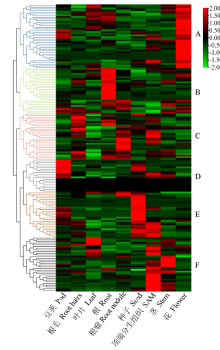
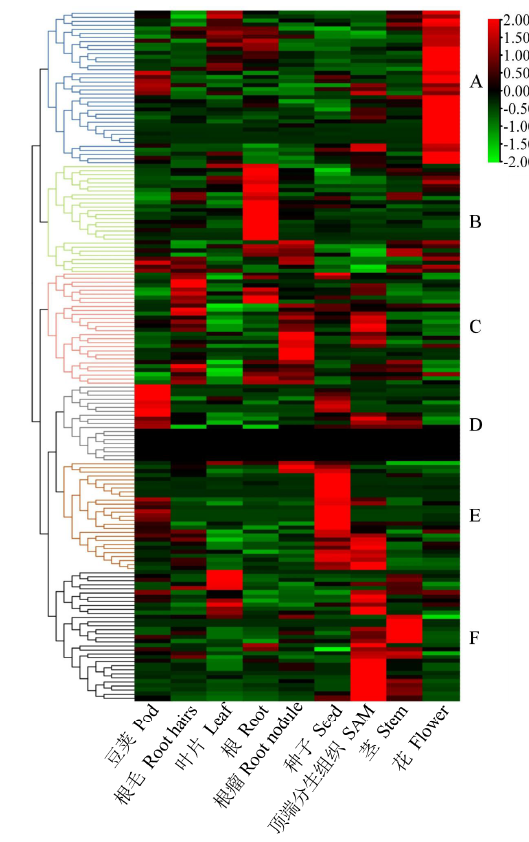
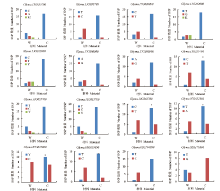
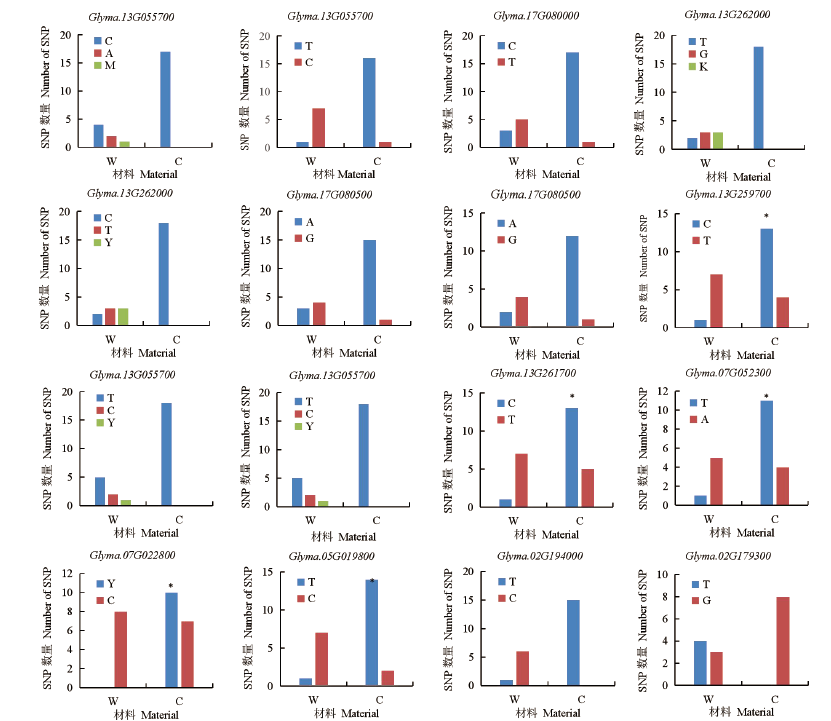
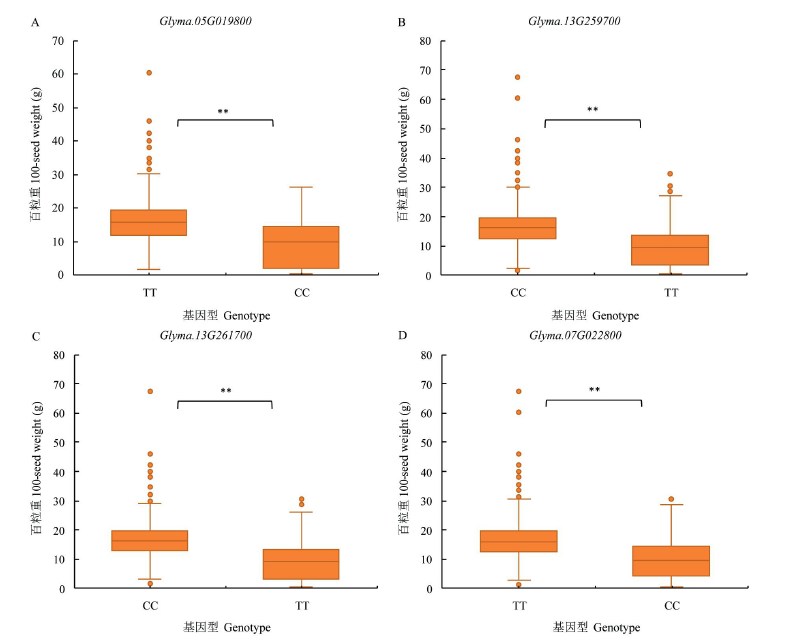
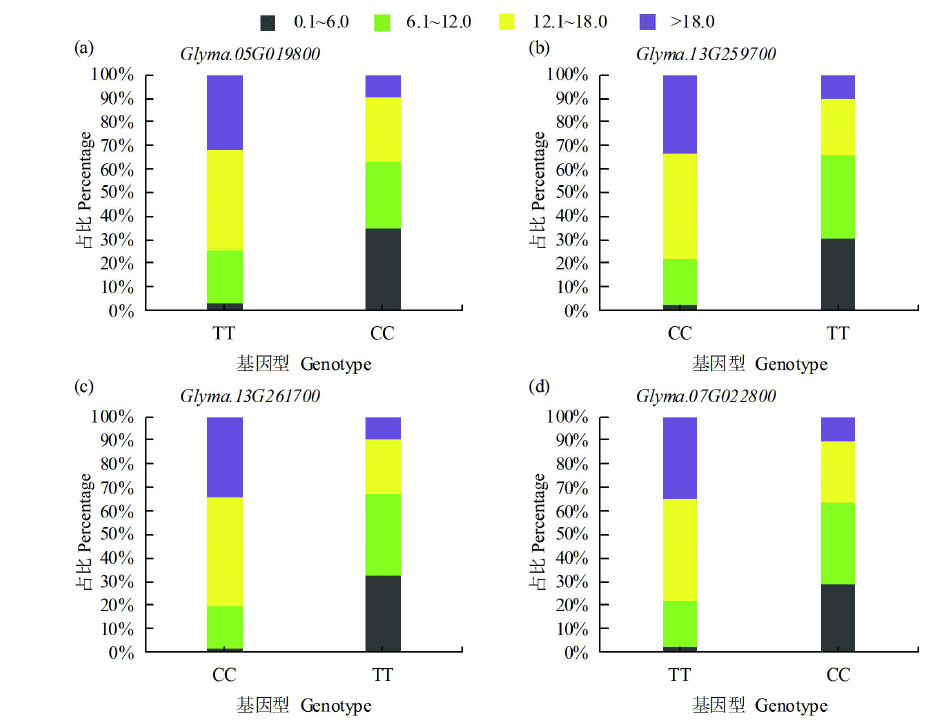
百粒重是大豆重要的产量性状之一,利用正向和反向遗传学方法鉴定与籽粒大小/粒重相关的基因具有重要的理论和实践意义。利用拟南芥和水稻等模式植物中已明确功能的调控籽粒大小/粒重的基因,基于序列相似和结构域相同的原则,在大豆全基因组内筛选到175个同源基因,通过基因表达谱分析发现有22个基因在大豆种子中特异性表达。利用56份大豆种质资源重测序数据查找这些基因内的SNP位点,共得到2769个SNP位点,从中筛选得到在野生大豆和栽培大豆中分化明显的SNP位点121个。通过扩增测序对其中的16个导致非同义变异的SNP位点进行验证,发现有5个SNP位点在野生大豆中为一种变异,而在栽培大豆中为另一种变异。利用2368份大豆种质资源的重测序数据获得了其中的4个SNP位点的变异数据,结合其中1695份材料的百粒重表型分析,发现每个SNP位点对应的野生和突变基因型材料的百粒重表型间都存在极显著差异,并且每个SNP位点中野生基因型材料的百粒重大部分≥12g,突变基因型材料的百粒重大部分<12g,因此上述4个SNP位点所在的基因(Glyma.05G019800、Glyma.07G022800、Glyma.13G259700和Glyma.13G261700)可能与大豆籽粒大小/粒重的调控有关。获得了与大豆百粒重相关的4个候选基因,为大豆百粒重QTL的精细定位和功能标记的开发以及调控大豆籽粒大小/粒重的基因的功能研究提供了参考。
| [1] |
Hymowitz T . On the domestication of the soybean. Economic Botany, 1970,24(4):408-421.
doi: 10.1007/BF02860745 |
| [2] | Meng L S, Wang Y B, Loake G J , et al. Seed embryo development is regulated via an AN3-MINI3 gene cascade. Frontiers in Plant Science, 2016,7:1645. |
| [3] |
Meng L S, Xu M K, Wan W , et al. Integration of environmental and developmental (or metabolic) control of seed mass by sugar and ethylene metabolisms in Arabidopsis. Journal of Agricultural and Food Chemistry, 2018,66(13):3477-3488.
doi: 10.1021/acs.jafc.7b05992 |
| [4] |
Pignocchi C, Minns G E, Nesi N , et al. ENDOSPERM DEFECTIVE1 is a novel microtubule-associated protein essential for seed development in Arabidopsis. The Plant Cell, 2009,21(1):90-105.
doi: 10.1105/tpc.108.061812 |
| [5] |
Jiang W B, Huang H Y, Hu Y W , et al. Brassinosteroid regulates seed size and shape in Arabidopsis. Plant Physiology, 2013,162(4):1965-1977.
doi: 10.1104/pp.113.217703 |
| [6] |
Fang W, Wang Z, Cui R , et al. Maternal control of seed size by EOD3/CYP78A6 in Arabidopsis thaliana. The Plant Journal, 2012,70(6):929-939.
doi: 10.1111/tpj.2012.70.issue-6 |
| [7] |
Ohto M, Floyd S K, Fischer R L , et al. Effects of APETALA2 on embryo,endosperm,and seed coat development determine seed size in Arabidopsis. Sexual Plant Reproduction, 2009,22(4):277-289.
doi: 10.1007/s00497-009-0116-1 |
| [8] |
Song X J, Huang W, Shi M , et al. A QTL for rice grain width and weight encodes a previously unknown RING type E3 ubiquitin ligase. Nature Genetics, 2007,39(5):623-630.
doi: 10.1038/ng2014 |
| [9] | Liu J, Chen J, Zheng X , et al. GW5 acts in the brassinosteroid signalling pathway to regulate grain width and weight in rice. Nature Plants, 2017,3(5):1-7. |
| [10] | Wu W, Liu X, Wang M , et al. A single-nucleotide polymorphism causes smaller grain size and loss of seed shattering during African rice domestication. Nature Plants, 2017,3(6):1-7. |
| [11] | 陈强, 闫龙, 冯燕 等. 大豆百粒重QTL定位及多样性评价. 中国农业科学, 2016,49(9):1646-1656. |
| [12] | 郭洁, 张继雨, 陈峰娜 等. 控制大豆油分含量和百粒重的QTL定位. 基因组学与应用生物学, 2017,36(7):2983-2988. |
| [13] |
Dhungana S K, Kulkarni K P, Park C W , et al. Mapping quantitative trait loci controlling soybean seed starch content in an interspecific cross of 'Williams 82' (Glycine max) and 'PI 366121' (Glycine soja). Plant Breeding, 2017,136(3):379-385.
doi: 10.1111/pbr.2017.136.issue-3 |
| [14] |
Liu D, Yan Y, Fujita Y , et al. Identification and validation of QTLs for 100-seed weight using chromosome segment substitution lines in soybean. Breeding Science, 2018,68(4):442-448.
doi: 10.1270/jsbbs.17127 |
| [15] |
Li J, Zhao J, Li Y , et al. Identification of a novel seed size associated locus SW9-1 in soybean. The Crop Journal, 2019,7(4):548-559.
doi: 10.1016/j.cj.2018.12.010 |
| [16] |
Wang X, Li Y, Zhang H , et al. Evolution and association analysis of GmCYP78A10 gene with seed size/weight and pod number in soybean. Molecular Biology Reports, 2015,42(2):489-496.
doi: 10.1007/s11033-014-3792-3 |
| [17] |
Lu X, Xiong Q, Cheng T , et al. A PP2C-1 allele underlying a quantitative trait locus enhances soybean 100-seed weight. Molecular Plant, 2017,10(5):670-684.
doi: 10.1016/j.molp.2017.03.006 |
| [18] |
Li Y, Zhao S, Ma J , et al. Molecular footprints of domestication and improvement in soybean revealed by whole genome re-sequencing. BMC Genomics, 2013,14(1):1-12.
doi: 10.1186/1471-2164-14-1 |
| [19] |
Li Y, Li D, Jiao Y , et al. Identification of loci controlling adaptation in Chinese soybean landraces via a combination of conventional and bioclimatic GWAS. Plant Biotechnology Journal, 2020,18(2):389-401.
doi: 10.1111/pbi.v18.2 |
| [20] |
Sun X, Shantharaj D, Kang X , et al. Transcriptional and hormonal signaling control of Arabidopsis seed development. Current Opinion in Plant Biology, 2010,13(5):611-620.
doi: 10.1016/j.pbi.2010.08.009 |
| [21] |
Savadi S . Molecular regulation of seed development and strategies for engineering seed size in crop plants. Plant Growth Regulation, 2018,84(3):401-422.
doi: 10.1007/s10725-017-0355-3 |
| [22] |
Orozco-Arroyo G, Paolo D, Ezquer I , et al. Networks controlling seed size in Arabidopsis. Plant Reproduction, 2015,28(1):17-32.
doi: 10.1007/s00497-015-0255-5 pmid: 25656951 |
| [23] |
Li N, Li Y . Signaling pathways of seed size control in plants. Current Opinion in Plant Biology, 2016,33:23-32.
doi: 10.1016/j.pbi.2016.05.008 |
| [24] |
Du L, Li N, Chen L , et al. The ubiquitin receptor DA1 regulates seed and organ size by modulating the stability of the ubiquitin-specific protease UBP15/SOD2 in Arabidopsis. The Plant Cell, 2014,26(2):665-677.
doi: 10.1105/tpc.114.122663 |
| [25] |
Li Y, Zheng L, Corke F , et al. Control of final seed and organ size by the DA1 gene family in Arabidopsis thaliana. Genes and Development, 2008,22(10):1331-1336.
doi: 10.1101/gad.463608 |
| [26] |
Xia T, Li N, Dumenil J , et al. The ubiquitin receptor DA1 interacts with the E3 ubiquitin ligase DA2 to regulate seed and organ size in Arabidopsis. The Plant Cell, 2013,25(9):3347-3359.
doi: 10.1105/tpc.113.115063 |
| [27] |
Liu S, Hua L, Dong S , et al. OsMAPK6,a mitogen-activated protein kinase,influences rice grain size and biomass production. The Plant Journal, 2015,84(4):672-681.
doi: 10.1111/tpj.2015.84.issue-4 |
| [28] |
Ishimaru K, Hirotsu N, Madoka Y , et al. Loss of function of the IAA-glucose hydrolase gene TGW6 enhances rice grain weight and increases yield. Nature Genetics, 2013,45(6):707-711.
doi: 10.1038/ng.2612 pmid: 23583977 |
| [29] |
Wang S, Li S, Liu Q , et al. The OsSPL16-GW7 regulatory module determines grain shape and simultaneously improves rice yield and grain quality. Nature Genetics, 2015,47(8):949-954.
doi: 10.1038/ng.3352 |
| [30] |
Wang S, Wu K, Yuan Q , et al. Control of grain size,shape and quality by OsSPL16 in rice. Nature Genetics, 2012,44(8):950-954.
doi: 10.1038/ng.2327 |
| [31] |
Huang X, Qian Q, Liu Z , et al. Natural variation at the DEP1 locus enhances grain yield in rice. Nature Genetics, 2009,41(4):494-497.
doi: 10.1038/ng.352 |
| [32] |
Zhong X, Dai X, Xv J , et al. Cloning and expression analysis of GmGAL1,SOC1 homolog gene in soybean. Molecular Biology Reports, 2012,39(6):6967-6974.
doi: 10.1007/s11033-012-1524-0 |
| [33] |
Deshmukh R K, Vivancos J, Guérin V , et al. Identification and functional characterization of silicon transporters in soybean using comparative genomics of major intrinsic proteins in Arabidopsis and rice. Plant Molecular Biology, 2013,83(4/5):303-315.
doi: 10.1007/s11103-013-0087-3 |
| [34] | 薛晨晨, 徐筋燕, 郭娜 等. 大豆GmGLDH基因的克隆,表达及生物信息学分析. 分子植物育种, 2019,17(6):1738-1745. |
| [35] |
Brim C A, Cockerham C C . Inheritance of quantitative characters in soybeans. Crop Science, 1961,1(3):187-190.
doi: 10.2135/cropsci1961.0011183X000100030009x |
| [36] |
Zhou Z, Jiang Y, Wang Z , et al. Resequencing 302 wild and cultivated accessions identifies genes related to domestication and improvement in soybean. Nature Biotechnology, 2015,33(4):408-414.
doi: 10.1038/nbt.3096 |
| [37] |
Kulkarni K P, Patil G, Valliyodan B , et al. Comparative genome analysis to identify SNPs associated with high oleic acid and elevated protein content in soybean. Genome, 2018,61(3):217-222.
doi: 10.1139/gen-2017-0158 |
| [1] | 李琼, 常世豪, 武婷婷, 耿臻, 杨青春, 舒文涛, 李金花, 张东辉, 张保亮. 120份大豆种质资源遗传多样性和亲缘关系分析[J]. 作物杂志, 2021, (4): 51–58 |
| [2] | 楚光红, 章建新, 王聪, 赵占营. 幼苗期不同节位摘心对高产春大豆根系生长和产量的影响[J]. 作物杂志, 2021, (3): 195–201 |
| [3] | 秦宁, 李俊茹, 李文龙, 杜汇, 李喜焕, 张彩英. 大豆籽粒生育酚及其组分含量鉴定与优异种质筛选[J]. 作物杂志, 2021, (3): 34–39 |
| [4] | 李灿东, 郭泰, 王志新, 郑伟, 赵海红, 张振宇, 徐杰飞, 郭美玲. 黑龙江省中东部地区主栽大豆品种产量指标筛选及评价[J]. 作物杂志, 2021, (2): 45–51 |
| [5] | 王财金, 弟文静, 马淑梅, 王洋. 发掘大豆资源中灰斑病1号生理小种抗性优异等位变异[J]. 作物杂志, 2020, (6): 189–196 |
| [6] | 赵宇杨, 宋健, 邱丽娟. 大豆G位点近等基因系叶片类囊体蛋白质组比较分析[J]. 作物杂志, 2020, (6): 8–16 |
| [7] | 曾艳华, 谢和霞, 江禹奉, 周锦国, 谢小东, 周海宇, 谭贤杰, 覃兰秋, 程伟东. 基于SNP标记的爆裂玉米农家品种遗传多样性[J]. 作物杂志, 2020, (5): 65–70 |
| [8] | 常世豪, 杨青春, 舒文涛, 李金花, 李琼, 张保亮, 张东辉, 耿臻. 黄淮海夏大豆品种(系)主要农艺性状的综合性分析[J]. 作物杂志, 2020, (3): 66–72 |
| [9] | 徐冉, 王彩洁, 张礼凤, 李伟, 张彦威, 林延慧, 刘薇. 应用大豆表型设计育种技术选育大豆品种齐黄34[J]. 作物杂志, 2020, (3): 73–78 |
| [10] | 黄俊霞,黄甜,饶德民,张鸣浩,孟凡钢,闫晓艳,张伟. 花后水肥一体化与化控措施对大豆产量及生理特征的影响[J]. 作物杂志, 2020, (2): 82–87 |
| [11] | 王明瑶,曹亮,于奇,邹京南,何松榆,秦彬,王孟雪,张玉先. 褪黑素浸种对盐碱胁迫下大豆种子萌发的影响[J]. 作物杂志, 2019, (6): 195–202 |
| [12] | 张永芳,钱肖娜,王润梅,史鹏清,杨荣. 不同大豆材料的抗旱性鉴定及耐旱品种筛选[J]. 作物杂志, 2019, (5): 41–45 |
| [13] | 刘念析,陈亮,厉志,刘宝泉,刘佳,衣志刚,董志敏,王曙明. 大豆抗病分子标记的研究进展[J]. 作物杂志, 2019, (4): 10–16 |
| [14] | 杨珺凯,沈阳,才晓溪,邬升杨,李建伟,孙明哲,贾博为,孙晓丽. 大豆PHD家族蛋白的全基因组鉴定及表达特征分析[J]. 作物杂志, 2019, (3): 55–65 |
| [15] | 蔡东芳,张书芬,王建平,曹金华,文雁成,张书法,何俊平,赵磊,王东国,朱家成. 利用SNP芯片解析油菜杂交种丰油10号的遗传基础[J]. 作物杂志, 2019, (3): 80–85 |
|
||