作物杂志,2026, 第2期: 154–159 doi: 10.16035/j.issn.1001-7283.2026.02.019
外源黄体酮影响小麦根系伸长生长的生理调控途径
王丹1( ), 王润2, 胡孝庆2, 于会勇1, 李江涛1, 程星1, 郭海悦1, 刘婷1, 胡珍珍1, 李华2(
), 王润2, 胡孝庆2, 于会勇1, 李江涛1, 程星1, 郭海悦1, 刘婷1, 胡珍珍1, 李华2( )
)
1濮阳市农林科学院, 457000, 河南濮阳 2河南农业大学生命科学学院, 450002, 河南郑州
Physiological Regulation Pathways of Exogenous Progesterone Affecting Wheat Root Elongation and Growth
Wang Dan1( ), Wang Run2, Hu Xiaoqing2, Yu Huiyong1, Li Jiangtao1, Cheng Xing1, Guo Haiyue1, Liu Ting1, Hu Zhenzhen1, Li Hua2(
), Wang Run2, Hu Xiaoqing2, Yu Huiyong1, Li Jiangtao1, Cheng Xing1, Guo Haiyue1, Liu Ting1, Hu Zhenzhen1, Li Hua2( )
)
1Puyang Academy of Agricultural and Forestry Sciences Puyang 457000, Henan, China 2College of Life Sciences ,Henan Agricultural University Zhengzhou 450002, Henan
摘要:
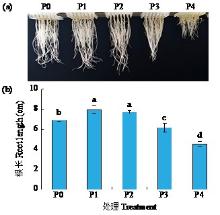
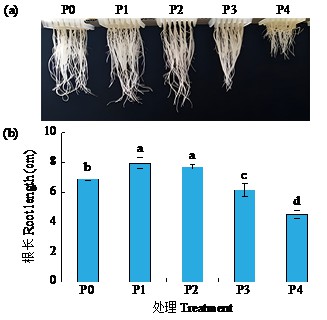
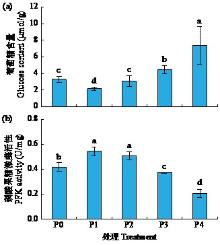
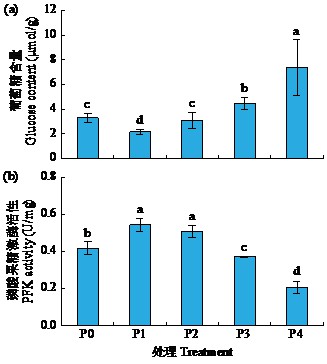
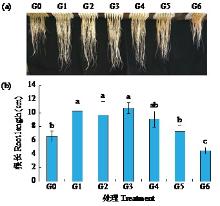
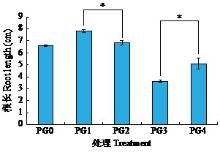
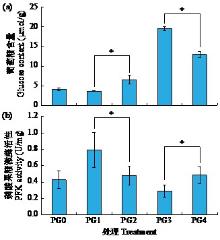
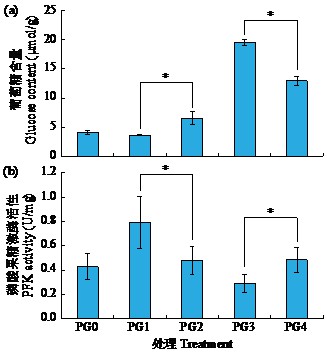
黄体酮是一种存在于植物体中的类固醇激素,与植物的生长发育紧密相关,对植物抵抗逆境胁迫也有积极作用。小麦根系生长状况直接影响产量与抗逆能力,本试验研究了外源黄体酮对小麦根系伸长生长的影响,并初步分析其调控途径。结果表明,不同浓度外源黄体酮对小麦根系生长的调控效应存在差异,低浓度(0.001和0.01 μmol/L)促进根系伸长生长,高浓度(0.1和1.0 μmol/L)抑制其生长。外源黄体酮处理显著影响小麦幼苗根系中的葡萄糖含量及磷酸果糖激酶(PFK)活性,随着黄体酮处理浓度升高,根系葡萄糖含量先降后升,而PFK活性变化趋势与之相反。进一步研究发现,10 μmol/L葡萄糖处理下幼苗根系葡萄糖含量降低,PFK活性升高;10 000 μmol/L葡萄糖处理下幼苗根系葡萄糖含量升高,PFK活性受到抑制。此外,添加0.1 μmol/L黄体酮可显著促进低浓度葡萄糖处理下根系葡萄糖的积累,并抑制葡萄糖对PFK活性的诱导作用;而添加0.001 μmol/L黄体酮能显著抑制高浓度葡萄糖处理下根系葡萄糖的累积,缓解葡萄糖对PFK活性的抑制作用。外源黄体酮可能通过作用于糖酵解途径的关键限速酶PFK影响糖酵解过程,进而调控根系中葡萄糖含量,实现对小麦根系伸长生长的调控。
| [1] |
Iino M, Nomura T, Tamaki Y, et al. Progesterone: Its occurrence in plants and involvement in plant growth. Phytochemistry, 2007, 68(12):1664-1673.
doi: 10.1016/j.phytochem.2007.04.002 pmid: 17512025 |
| [2] |
Simons R G, Grinwich D L. Immunoreactive detection of four mammalian steroids in plants. Canadian Journal of Botany, 1989, 67(2):288-296.
doi: 10.1139/b89-042 |
| [3] |
Erdal S. Alleviation of salt stress in wheat seedlings by mammalian sex hormones. Journal of the Science of Food and Agriculture, 2012, 92(7):1411-1416.
doi: 10.1002/jsfa.4716 pmid: 22102166 |
| [4] |
Filek M, Rudolphi-Skórska E, Sieprawska A, et al. Regulation of the membrane structure by brassinosteroids and progesterone in winter wheat seedlings exposed to low temperature. Steroids, 2017, 128:37-45.
doi: S0039-128X(17)30182-4 pmid: 29030144 |
| [5] |
Xue R L, Wang S Q, Xu H L, et al. Progesterone increases photochemical efficiency of photosystem II in wheat under heat stress by facilitating D1 protein phosphorylation. Photosynthetica, 2017, 55(4):664-670.
doi: 10.1007/s11099-016-0681-0 |
| [6] |
Su X Y, Wu S, Yang L, et al. Exogenous progesterone alleviates heat and high light stress-induced inactivation of photosystem II in wheat by enhancing antioxidant defense and D1 protein stability. Plant Growth Regulation, 2014, 74(3):311-318.
doi: 10.1007/s10725-014-9920-1 |
| [7] |
Janeczko A, Tobias I, Skoczowski A, et al. Progesterone moderates damage in Arabidopsis thaliana caused by infection with Pseudomonas syringae or P. fluorescens. Biologia Plantarum, 2013, 57(1):169-173.
doi: 10.1007/s10535-012-0142-y |
| [8] |
Shpakovski G V, Spivak S G, Berdichevets I N, et al. A key enzyme of animal steroidogenesis can function in plants enhancing their immunity and accelerating the processes of growth and development. BMC Plant Biology, 2017, 17(S1):189.
doi: 10.1186/s12870-017-1123-2 |
| [9] |
Hao J S, Li X, Xu G Z, et al. Exogenous progesterone treatment alleviates chilling injury in postharvest banana fruit associated with induction of alternative oxidase and antioxidant defense. Food Chemistry, 2019, 286(15):329-337.
doi: 10.1016/j.foodchem.2019.02.027 |
| [10] |
Erdal S, Genisel M. The property of progesterone to mitigate cold stress in maize is linked to a modulation of the mitochondrial respiratory pathway. Theoretical and Experimental Plant Physiology, 2016, 28(4):385-393.
doi: 10.1007/s40626-016-0076-4 |
| [11] |
Genisel M, Turk H, Erdal S. Exogenous progesterone application protects chickpea seedlings against chilling-induced oxidative stress. Acta Physiologiae Plantarum, 2013, 35(1):241-251.
doi: 10.1007/s11738-012-1070-3 |
| [12] |
Bhattacharya B, Gupta K. Steroid hormone effects on growth and apical dominance of sunflower. Phytochemistry, 1981, 20(5):989-991.
doi: 10.1016/0031-9422(81)83014-2 |
| [13] |
Ylstra B, Brinkmann A O, Heberlebors E, et al. Steroid hormones stimulate germination and tube growth of in vitro matured tobacco pollen. Plant Physiology, 1995, 107(2):639-643.
pmid: 12228388 |
| [14] |
Speranza A, Crosti P, Malerba M, et al. The environmental endocrine disruptor, bisphenol A, affects germination, elicits stress response and alters steroid hormone production in kiwifruit pollen. Plant Biology, 2011, 13(1):209-217.
doi: 10.1111/j.1438-8677.2010.00330.x pmid: 21143743 |
| [15] |
Janeczko A, Oklestkova J, Novak O, et al. Disturbances in production of progesterone and their implications in plant studies. Steroids, 2015, 96:153-163.
doi: 10.1016/j.steroids.2015.01.025 pmid: 25676788 |
| [16] |
Janeczko A, Filek W. Stimulation of generative development in partly vernalized winter wheat by animal sex hormone. Acta Physiologiae Plantarum, 2002, 24:291-295.
doi: 10.1007/s11738-002-0054-0 |
| [17] |
Janeczko A, Filek W, Biesaga-Kościelniak J, et al. The influence of animal sex hormones on the induction of flowering in Arabidopsis thaliana: comparison with the effect of 24- epibrassinolide. Plant Cell Tissue and Organ Culture, 2003, 72 (2):147-151.
doi: 10.1023/A:1022291718398 |
| [18] |
Janeczko A. The presence and activity of progesterone in the plant kingdom. Steroids, 2012, 77(3):169-173.
doi: 10.1016/j.steroids.2011.10.012 pmid: 22100776 |
| [19] | 王晨阳, 郭天财, 马冬云, 等. 小麦苗期根系生长与地上部发育的协同关系. 麦类作物学报, 2019, 39(5):628-636. |
| [20] | 张宾, 陈庆全, 李友军, 等. 小麦苗期根系形态建成及其对干旱胁迫的响应. 植物生理学报, 2022, 58(3):589-598. |
| [21] | 赵广远, 周苏玫, 王志伟. 小麦根系下扎深度与水肥利用效率的关系. 作物学报, 2020, 46(8):1121-1130. |
| [22] | 董藏运, 张向前, 张英华. 小麦苗期促根栽培对群体质量及产量的影响. 华北农学报, 2021, 36(增1):156-161. |
| [23] |
Garay-Arroyo A, De La Paz Sánchez M, García-Ponce B, et al. Hormone symphony during root growth and development. Developmental Dynamics, 2012, 241(12):1867-1885.
doi: 10.1002/dvdy.23878 pmid: 23027524 |
| [24] |
Pacifici E, Polverari L, Sabatini S. Plant hormone cross-talk: The pivot of root growth. Journal of Experimental Botany, 2015, 66 (4):1113-1121.
doi: 10.1093/jxb/eru534 pmid: 25628331 |
| [25] | 赵敏, 米青荣. 生长素类物质对根芽生长的不同影响. 邯郸农业高等专科学校学报, 1996(3):14-15. |
| [26] |
Saksena H B, Sharma M, Singh D, et al. The versatile role of glucose signalling in regulating growth, development and stress responses in plants. Journal of Plant Biochemistry and Biotechnology, 2020, 29(4):1-13.
doi: 10.1007/s13562-020-00550-3 |
| [27] | Sami F, Siddiqui H, Hayat S. Interaction of glucose and phytohormone signaling in plants. Plant Physiology and Biochemistry, 2019, 13:119-126. |
| [28] |
Mishra B S, Singh M, Aggrawal P, et al. Glucose and auxin signaling interaction in controlling Arabidopsis thaliana seedlings root growth and development. PLoS ONE, 2009, 4(2):e4502.
doi: 10.1371/journal.pone.0004502 |
| [29] |
Yuan T T, Xu H H, Zhang K X, et al. Glucose inhibits root meristem growth via ABA INSENSITIVE 5, which represses PIN1 accumulation and auxin activity in Arabidopsis. Plant Cell & Environment, 2014, 37(6):1338-1350.
doi: 10.1111/pce.2014.37.issue-6 |
| [30] |
Lang D M, Lyu D, Zhu Z T, et al. Exogenous Glucose mediates the regulation of root morphology and carbon-nitrogen metabolism by Indole-3-Acetic Acid (IAA) in Malus baccata (L.) Borkh. in soil with low organic carbon content. Journal of Plant Growth Regulation, 2019, 38(4):1598-1615.
doi: 10.1007/s00344-019-10005-2 |
| [1] | 袁博, 张晓, 方正武, 李曼, 寿路路, 汪尊杰, 江伟, 肖龙飞, 高德荣. 小麦面粉和鲜面片色泽及相关基因组成分析[J]. 作物杂志, 2026, (2): 1–11 |
| [2] | 喻奇伟, 张月华, 贺帅, 张梁, 景延秋, 马黔, 马关凯, 朱国志, 王茂贤, 王秋领. 聚天门冬氨酸与壳聚糖配施对烤烟生长及烤后烟叶品质的影响[J]. 作物杂志, 2026, (2): 109–119 |
| [3] | 王静, 王志红, 侯现军, 艾振光, 闫丽慧, 王昌亮, 张国合, 常建智. 玉米自交系气生根性状与抗倒伏性的相关性及通径分析[J]. 作物杂志, 2026, (2): 23–29 |
| [4] | 代丽婷, 车京玉, 刘宁涛, 田超, 尹雪巍, 马勇, 王志坤, 张金鹏, 张起昌. 黑龙江省小麦根腐病病原菌鉴定[J]. 作物杂志, 2026, (2): 230–237 |
| [5] | 孙娟, 王新栋, 郄彦敏, 牛雪婧, 王丽娜, 耿立格. 引发处理对低发芽力小麦种子萌发和生理特性的影响[J]. 作物杂志, 2026, (2): 259–264 |
| [6] | 袁谦, 赵永涛, 张中州, 甄士聪, 望俊森, 张锋, 陈莉, 刘迪, 周扬. 2020-2024年黄淮麦区强筋和中强筋国审小麦品种的品质及育种策略分析[J]. 作物杂志, 2026, (2): 39–50 |
| [7] | 金艳, 宋全昊, 宋佳静, 郜战宁, 马红珍, 陈亮, 朱统泉. 黄淮南片麦区新育成小麦品种(系)高分子量麦谷蛋白遗传多样性分析[J]. 作物杂志, 2026, (2): 59–67 |
| [8] | 周喜旺, 刘鸿燕, 王娜, 魏志平, 王希恩, 岳维云, 王伟, 汪石俊, 孙振宇, 张耀辉. 73份甘肃陇南冬小麦品种(系)抗条锈病基因分子检测[J]. 作物杂志, 2026, (2): 82–89 |
| [9] | 陆楚盛, 赖嘉俊, 唐偲, 梁开明, 覃元钰, 钟旭华, 潘俊峰, 刘彦卓, 胡香玉, 胡锐, 李妹娟, 王昕钰, 尹媛红, 叶群欢, 沈宏, 傅友强. 铵、硝营养条件下不同酸碱度对水稻苗期生长和养分吸收的影响[J]. 作物杂志, 2026, (2): 90–97 |
| [10] | 孙茹梦, 张男, 殷佳, 汝艳, 景文疆, 张耗. 水稻根系分泌物对干旱胁迫的响应研究进展[J]. 作物杂志, 2026, (1): 1–8 |
| [11] | 吴昊, 贺金萍, 廖朝霞, 薛承康, 吴瑶瑶, 李宗芸, 刘敬然. 块根分化建成期干旱―复水对甘薯根系活性氧代谢与内源激素的影响[J]. 作物杂志, 2026, (1): 111–117 |
| [12] | 周文丽, 郝淼艺, 张仁和. 高密度种植下氮肥对玉米根系生长及氮代谢的影响[J]. 作物杂志, 2026, (1): 125–132 |
| [13] | 贾永红, 魏海鹏, 曾潮武, 刘俊, 陈艳妮, 李建疆, 梁晓东. 基于KASP技术鉴定新疆春小麦材料抗病基因[J]. 作物杂志, 2026, (1): 15–19 |
| [14] | 叶晓娟, 刘强. 不同降水年型下春小麦产量对降水、施氮及秸秆覆盖的响应模拟[J]. 作物杂志, 2026, (1): 217–224 |
| [15] | 王云江, 王玉莹, 刘畅, 敬雪妍, 羊辰, 孙彩霞, 王春平. 施氮量对小麦籽粒主要矿质元素含量的影响和有效性分析[J]. 作物杂志, 2026, (1): 240–248 |
|
||