作物杂志,2018, 第4期: 53–61 doi: 10.16035/j.issn.1001-7283.2018.04.010
甘蔗ScHAK10基因克隆及表达分析
罗海斌1,蒋胜理1,黄诚梅1,曹辉庆1,邓智年2,吴凯朝2,徐林2,陆珍1,魏源文1
- 1 广西作物遗传改良生物技术重点开放实验室,530007,广西南宁
2 广西农业科学院甘蔗研究所/农业农村部广西甘蔗生物技术与遗传改良重点实验室/广西甘蔗遗传改良重点实验室,530007,广西南宁
Cloning and Expression of ScHAK10 Gene in Sugarcane
Luo Haibin1,Jiang Shengli1,Huang Chengmei1,Cao Huiqing1,Deng Zhinian2,Wu Kaichao2,Xu Lin2,Lu Zhen1,Wei Yuanwen1
- 1 Guangxi Crop Genetic Improvement and Biotechnology Laboratory, Nanning 530007, Guangxi, China
2 Sugarcane Research Institute, Guangxi Academy of Agricultural Sciences/Key Laboratory of Sugarcane Biotechnology and Genetic Improvement of Guangxi, Ministry of Agriculture and Rural Affairs, Nanning 530007, Guangxi, China
摘要:
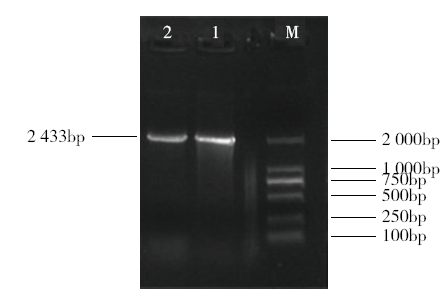
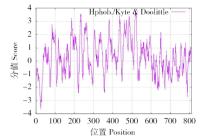
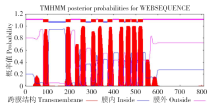
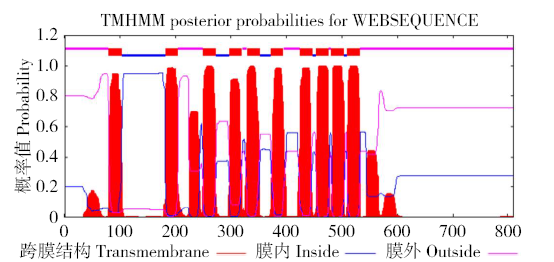
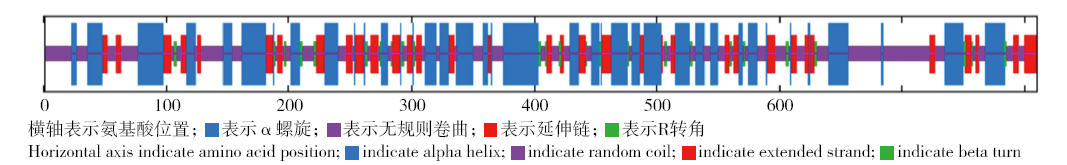
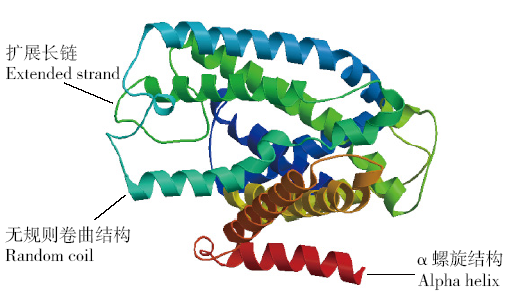
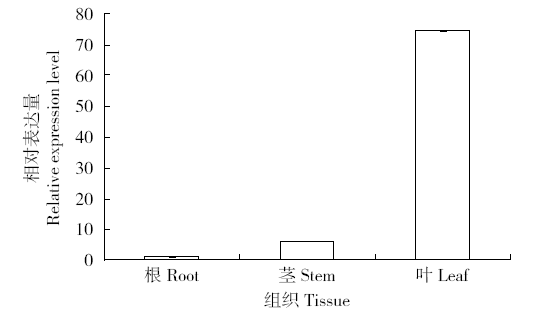
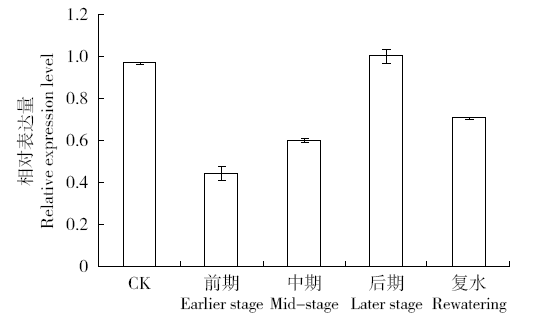
为探究甘蔗HAK基因家族中ScHAK10基因的功能及干旱环境下的响应机制,利用同源克隆技术(homologous cloning)从新台糖22号中克隆得到ScHAK10基因的完整编码序列,并对其进行生物信息学分析,同时采用q-PCR技术分析基因在不同组织以及在不同干旱阶段的表达情况。结果表明,ScHAK10基因编码区全长为2 433bp,编码810个氨基酸,相对分子量89.83kD,等电点(pI)为8.46,预测其为疏水碱性蛋白;核苷酸同源性分析表明ScHAK10基因与玉米(Zea mays L.)、高粱[Sorghum bicolor (L.) Moench]等其他作物中HAK基因具有高度的同源性;该蛋白具有跨膜结构域,定位在细胞质膜上。蛋白质二级结构分析发现其主要由α螺旋(38.15%)、扩展长链(19.26%)和无规则卷曲(37.16%)组成;蛋白功能预测显示其与阳离子运输通道相关,有较大的概率显示其介入转录、信号传导、免疫应答等生物活动。qPCR表达分析显示,ScHAK10基因在不同部位都有表达,叶片中的表达量最高,其次为茎,根系中的表达量最低。干旱胁迫下ScHAK10基因受到诱导表达,表达水平根据胁迫程度的加深而呈现出先上调后下降的表达趋势,复水后,基因表达量降低。本研究为进一步阐明甘蔗ScHAK10基因的功能及甘蔗耐旱调控相关分子机制奠定了基础。
| [1] | 姚瑞亮, 李杨瑞, 陈如凯 . 广西旱坡地甘蔗高产栽培措施的探讨. 广西农业生物科学, 1999,18(S1):29-31. |
| [2] | 陈如凯, 张木清, 陆裔波 . 甘蔗品种的御旱性差异研究. 甘蔗, 1996,3(1):1-4. |
| [3] | Malik K B . Some anatomical characteristics of sugarcane varieties in relation to drought resistance. Agriculture Research, 1986,11(1):43-49. |
| [4] | 罗海斌, 曹辉庆, 魏源文 , 等. 干旱胁迫下甘蔗苗期根系全长cDNA文库构建及EST序列分析. 南方农业学报, 2012,43(6):733-737. |
| [5] |
Maser P, Thomine S, Schroeder J I , et al. Phylogenetic relationships within cation transporter families of Arabidopsis. Plant Physiology, 2001,126(4):1646-1667.
doi: 10.1104/pp.126.4.1646 pmid: 11500563 |
| [6] |
Yang Z F, Gao Q S, Sun Ch S , et al. Molecular evolution and functional divergence of HAK potassium transporter gene family in rice (Oryza sativa L.). Journal of Genetics and Genomics, 2009,36(3):161-172.
doi: 10.1016/S1673-8527(08)60103-4 |
| [7] |
Ahn S J, Shin R, Schachtman D P . Expression of KT/KUP genes in Arabidopsis and the role of root hairs in K+ uptake . Plant Physiology, 2004,134(3):1135-1145.
doi: 10.1104/pp.103.034660 pmid: 14988478 |
| [8] |
Gierth M, Mäser P, Schroeder J I . The potassium transporter AtHAK5 functions in K+ deprivation-induced high-affinity K+ uptake and AKT1 K+ channel contribution to K+ uptake kinetics in Arabidopsis roots . Plant Physiology, 2005,137(3):1105-1114.
doi: 10.1104/pp.104.057216 |
| [9] | Fu H H, Luan S . AtHUPl:a dual-affinity K+ transporter from Arabidopsis . Plant Cell, 1998,10(1):63-73. |
| [10] |
Santa-Maria G E, Rubio F, Dubcovsky J , et al. The HAK1 gene of barley is a member of a large gene family and encodes a high-affinity potassium transporter. Plant Cell, 1997,9(12):2281-2289.
doi: 10.1105/tpc.9.12.2281 |
| [11] |
Kim E J, Kwak J M, Uozumi N , et al. AtKUP1:an Arabidopsis gene encoding high-affinity potassium trans-port activity. Plant Cell, 1998,10(1):51-62.
doi: 10.1105/tpc.10.1.51 |
| [12] |
Zhang Z, Zhang J, Chen Y , et al. Genome-wide analysis and identification of HAK potassium transporter gene family in maize (Zea mays L.). Moleculur Biology Reports, 2012,39(8):8465-8473.
doi: 10.1007/s11033-012-1700-2 |
| [13] |
Yang Z, Gao Q, Sun C , et al. Molecular evolution and functional divergence of HAK potassium transporter gene family in rice (Oryza sative L.). Journul of Genetics and Genomics, 2009,36(3):161-172.
doi: 10.1016/S1673-8527(08)60103-4 |
| [14] |
Bañuelos M A, Garciadeblas B, Band C , et al. Inventory and functional characterization of the HAK potassium transporters of rice. Physiologia Plantarum, 2002,130(2):784-795.
doi: 10.1104/pp.007781 |
| [15] |
张高华, 王鹤, 王旭达 , 等. 獐茅HAK基因表达调控区的分离及其在水稻中的功能分析. 遗传, 2012,34(6):742-748.
doi: 10.3724/SP.J.1005.2012.00742 |
| [16] |
Wang Y H, Garvin D F, Kochian L V . Rapid induction of regulatory and transporter genes in response to phosphorus,potassium,and iron deficiencies in tomato roots. Evidence for cross talk and root/rhizosphere-mediated signals. Physiologia Plantarum, 2002,130(3):1361-1370.
doi: 10.1104/pp.008854 |
| [17] | 王兰 . 璋茅高亲和性K+转运蛋自基因的表达调控 . 大连:大连理工大学, 2009. |
| [18] |
Maathuis F J M . The role of monovalent cation transporters in plant responses to salinity. Journal of Experimental Botany, 2016,57(5):1137-1147.
doi: 10.1093/jxb/erj001 pmid: 16263900 |
| [19] |
Su H, Golldack D, Zhao C S , et al. The expression of HAK-type K+ transporters is regulated in response to salinity stress in common ice plant. Plant Physiology, 2002,129(4):1482-1493.
doi: 10.1104/pp.001149 |
| [20] |
宋毓峰, 张良, 董连红 , 等. 植物KUP/HAK/KT家族钾转运体研究进展. 中国农业科技导报, 2013,15(6):92-98.
doi: 10.3969/j.issn.1008-0864.2013.06.14 |
| [21] | 韦丽君 . 甘蔗干旱胁迫的形态结构及生理机制研究. 长沙:湖南农业大学, 2014. |
| [22] |
苏炜华, 刘峰, 黄珑 , 等. 甘蔗Ca2+/H+反向运转体基因的克隆与表达分析 . 作物学报, 2016,42(7):1074-1082.
doi: 10.3724/SP.J.1006.2016.01074 |
| [23] | 鲁黎明, 杨铁钊 . 烟草钾转运体基因NtHAK1的克隆及表达模式分析. 核农学报, 2011,25(3):469-476. |
| [24] |
Sato Y, Nanatani K, Hamamoto S , et al. Defining membrane spanning domains and crucial membrane-localized acidic amino acid residues for K+ transport of a KUP/HAK/KT-type Escherichia coli potassium transporter . Journal of Biochemistry, 2014,155(5):315-323.
doi: 10.1093/jb/mvu007 |
| [25] | Elumalai R P, Nagpal P, Reed J W . A mutation in the Arabidopsis KT2/KUP2 potassium transporter gene affects shoot cell expansion. Plant Cell, 2002,14(1):119-131. |
| [26] |
Very A A, Sentenac H . Molecular mechanisms and regulation of K+ transport in higher plants . Annual Review of Plant Biology, 2003,54(1):575-603.
doi: 10.1146/annurev.arplant.54.031902.134831 |
| [27] | Gupta M, Qiu X H, Wang L , et al. KT/HAK/KUP potassium transporters gene family and their whole-life cycle expression profile in rice (Oryza sativa). Molecular Genetics and Genomics, 2008,208(5):437-452. |
| [28] | 晁毛妮, 张志勇, 宋海娜 , 等. 陆地棉Pht1家族成员的全基因组鉴定及表达分析. 棉花学报, 2017,29(1):59-69. |
| [29] | 张选 . 胡杨在盐碱胁迫下的生理响应及其钾离子转运基因HAK克隆和功能分析. 北京:中国科学院大学, 2015. |
| [30] |
Osakabe Y, Arinaga N, Umezawa T , et al. Osmotic stressresponses and plant growth controlled by potassium transporters in Arabidopsis. The Plant Cell, 2013,25(2):609-624.
doi: 10.1105/tpc.112.105700 |
| [1] | 朱鹏锦,庞新华,梁春,谭秦亮,严霖,周全光,欧克维. 低温胁迫对甘蔗幼苗活性氧代谢和抗氧化酶的影响[J]. 作物杂志, 2018, (4): 131–137 |
| [2] | 曹哲群,肖芙荣,陈疏影,何丽莲,李富生. 7个蔗茅野生种及其后代材料苗期耐寒性鉴定[J]. 作物杂志, 2017, (5): 43–48 |
| [3] | 李雪妹,刘畅,刘倩雯,张煜笛,李雪梅. PEG预处理对水分胁迫下水稻叶片抗氧化酶同工酶及其表达的影响[J]. 作物杂志, 2016, (6): 107–111 |
| [4] | 石景雨,何丽莲,王先宏,李富生. 不同甘蔗品种叶片中总黄酮含量与提取工艺的优化研究[J]. 作物杂志, 2016, (5): 19–24 |
| [5] | 朱畇昊,爼梦航,苏秀红,董诚明,陈随清. 冬凌草HMGS基因的克隆与表达分析[J]. 作物杂志, 2016, (5): 25–30 |
| [6] | 李钰,郑文寅,冯春,王荣富,李娟. 非生物逆境胁迫下普通小麦烟农19幼苗FeSOD基因表达分析[J]. 作物杂志, 2016, (4): 75–79 |
| [7] | 胡海洋,付青青,何丽莲,李富生. 新型肥料海生源对甘蔗耐寒性的影响[J]. 作物杂志, 2016, (3): 73–78 |
| [8] | 庞新华,朱鹏锦,周全光,谭秦亮,梁春,欧克维,黄强,欧景莉. 广西蔗区引进甘蔗品种(系)的比较试验[J]. 作物杂志, 2016, (2): 73–78 |
| [9] | 杨善, 江永, 周鸿凯, 等. 甘蔗光合因子与品质性状的典型相关性分析[J]. 作物杂志, 2015, (4): 69–73 |
| [10] | 王雪丽, 阮红燕, 黄智刚. 基于AEz模型的广西甘蔗生产潜力分析[J]. 作物杂志, 2015, (1): 121–126 |
| [11] | 沈雪峰, 方越, 董朝霞, 等. 甘蔗/花生间作对土壤微生物和土壤酶活性的影响[J]. 作物杂志, 2014, (5): 55–58 |
| [12] | 李翔, 黄伟华, 黄家雍, 等. 2013年广西甘蔗中期生长情况调查[J]. 作物杂志, 2014, (4): 148–152 |
| [13] | 王红, 杨镇, 肖军, 等. 两种植物内生真菌的醇提取物对水稻根部基因表达谱的分析[J]. 作物杂志, 2014, (2): 48–52 |
| [14] | 向鹏, 龙承波, 罗红丽. 水稻OsMED7基因的克隆及表达分析[J]. 作物杂志, 2013, (3): 21–24 |
| [15] | 黄诚梅, 江文, 杨丽涛, 李杨瑞, 杨翠芳,. 转入反义ScP5CS基因烟草植株对PEG和NaCl处理的反应[J]. 作物杂志, 2013, (3): 42–45 |
|