作物杂志,2019, 第3期: 73–79 doi: 10.16035/j.issn.1001-7283.2019.03.012
农杆菌介导谷子成熟胚遗传转化体系的建立与优化
李颜方,杜艳伟,张正,王高鸿,赵根有,赵晋锋,余爱丽
- 山西省农业科学院谷子研究所,046011,山西长治
Establishment and Optimization of Agrobacterium Mediated Transformation System for Mature Embryo of Foxtail Millet
Li Yanfang,Du Yanwei,Zhang Zheng,Wang Gaohong,Zhao Genyou,Zhao Jinfeng,Yu Aili
- Millet Research Institute, Shanxi Academy of Agricultural Sciences, Changzhi 046011, Shanxi, China
摘要:
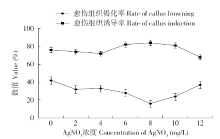
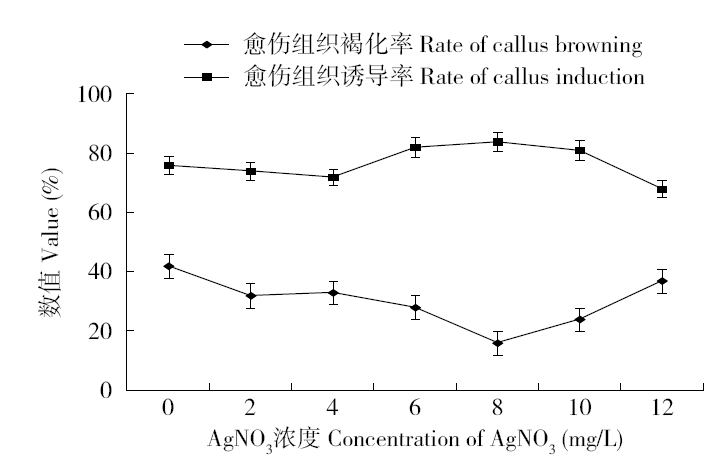
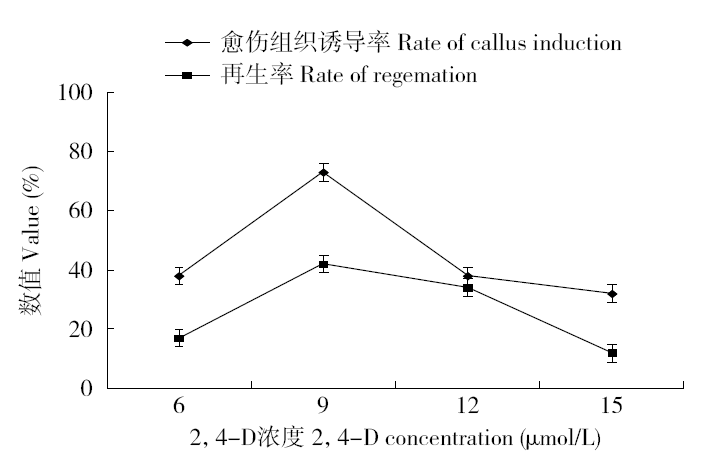
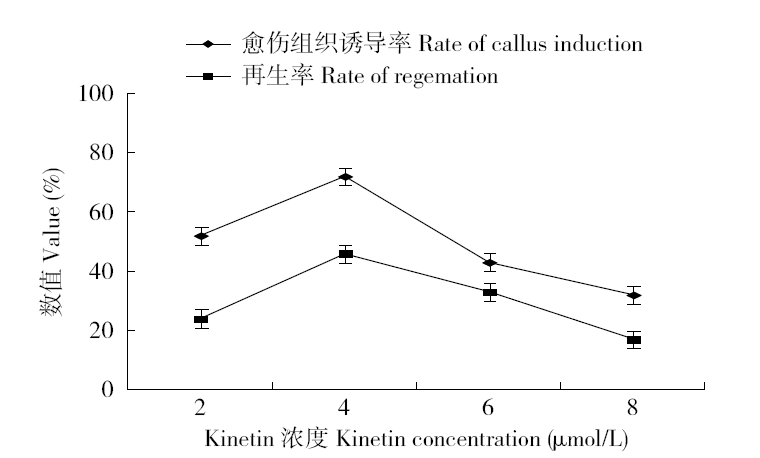
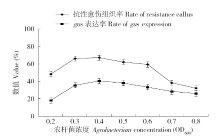
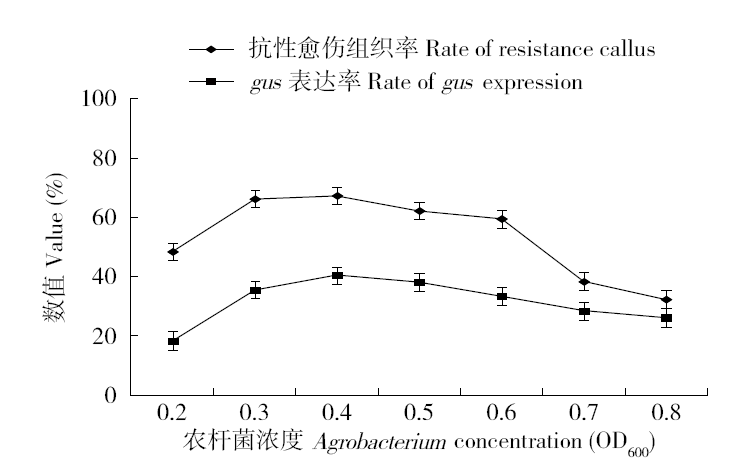
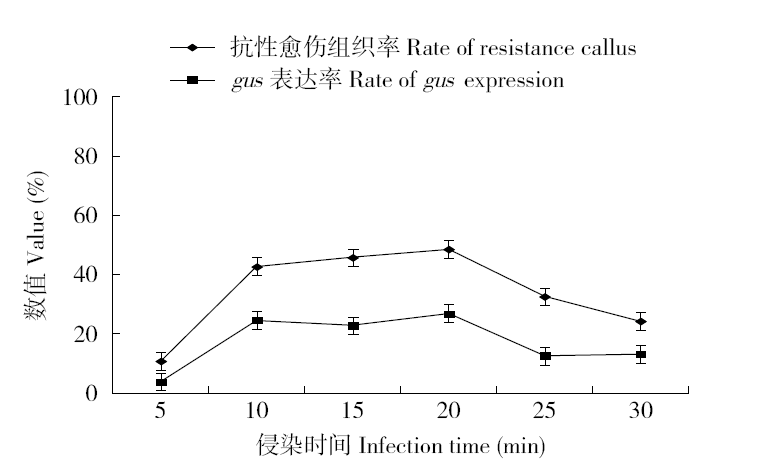
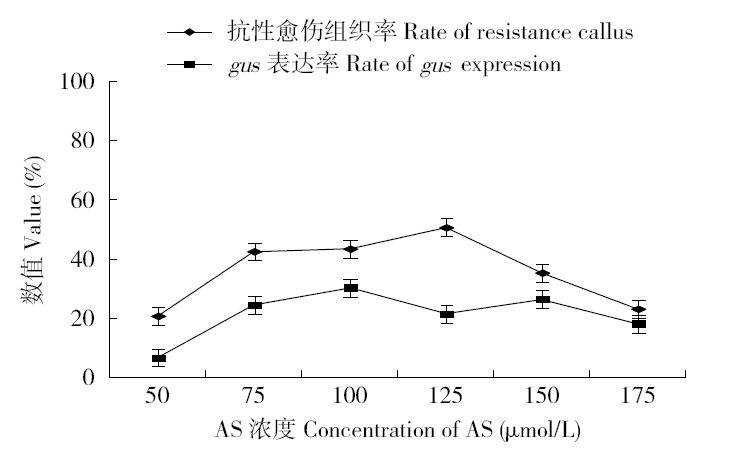
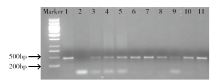
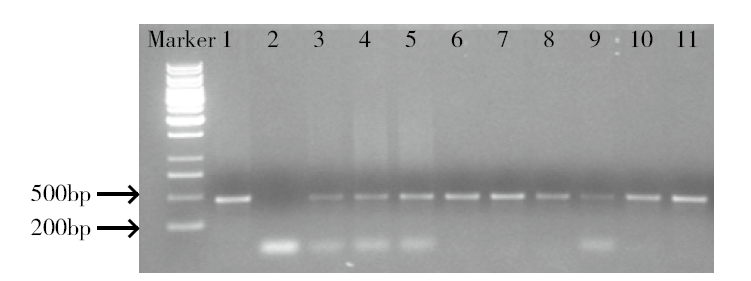
为建立一个稳定的谷子遗传转化体系,对216份谷子种质资源进行筛选,确定材料178成熟胚为受体材料,从植物激素浓度、防褐化剂、农杆菌菌液浓度、侵染时间、乙酰丁香酮(AS)浓度等方面对谷子再生及转化体系进行优化。结果表明,在MS培养基中加入生长素2,4-D 9μmol/L、细胞分裂素Kinetin 4μmol/L、硝酸银8mg/L,愈伤组织诱导率最高且能有效防止褐化。在侵染液和共培养基中加入100μmol/L AS,gus表达效果最好。农杆菌溶液OD600为0.3~0.5,侵染15min,共培养3d时,抗性愈伤组织率最高。获得的转基因植株,经PCR检测及gus染色分析,确定外源bar基因已成功整合到谷子基因组中。初步建立了一套谷子遗传转化体系,为今后谷子遗传转化提供一些参考。
| [1] |
智慧, 牛振刚, 贾冠清 , 等. 谷子干草饲用品质性状变异及相关性分析. 作物学报, 2012,38(5):800-807.
doi: 10.3724/SP.J.1006.2012.00800 |
| [2] |
Jayaraman A, Puranik S, Rai N K , et al. cDNA-AFLP analysis reveals differential gene expression in response to salt stress in foxtail millet (Setaria italica L.). Molecular Biotechnology, 2008,40:241-251.
doi: 10.1007/s12033-008-9081-4 |
| [3] |
Muthamilarasan M, Prasad M . Advances in setaria genomics for genetic improvement of cereals and bioenergy grasses. Theoretical and Applied Genetics, 2015,128(1):1-14.
doi: 10.1007/s00122-014-2399-3 |
| [4] | Taylor M, Vasil I K . GUS gene expression in cereal cell suspensions and immature embryos using the maize ubiquiton-based plasmid pAHC25. Plant Cell Reports, 1993,12:491-495. |
| [5] | 许智宏, 夏镇澳 . 谷子与狗尾草的组织培养. 植物生理学通讯, 1983(2):12-13. |
| [6] | 刁现民, 段胜军, 陈振玲 , 等. 谷子体细胞无性系变异分析. 中国农业科学, 1999,32(3):21-26. |
| [7] | 赵连元, 纪芸, 段胜军 . 谷子原生质体植株再生研究. 华北农学报, 1990(5):27-29. |
| [8] | 王永芳, 李伟, 刁现民 . 根癌农杆菌共培养转化谷子技术体系的建立. 河北农业科学, 2003,7(4):1-6. |
| [9] | 袁进成, 刘颖慧, 董志平 , 等. 谷子成熟胚诱导愈伤组织及植株再生的研究和条件的优化. 生物技术通报, 2013(3):77-82. |
| [10] | 刘颖慧, 于静娟, 赵倩 , 等. 根癌农杆菌介导谷子的遗传转化. 农业生物技术学报, 2005,13(1):32-37. |
| [11] | Jefferson R A . Assaying chimerical gene sin plants:the GUS gene fusion system. Plant Molecular Biology Reports, 1987,11:38-47. |
| [12] | 魏开发, 刘逸萍, 林子英 , 等. 农杆菌介导单子叶植物遗传转化问题与对策. 植物学通报, 2008,25(4):491-496. |
| [13] |
Zhang Y M, Linghu J J, Wang D , et al. Foxtail millet CBL4 (SiCBL4) interacts with SiCIPK24,modulates plant salt stress tolerance. Plant Molecular Biology Reporter, 2017,35:634-646.
doi: 10.1007/s11105-017-1051-1 |
| [14] |
Lakkakula S, Periyasamy R, Arokiam S R , et al. Somatic embryogenesis and regeneration using Gracilaria edulis and Padina boergesenii seaweed liquid extracts and genetic fidelity in finger millet (Eleusine coracana). Journal of Applied Phycology, 2016,28:2083-2098.
doi: 10.1007/s10811-015-0696-0 |
| [15] | 赵巧阳, 赖钟雄 . 硝酸银在离体培养和转化中的作用及其机理. 亚热带农业研究, 2008,4(1):62-66. |
| [16] | 王景雪, 孙毅, 胡晶晶 , 等. 玉米自交系高频率再生因素研究. 作物学报, 2004,30(6):398-402. |
| [17] | 王文星, 屈山, 曹成有 , 等. 硝酸银对离体培养烟草叶片愈伤组织形成和芽再生及其脯氨酸和丙二醛含量的影响. 植物生理学通讯, 2006,42(4):668-670. |
| [18] | 孔维龙, 王翠, 但乃震 , 等. 根癌农杆菌介导的香石竹遗传转化体系建立. 中国农业大学学报, 2018,23(4):34-44. |
| [19] | 朱军, 韩锁义, 袁美 , 等. 农杆菌介导的花生遗传转化条件优化. 中国油料作物学报, 2018,40(2):191-198. |
| [20] | Shweta B, Suresh K . Agrobacterium-mediated transient GUS gene expression in buffel grass (Cenchrus ciliaris L.). Journal of Application Genetics, 2003,44:449-458. |
| [21] |
Smith R, Hood E . Agrobacterium tumefaciens transformation of monocotyledons. Crop Science, 1995,35:301-309.
doi: 10.2135/cropsci1995.0011183X003500020001x |
| [22] |
Stachel S E, Messens E, Montagu M , et al. Identification of the signal molecules produced by wounded plant cell that activate T-DNA transfer in Agrobacterium tumefaciens. Nature, 1985,318:624-629.
doi: 10.1038/318624a0 |
| [23] |
Cheng M, Lowe B A, Speneer T M , et al. Factors in flueneing Agrobacterium-mediated transformation of monoeotyledonous speeies. In Vitro Cellular & Developmental Biology-Plant, 2004,40:31-45.
doi: 10.1079/IVP2003501 |
| [24] |
Shrawat A K, Lorz H . Agrobacterium-mediated transformation of cereals:a promising approach crossing barriers. Plant Biotechnology Journal, 2006,4:575-603.
doi: 10.1111/pbi.2006.4.issue-6 |
| [25] |
Hensel G, Valkov V, Kumlehn J , et al. Efficient generation of transgenic barley:the way forward to modulate plant microbe interactions. Journal of Plant Physiology, 2008,165:71-82.
doi: 10.1016/j.jplph.2007.06.015 |
| [26] |
Ishida Y, Tsunashima M, Hiei Y , et al. Wheat (Triticum aestivum L.) transformation using immature embryos. Agrobacterium Protocols, 2015,1223:189-198.
doi: 10.1007/978-1-4939-1695-5 |
| [27] |
Lakkakula S, Arokiam S R, Ceasar S A , et al. Influence of plant growth regulators and spermidine on somatic embryogenesis and plant regeneration in four Indian genotypes of finger millet (Eleusine coracana (L.) Gaertn). Plant Cell Tissue and Organ Culture, 2016,124(1):15-31.
doi: 10.1007/s11240-015-0870-8 |
| [28] | 李颜方, 赵晋锋, 王高鸿 , 等. 农杆菌介导谷子遗传转化的研究进展. 中国农学通报, 2016,32(36):66-72. |
| [29] |
Sweta D, Chawla H S . In vitro plant regeneration and transformation studies in millets:current status and future prospects. India Journal of Plant Physiology, 2016,21(3):239-254.
doi: 10.1007/s40502-016-0240-5 |
| [30] |
Ceasar S A, Ignacimuthu S, Coll L , et al. Agrobacterium-mediated transformation of finger millet (Eleusine coracana (L.) Gaertn.) using shoot apex explants. Plant Cell Report, 2011,30:1759-1770.
doi: 10.1007/s00299-011-1084-0 |
| [31] |
Martins P K, Ribeiro A P, Dias B A , et al. A simple and highly efficient Agrobacterium-mediated transformation protocol for Setaria viridis. Biotechnology Reports, 2015,6:41-44.
doi: 10.1016/j.btre.2015.02.002 |
| [32] |
Pooja J, Rustagi A, Agnihotri P K , et al. Efficient Agrobacterium-mediated transformation of Pennisetum glaucum (L.) R. Br. using shoot apices as explant source. Plant Cell Tissue and Organ Culture, 2011,107:501-512.
doi: 10.1007/s11240-011-0001-0 |
| [33] |
Saha P, Blumwald E . Spike-dip transformation of Setaria viridis. The Plant Journal, 2016,86(1):89-101.
doi: 10.1111/tpj.2016.86.issue-1 |
| [34] | Lakkakula S, Periyasamy R, Ceasar S A , et al. Effects of cefotaxime,amino acids and carbon source on somatic embryogenesis and plant regeneration in four Indian genotypes of foxtail millet (Setaria italica L.). In Vitro Cellular & Developmental Biology-Plant, 2016,52(2):140-153. |
| [35] | Van E J, Swartwood K, Pidgeon K , et al. Agrobacterium tumefaciens-mediated transformation of Setaria viridis. Plant Genetics and Genomics, 2017,19:343-356. |
| [36] |
Zhang X, Henriques R, Lin S S , et al. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nature Protocols, 2006,1(2):641-646.
doi: 10.1038/nprot.2006.97 |
| [37] |
Martins P K, Nakayama T J, Ribeiro A P , et al. Setaria viridis floral-dip:a simple and rapid Agrobacterium mediated transformation method. Biotechnology Reports, 2015,6:61-63.
doi: 10.1016/j.btre.2015.02.006 |
| [1] | 刘韶光,赵夏童,宋喜娥,原向阳,董淑琦,郭美俊,郭平毅. 膜间喷施芽前除草剂对谷子安全性及对杂草防效的影响[J]. 作物杂志, 2019, (2): 173–178 |
| [2] | 杜艳伟,赵晋锋,王高鸿,李颜方,赵根有,阎晓光. 春播谷子成熟期抗倒伏性研究[J]. 作物杂志, 2019, (1): 141–145 |
| [3] | 王小林,纪晓玲,张盼盼,张雄,张静. 黄土高原旱地谷子品种地上器官干物质分配与产量形成相关性分析[J]. 作物杂志, 2018, (5): 150–155 |
| [4] | 魏萌涵, 解慧芳, 邢璐, 宋慧, 王淑君, 王素英, 刘海萍, 付楠, 刘金荣. 华北地区谷子产量与农艺性状的综合评价分析[J]. 作物杂志, 2018, (4): 42–47 |
| [5] | 李志华,穆婷婷,刘鑫,李会霞,田岗. 4个谷子不育系主要农艺性状的配合力分析[J]. 作物杂志, 2018, (3): 61–67 |
| [6] | 岳茂林,薛蔚荣,张瑞栋,岳忠孝,吕瑞洲,郭鹏燕. 不同行距配置对谷子农艺性状及产量的影响[J]. 作物杂志, 2018, (2): 93–96 |
| [7] | 张微,王良群,刘勇,郝艳芳,杨伟,白鸿雁,武擘. 农杆菌介导高粱遗传转化的相关因素优化[J]. 作物杂志, 2018, (1): 56–61 |
| [8] | 孙冬雪,张爱军. 底施锌肥对谷子抗氧化性及干物质的影响[J]. 作物杂志, 2017, (6): 160–164 |
| [9] | 郭瑞锋,张永福,任月梅,杨忠. 混合盐碱胁迫对谷子萌发、幼芽生长的影响及耐盐碱品种筛选[J]. 作物杂志, 2017, (4): 63–66 |
| [10] | 宋国亮,冯小磊,范光宇,史高雷,李双东,王峰,王晓明,赵治海. 谷子新不育系的配合力分析[J]. 作物杂志, 2017, (2): 44–50 |
| [11] | 代小冬,朱灿灿,秦娜,杨育峰,王雁楠,杨国红,司冰,刘诗慧,李君霞. 烯效唑和密度对谷子产量及产量相关性状的影响[J]. 作物杂志, 2017, (2): 104–108 |
| [12] | 邱双,闫双堆,刘利军. 不同谷子品种耐低磷能力研究[J]. 作物杂志, 2017, (2): 139–144 |
| [13] | 赵丽娟,马金丰,李延东,李祥羽,李志江,袁红梅,郭文栋. 60Co-γ射线辐射谷子干种子诱变效应的研究[J]. 作物杂志, 2017, (1): 38–43 |
| [14] | 穆婷婷,杜慧玲,景小兰,李志华,郭琦,田岗,李会霞,刘璋. 外源硒对谷子产量因子及硒含量的影响[J]. 作物杂志, 2017, (1): 73–78 |
| [15] | 李志华,景小兰,李会霞,田岗,刘鑫,穆婷婷. 谷子苗期除草剂的安全性及杂草防效研究[J]. 作物杂志, 2017, (1): 150–154 |
|