作物杂志,2022, 第4期: 90–98 doi: 10.16035/j.issn.1001-7283.2022.04.013
玉米杂交种及其亲本苗期性状对低氮胁迫的动态响应
- 山西农业大学农学院/黄土高原特色作物优质高效生产省部共建协同创新中心,030801,山西晋中
Dynamic Changes of Seedling Traits among Maize Hybrids and Their Parents in Response to Low Nitrogen Stress
Xu Shiying( ), Wang Ning, Cheng Hao, Feng Wanjun(
), Wang Ning, Cheng Hao, Feng Wanjun( )
)
- College of Agriculture, Shanxi Agricultural University/Ministerial and Provincial Co-Innovation Center for Endemic Crops Production with High-Quality and Efficiency in Loess Plateau, Jinzhong 030801, Shanxi, China
摘要:
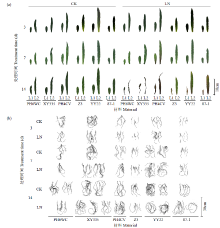
以玉米杂交种先玉335及其亲本(PH6WC、PH4CV)和豫玉22及其亲本(综3、87-1)为试验材料,通过水培方法,设置低氮(0.04mmol/L,LN)和正常氮素(2mmol/L,CK)水平2个处理,分别在培养3、7和14d后,对其幼苗生物量积累、叶片和根系表型、叶绿素及氮素含量等进行分析,探究低氮胁迫下玉米杂交种及其亲本幼苗性状的动态变化。结果表明,玉米苗期根系对低氮的响应要早于地上部,且杂交种对低氮的响应比亲本更为迅速。在CK和LN处理下,杂交种幼苗的多个性状均存在中亲和超亲优势,豫玉22的根尖数、根系氮积累量占比和先玉335的总根长、根表面积及根尖数的杂种优势在3个时间点与CK处理相比均有较大幅度提高。通过耐低氮指数分析发现,杂交种的耐低氮能力介于2个亲本之间或与其中1个亲本接近。
| [1] | George H S. The composition of a field of maize. Journal of Heredity, 1908, 4(1):296-301. |
| [2] |
Birchler J A, Auger D L, Riddle N C. In search of the molecular basis of heterosis. Plant Cell, 2003, 15(10):2236-2239.
pmid: 14523245 |
| [3] |
Hochholdinger F, Hoecker N. Towards the molecular basis of heterosis. Trends in Plant Science, 2007, 12(9):427-432.
pmid: 17720610 |
| [4] | Suresh K S, Renu K. Physiological,biochemical,and genetic basis of heterosis. Advances in Agronomy, 1975, 27:123-174. |
| [5] |
Tollenaar M A, Ahmadzadeh E A. Physiological basis of heterosis for grain yield in maize. Crop Science, 2004, 44(6):2086-2094.
doi: 10.2135/cropsci2004.2086 |
| [6] | Tollenaar M A, Lee E A. Dissection of physiological processes underlying grain yield in maize by examining genetic improvement and heterosis. Maydica, 2006, 51(2):399-408. |
| [7] |
Wang F H. Embryological development of inbred and hybrid Zea mays L.. American Journal of Botany, 1947, 34(3):113-125.
doi: 10.1002/j.1537-2197.1947.tb12966.x |
| [8] |
Meyer S, Pospisil H, Scholten S. Heterosis associated gene expression in maize embryos 6 days after fertilization exhibits additive,dominant and overdominant pattern. Plant Molecular Biology, 2007, 63(3):381-391.
doi: 10.1007/s11103-006-9095-x |
| [9] |
Hoecker N, Keller B, Piepho H P, et al. Manifestation of heterosis during early maize (Zea mays L.) root development. Theoretical and Applied Genetics, 2006, 112(3):421-429.
pmid: 16362278 |
| [10] |
Hoecker N, Keller B, Muthreich N, et al. Comparison of maize (Zea mays L.) F1-hybrid and parental inbred line primary root transcriptomes suggests organ-specific patterns of nonadditive gene expression and conserved expression trends. Genetics, 2008, 179(3):1275-1283.
doi: 10.1534/genetics.108.088278 pmid: 18562640 |
| [11] |
Paschold A, Marcon C, Hoecker N, et al. Molecular dissection of heterosis manifestation during early maize root development. Theoretical and Applied Genetics, 2010, 120(2):383-388.
doi: 10.1007/s00122-009-1082-6 pmid: 19526205 |
| [12] | Springer N M, Stupar R M. Allelic variation and heterosis in maize:how do two halves make more than a whole?. Genome Research, 2007, 17(3):264-275. |
| [13] |
Araus J L, Sanchez C, Cabrera-Bosquet L. Is heterosis in maize mediated through better water use?. New Phytologist, 2010, 187(2):392-406.
doi: 10.1111/j.1469-8137.2010.03276.x pmid: 20456048 |
| [14] |
Liu W D, Tollenaar M. Response of yield heterosis to increasing plant density in maize. Crop Science, 2009, 49(5):1807-1816.
doi: 10.2135/cropsci2008.07.0422 |
| [15] | 李潮海, 尹飞, 王群. 不同耐旱性玉米杂交种及其亲本叶片活性氧代谢对水分胁迫的响应. 生态学报, 2006(6):1912-1919. |
| [16] | 唐连顺, 李广敏. 干旱对玉米杂交种及其亲本自交系幼苗膜脂过氧化及其保护酶活性的影响. 作物学报, 1995(4):509-512. |
| [17] | 马建华, 孙毅, 王玉国, 等. 低磷胁迫对玉米自交系及其杂交种苗期生理特性的影响. 山西农业科学, 2014, 42(3):220-222. |
| [18] | 陈范骏, 米国华, 春亮, 等. 玉米氮效率的杂种优势分析. 作物学报, 2004(10):1014-1018. |
| [19] |
Wang Z, Ma B L, Yu X, et al. Physiological basis of heterosis for nitrogen use efficiency of maize. Scientific Reports, 2019, 9(1):18708.
doi: 10.1038/s41598-019-54864-x |
| [20] | Duvick D N. Genetic progress in yield of United States maize (Zea mays L.). Maydica, 2005, 50(3):193-202. |
| [21] |
Han M, Okamoto M, Beatty P H, et al. The genetics of nitrogen use efficiency in crop plants. Annual Review of Genetics, 2015, 49:269-289.
doi: 10.1146/annurev-genet-112414-055037 |
| [22] | Good A G, Beatty P H. Fertilizing nature:A tragedy of excess in the commons. Working Papers, 2011, 9(8):e1001124. |
| [23] |
Mulvaney R L, Khan S A, Ellsworth T R. Synthetic nitrogen fertilizers deplete soil nitrogen:a global dilemma for sustainable cereal production. Journal of Environmental Quality, 2009, 38(6):2295-2314.
doi: 10.2134/jeq2008.0527 pmid: 19875786 |
| [24] |
Li H, Hu B, Chu C. Nitrogen use efficiency in crops:lessons from Arabidopsis and rice. Journal of Experimental Botany, 2017, 68(10):2477-2488.
doi: 10.1093/jxb/erx101 |
| [25] | 赵泽群, 师赵康, 王雯, 等. 低氮胁迫下玉米幼苗氮素和蔗糖分配特性. 植物营养与肥料学报, 2020, 26(4):783-796. |
| [26] | 冯万军, 张义荣, 姚颖垠, 等. 玉米杂交种与亲本苗期根系蛋白差异表达谱分析. 自然科学进展, 2009, 19(6):619-627. |
| [27] | 薛玲珠, 孙敏, 高志强, 等. 深松蓄水增量播种对旱地小麦植株氮素吸收利用、产量及蛋白质含量的影响. 中国农业科学, 2017, 50(13):2451-2462. |
| [28] | 胡成梅, 连盈, 程鹏飞, 等. 小麦苗期性状与耐低氮性的遗传相关分析. 中国农业大学学报, 2020, 25(4):11-22. |
| [29] | Li H, Sun Y, Yu X, et al. Effects of exogenous calcium on the growth and physiological traits of garlic seedlings under cadmium stress. Journal of Animal and Plant Sciences, 2015, 25(3):107-113. |
| [30] |
Procházková D, Sairam R K, Lekshmy S, et al. Differential response of maize hybrid and its parental lines to salinity stress. Czech Journal of Genetics and Plant Breeding, 2013, 54:9-15.
doi: 10.17221/30/2017-CJGPB |
| [31] |
Betrán F J, Beck D, Bänziger M, et al. Secondary traits in parental inbreds and hybrids under stress and non-stress environments in tropical maize. Field Crops Research, 2003, 83(1):51-65.
doi: 10.1016/S0378-4290(03)00061-3 |
| [32] |
Sabermanesh K, Holtham L R, George J, et al. Transition from a maternal to external nitrogen source in maize seedlings. Journal of Integrative Plant Biology, 2017, 59(4):261-274.
doi: 10.1111/jipb.12525 |
| [33] |
Li P, Zhuang Z, Cai H, et al. Use of genotype-environment interactions to elucidate the pattern of maize root plasticity to nitrogen deficiency. Journal of Integrative Plant Biology, 2016, 58(3):242-253.
doi: 10.1111/jipb.12384 |
| [34] |
Chun L, Mi G, Li J, et al. Genetic analysis of maize root characteristics in response to low nitrogen stress. Plant and Soil, 2005, 276(1-2):369-382.
doi: 10.1007/s11104-005-5876-2 |
| [35] | Liu J, Li J, Chen F, et al. Mapping QTLs for root traits under different nitrate levels at the seedling stage in maize (Zea mays L.). 2008, 305(1):253-265. |
| [36] |
Zhan A, Lynch J P. Reduced frequency of lateral root branching improves N capture from low-N soils in maize. Journal of Experimental Botany, 2015, 66(7):2055-2065.
doi: 10.1093/jxb/erv007 pmid: 25680794 |
| [1] | 王元元, 谷子寒, 陈平平, 易镇邪. 镉污染稻田玉米对水稻的季节性替代种植可行性研究[J]. 作物杂志, 2022, (4): 187–192 |
| [2] | 王家保, 冀怀远, 梅家法, 陶志国, 疏志峰, 江三桥. 玉米新品种荃科玉900的选育与栽培制种技术[J]. 作物杂志, 2022, (4): 267–270 |
| [3] | 雷蕾, 关哲允, 曹士亮, 王玉民, 林春晶, 彭宝, 刘鹏, 赵丽梅, 李志刚, 张春宝. 基于产量相关性状SSR分子标记的大豆杂种优势群划分[J]. 作物杂志, 2022, (4): 54–61 |
| [4] | 顾银山, 张士龙, 贾海涛, 李小琴, 贺正华, 焦春海, 田小海, 黄益勤, 危文亮. 超甜玉米果皮柔嫩度与成分含量动态变化及相互关系[J]. 作物杂志, 2022, (3): 134–142 |
| [5] | 杨奥军, 常巧玲, 王鹏, 王芳, 高妍婷, 周广阔, 宋小佳, 韦恩成. 外源5-ALA对干旱胁迫下玉米种子萌发及幼苗生长的影响[J]. 作物杂志, 2022, (3): 194–199 |
| [6] | 张军, 陈顺全, 张文庆, 李高超, Bell. 10份玉米品种在喀麦隆的适应性研究[J]. 作物杂志, 2022, (3): 87–91 |
| [7] | 曹丽茹, 鲁晓民, 王国瑞, 党尊, 邱天, 邱建军, 田云峰, 王振华, 党永富. 叶面喷施炭吸附聚谷氨酸对玉米生长发育的影响[J]. 作物杂志, 2022, (2): 158–166 |
| [8] | 李忠南, 王越人, 车丽梅, 邬生辉, 曲海涛, 宋涛, 李福林, 李光发. 通玉179六世代和DH世代4个穗部性状对比分析研究[J]. 作物杂志, 2022, (2): 64–68 |
| [9] | 房孟颖, 闫鹏, 卢霖, 王庆燕, 董志强. 乙矮合剂对不同氮水平夏玉米氮代谢及产量的调控效应[J]. 作物杂志, 2022, (2): 96–103 |
| [10] | 刘子刚, 卢海博, 武敏桦, 赵海超, 魏东, 黄智鸿. 化控剂玉黄金对春玉米抗倒伏性状及产量的影响[J]. 作物杂志, 2022, (1): 142–146 |
| [11] | 段琉颖, 吴婷, 李霞, 谢建坤, 胡标林. 水稻细胞质雄性不育及其育性恢复基因的研究进展[J]. 作物杂志, 2022, (1): 20–30 |
| [12] | 段雅娟, 曹士亮, 于滔, 李文跃, 杨耿斌, 王成波, 刘宝民, 刘长华. 玉米自交系萌发期耐盐性鉴定[J]. 作物杂志, 2022, (1): 213–219 |
| [13] | 冯素芬, 刘元剑, 许蕊淇, 张薇. 云南省近年审定鲜食玉米品种的主要性状分析[J]. 作物杂志, 2022, (1): 220–226 |
| [14] | 周德龙, 孟令聪, 郑淑波, 王楠, 李穆, 王薪淇, 卢实, 王敏, 刘文国, 路明. 一种快速高效检测转基因玉米方法的建立[J]. 作物杂志, 2022, (1): 65–69 |
| [15] | 黄莉群, 马玥, 戚新蕾, 孙秋玉, 董佳玉, 李冠甲, 马占鸿. 玉米品种对不同地区玉米南方锈菌的抗性评价[J]. 作物杂志, 2021, (6): 205–210 |
|
||