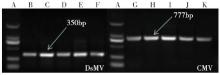
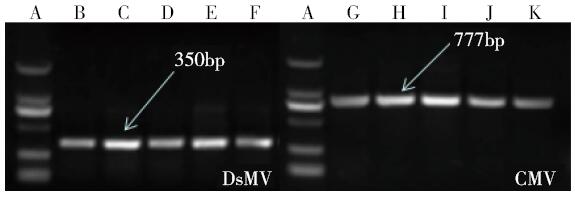
Crops ›› 2016, Vol. 32 ›› Issue (3): 51-57.doi: 10.16035/j.issn.1001-7283.2016.03.010
Previous Articles Next Articles
Establishment of Virus-Elimination and Rapid Propagation System of Pinellia pedatisecta Schott
Li Hui,Zhang Geng,Meng Yijiang,Ge Shujun
- College of Agronomy,Agricultural University of Hebei/North China Key Laboratory for Crop Germplasm Resources of Education Ministry,Baoding 071001,Hebei,China
| [1] | 中国药用植物志编委会. 中国药用植物志(第十二卷).北京:北京大学医学出版社, 2013: 95-97. |
| [2] | 李永伟, 桂明, 刘文洪 , 等. 天南星科植物病毒研究:芋花叶病毒(DsMV)与黄瓜花叶病毒(CMV)分子检测和脱病毒处理.//中国植物病理学会.中国植物病理学会第六届青年学术研讨会论文集—植物病理学研究进展(第五卷). 中国植物病理学会, 2003: 2. |
| [3] | Zettler F W, Hartman R D . Dasheen mosaic virus as a pathogen of cultivated aroids and control of the virus by tissue culture. Plant Disease, 1987,71:958-963. |
| [4] | Luis F S( 阎文昭等译). 马铃薯病毒及其防治.北京:中国农业科技出版社, 2000: 183-184. |
| [5] | Zilkah S, Evgenia F, Rotbaum A , et al. In vitro micropropagation of indicator plants for indexing Prunus necrotic ring spot virus. Acta Horticulturae , 1993,336:121-125. |
| [6] | 陈君帜, 李青 .李属植物脱毒技术及病毒检测研究进展.北京林业大学学报,2001(5):71-74. |
| [7] | 林蓉, 谢春梅, 谢世清 .马铃薯茎尖脱毒培养关键因子分析.中国农学通报,2005(7):338-340. |
| [8] | 齐恩芳, 王一航, 张武 , 等.马铃薯茎尖脱毒培养方法优化研究.中国马铃薯,2007(4):200-203. |
| [9] | 王娟, 汪仲敏, 王瑞英 , 等.马铃薯茎尖脱毒培养影响因素研究.中国马铃薯,2010(3):172-175. |
| [10] | 刘志文, 陈阳, 侯英敏 .不同培养基和培养条件对脱毒马铃薯快繁生长的影响.中国农学通报,2011(24):179-182. |
| [11] | 欧建龙, 黄振霖, 赵雨佳 , 等.几种因素对马铃薯试管薯诱导的影响.中国马铃薯,2009(2):94-95. |
| [12] |
陈春伶, 徐美隆, 李永华 .马铃薯试管薯诱导体系研究.中国农学通报,2012(27):53-56.
doi: 10.3969/j.issn.1000-6850.2012.27.011 |
| [13] |
胡建军, 何卫, 王克秀 , 等.马铃薯脱毒种薯快繁技术及其数量经济关系研究.西南农业学报,2008(3):737-740.
doi: 10.3969/j.issn.1001-4829.2008.03.042 |
| [14] |
谢宏峰, 吴菊香, 迟玉成 , 等.双重RT-PCR方法检测花生条纹病毒和黄瓜花叶病毒.花生学报,2012(1):16-20.
doi: 10.3969/j.issn.1002-4093.2012.01.006 |
| [15] | 陈集双 . 天南星科植物病毒的分子诊断和半夏研究.杭州:浙江大学出版社, 2006: 24-25. |
| [16] | 徐平东, 周仲驹, 林奇英 , 等.黄瓜花叶病毒亚组Ⅰ和Ⅱ分离物外壳蛋白基因的序列分析与比较.病毒学报,1999(2):164-170. |
| [17] | 陈耀锋 . 植物组织与细胞培养.北京:中国农业出版社, 2012: 58. |
| [18] | 孙秀梅 . 马铃薯茎尖剥离脱毒效果的影响因素分析.中国马铃薯,2005(4):226-227. |
| [19] | 王海丽 . 三叶半夏脱毒快繁及离体块茎诱导. 杭州:浙江大学, 2005. |
| [20] |
赵庆芳, 马平霞 .百合脱毒及病毒鉴定的研究进展.北方园艺,2007(3):77-79.
doi: 10.3969/j.issn.1001-0009.2007.03.030 |
| [21] |
Bhardwaj S V, Rai S J, Thakur P D , et al. Meristem tip culture and heat therapy for production of apple mosaic virus free plants in India. Acta Horticulturae, 1998,472:135-142.
doi: 10.17660/ActaHortic.1998.472.13 |
| [22] | 高尚士 . 花卉病毒病的检疫及其消除方法.植物检疫,1994(6):340-341. |
| [23] |
解红娥, 解晓红, 李江辉 , 等.半夏的病毒危害及脱毒快繁技术研究.中草药,2005(11):1697-1700.
doi: 10.7501/j.issn.0253-2670.2005.11.2005011738 |
| [24] | 陈芳, 党占海, 张建平 , 等.不同基因型亚麻下胚轴不定芽诱导的研究.作物杂志,2014(3):39-43,153. |
| [25] | 王罡, 张栩, 王萍 , 等.黄瓜子叶节不定芽的诱导及对甘露糖耐性的研究.作物杂志,2014(6):32-35,161. |
| [26] |
Xu X, André A M, Lammeren V , et al. The role of gibberellin,abscisic acid,and source in the regulation of potato tuber formation in virto. Plant Physiology, 1998,117:575-584.
doi: 10.1104/pp.117.2.575 |
| [27] |
柳俊, 谢从华 .马铃薯块茎发育机理及其基因表达.植物学通报,2001(5):531-539.
doi: 10.3969/j.issn.1674-3466.2001.05.003 |
| [28] |
盛玮, 薛建平, 张爱民 , 等.半夏试管块茎形成过程中内源激素的变化.中国中药杂志,2010(8):943-946.
doi: 10.4268/cjcmm20100801 |
| [29] | 程霞英, 牛振刚, 邵红军 , 等.三叶半夏离体块茎诱导研究.天津农业科学,2010(2):29-31. |
| [30] |
连勇, 邹颖, 东惠茹 , 等.马铃薯试管薯形成过程中几种内源激素的变化.园艺学报,2002(6):537-541.
doi: 10.3321/j.issn:0513-353X.2002.06.008 |
| [1] | Geng Zhang,Yijiang Meng,Xiaosha Jin,Shujun Ge. Effects of Seed Stem Type on Yield and Quality of Medicinal Plant Pinellia pedatisecta Schott [J]. Crops, 2017, 33(2): 168-172. |
| [2] | Xiaoliang Li,Junyun Zhang,Zhong Zhang,Chunfu Dong,Shixian Yang,Wenzhi Wang,Jiankang Zhang. Study on Technology of Rapid Propagation in Tissue Culture of Cut Rose Hongchun [J]. Crops, 2016, 32(6): 58-66. |
| [3] | Xiaoliang Li,Junyun Zhang,Zhong Zhang,Cuiping Zhang,Shixian Yang,Wenzhi Wang,Jiankang Zhang,Yonghua Duan. Tissue Culture and Rapid Propagation of Strawberry [J]. Crops, 2016, 32(4): 68-74. |
|