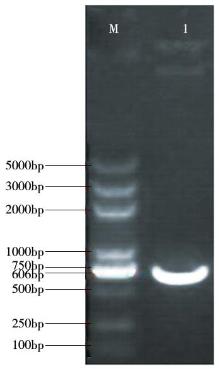
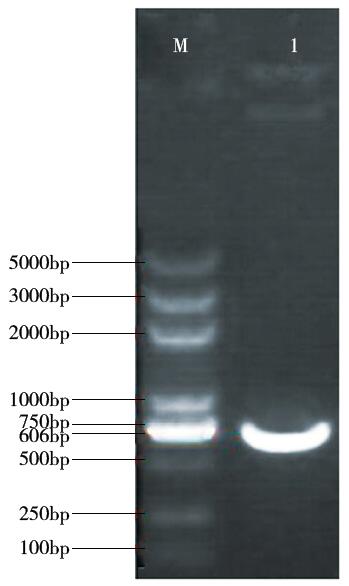
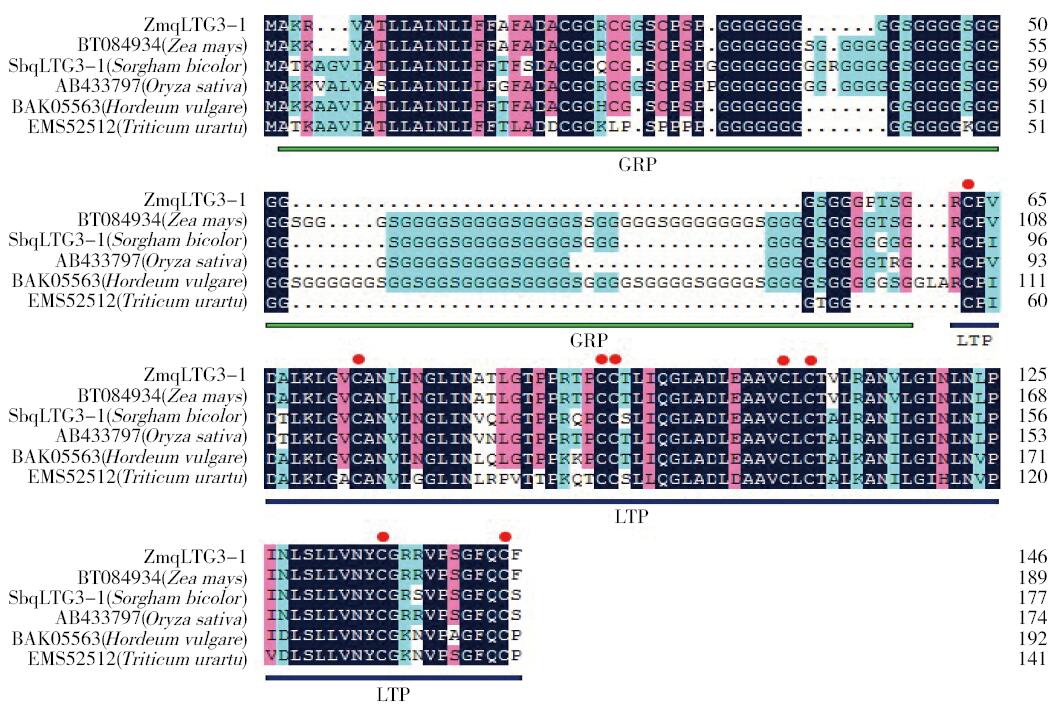
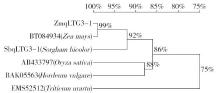
Crops ›› 2016, Vol. 32 ›› Issue (4): 47-55.doi: 10.16035/j.issn.1001-7283.2016.04.008
Previous Articles Next Articles
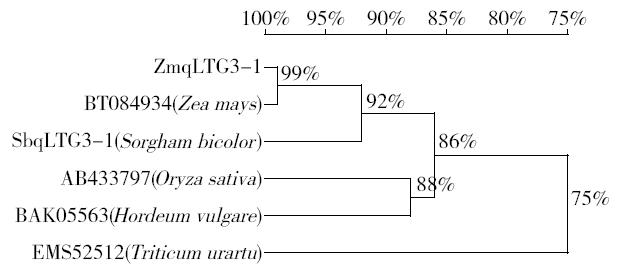
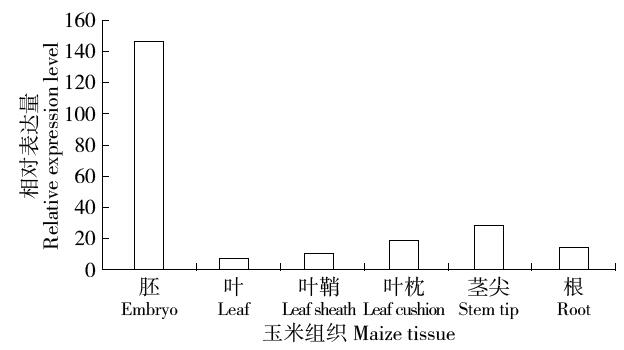
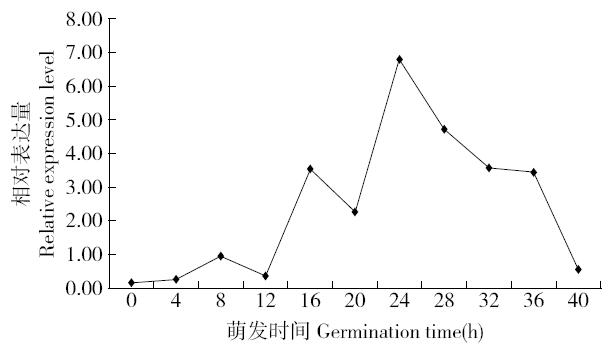
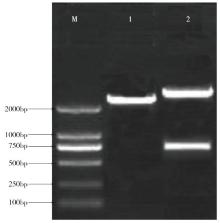
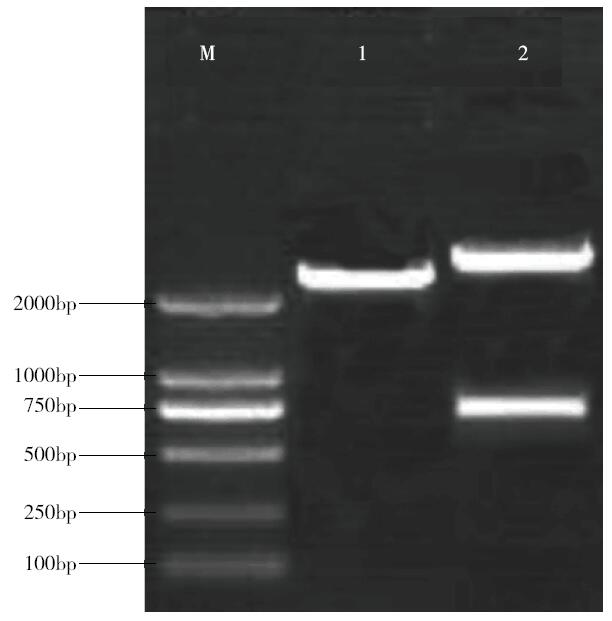
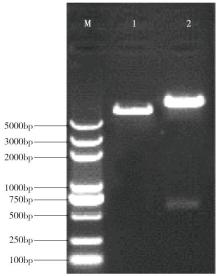
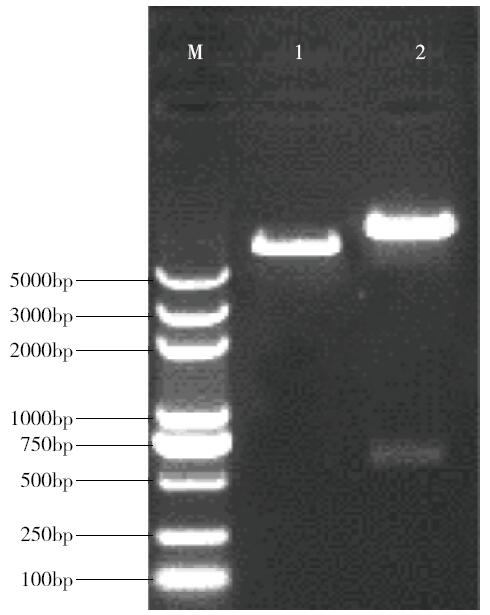
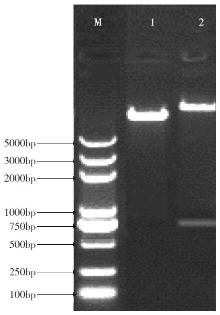
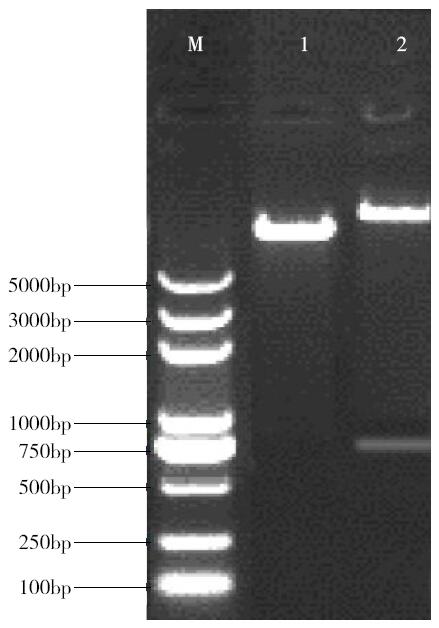
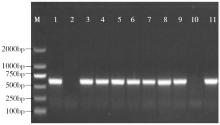
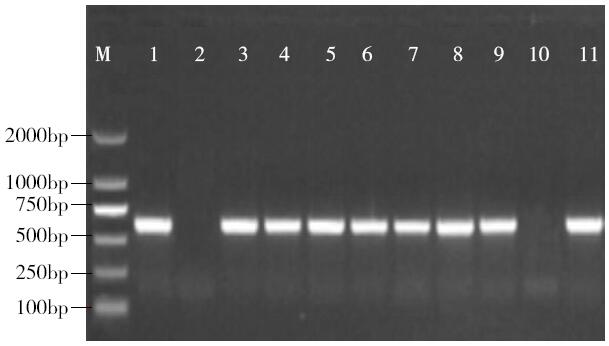
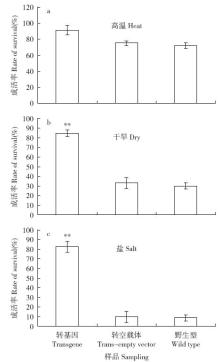
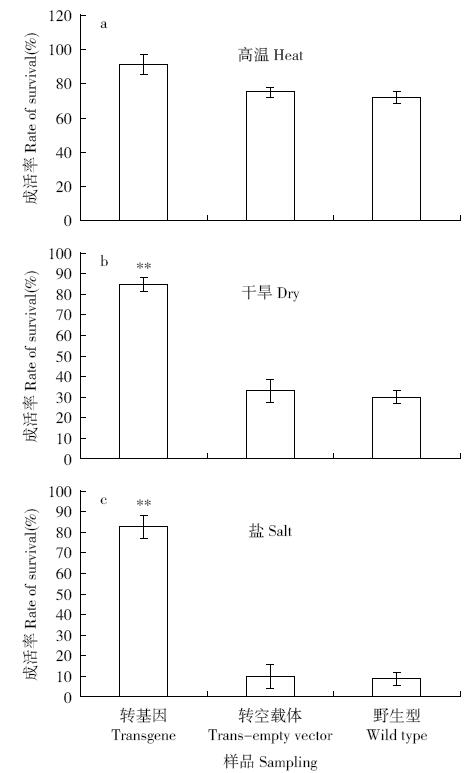
Cloning and Function Analysis of Resistance Gene ZmqLTG3-1 in Maize
Han Zanping1,2,Chen Yanhui2,Guo Shulei2,Zu Xiaofeng2,Wang Shunxi2,Zhao Xiyong2
- 1 Agricultural College,Henan University of Science and Technology,Luoyang 471003,Henan,China
2 Agronomy College of Henan Agricultural University,Zhengzhou 450002,Henan,China
| [1] | 关贤交, 欧阳西荣 .玉米低温冷害研究进展.作物研究,2004(5):353-357. |
| [2] | 史占忠, 贲显明, 张敬涛 , 等.三江平原春玉米低温冷害发生规律及防御措施.黑龙江农业科学,2003(2):7-10. |
| [3] |
Ashfield T, Bocian A, Held D , et al. Genetic and physical localization of the soybean Rpg1-b disease resistance gene reveals a complex locus containing several tightly linked families of NBS-LRR genes. Molecular Plant-Microbe Interactions, 2003,16(9):817-826.
doi: 10.1094/MPMI.2003.16.9.817 |
| [4] |
Radwan O, Bouzidi M F, Nicolas P , et al. Development of PCR markers of the PI5/PI8 locus for resistance to plasmopara halstedii in sunflower,Helianthus annuus L.from complete CC-NBS-LRR sequences. Theoretical and Applied Genetics, 2004,109(1):176-185.
doi: 10.1007/s00122-004-1613-0 |
| [5] | 祖小峰 .玉米促分裂原活化蛋白激酶ZmMAPK4的克隆及功能研究. 郑州:河南农业大学, 2013. |
| [6] |
Fujino K, Sekiguchi H, Matsuda Y , et al. Molecular identification of a major quantitative trait locus,qLTG3-1,controlling low-temperature germinability in rice. Proceedings of the National Academy of Sciences of the United States of America, 2008,105(34):12623-12628.
doi: 10.1073/pnas.0805303105 |
| [7] |
Fujino K, Matsuda Y . Genome-wide analysis of genes targeted by qLTG3-1 controlling low-temperature germinability in rice. Plant Molecular Biology, 2010,72:137-152.
doi: 10.1007/s11103-009-9559-x |
| [8] |
Fujino K, Sekiguchi H . Origins of functional nucleotide polymorphisms in a major quantitative trait locus,qLTG3-1,controlling low-temperature germinability in rice. Plant Molecular Biology, 2011,75:1-10.
doi: 10.1007/s11103-010-9697-1 |
| [9] |
Chen F, Bradford K J . Expression of an expansin is associated with endosperm weakening during tomato seed germination. Plant Physiology, 2000,124(3):1265-1274.
doi: 10.1104/pp.124.3.1265 |
| [10] |
Chen F, Nonogaki H, Bradford K J . A gibberellin-regulated xyloglucan endotransglycosylase gene is expressed in the endosperm cap during tomato seed germination. Journal of Experimental Botany, 2002,53(367):215-223.
doi: 10.1093/jexbot/53.367.215 |
| [11] |
Ogawa M, Hanada A, Yamauchi Y , et al. Gibberellin biosynthesis and response during Arabidopsis seed germination. The Plant Cell, 2003,15(7):1591-1604.
doi: 10.1105/tpc.011650 |
| [12] |
Bethke P C, Libourel I G L,Aoyama N, et al. The Arabidopsis aleurone layer responds to nitric oxide,gibberellin,and abscisic acid and is sufficient and necessary for seed dormancy. Plant Physiology, 2007,143(3):1173-1188.
doi: 10.1104/pp.106.093435 |
| [13] | Nonogaki H, Chen F, Bradford K J . Mechanisms and genes involved in germination sensu stricto. Annual Plant Reviews, 2007,27:264-304. |
| [14] |
Qing D J, Lu H F, Li N , et al. Comparative profiles of gene expression in leaves and roots of maize seedlings under conditions of salt stress and the removal of salt stress. Plant and Cell Physiology, 2009,50(4):889-903.
doi: 10.1093/pcp/pcp038 |
| [15] |
Adie B A T, Perez-Perez J, Perez-Perez M M , et al. ABA is an essential signal for plant resistance to pathogens affecting JA biosynthesis and the activation of defenses in Arabidopsis. The Plant Cell, 2007,19:1665-1681.
doi: 10.1105/tpc.106.048041 |
| [16] |
Mohr M, Krustrup P, Nielsen J J , et al. Effect of two different intense training regimens on skeletal muscle ion transport proteins and fatigue development. American Journal of Physiology Regulatory Integrative and Comparative Physiology, 2007,292:1594-1602.
doi: 10.1152/ajpregu.00251.2006 |
| [17] | Liu T, Carlsson J, Takeuchi T , et al. Direct regulation of abiotic responses by the Arabidopsis circadian clock component PRR7. The Plant Journal, 2013,76:101-114. |
| [1] | Haibin Luo, Shengli Jiang, Chengmei Huang, Huiqing Cao, Zhinian Deng, Kaichao Wu, Lin Xu, Zhen Lu, Yuanwen Wei. Cloning and Expression of ScHAK10 Gene in Sugarcane [J]. Crops, 2018, 34(4): 53-61. |
| [2] | Hongmei Yuan,Wendong Guo,Lijuan Zhao,Ying Yu,Jianzhong Wu,Lili Cheng,Dongsheng Zhao,Qinghua Kang,Wengong Huang,Yubo Yao,Xixia Song,Weidong Jiang,Yan Liu,Tingfen Ma,Guangwen Wu,Fengzhi Guan. Cloning and Expression Analysis of the Glycosyltransferase Gene LuUGT72E1 in Flax [J]. Crops, 2016, 32(4): 62-67. |
|