作物杂志,2023, 第1期: 38–45 doi: 10.16035/j.issn.1001-7283.2023.01.006
干旱胁迫及复水对马铃薯糖代谢途径中基因表达影响的研究
- 1内蒙古师范大学生命科学与技术学院,010022,内蒙古呼和浩特
2内蒙古自治区高等学校生物多样性保护与可持续利用重点实验室,010022,内蒙古呼和浩特
3内蒙古农业大学农学院,010020,内蒙古呼和浩特
Research on the Effects of Gene Expression in Sugar Metabolism Pathway of Potato by Drought Stress and Rehydration
Liu Sujun1,2( ), Meng Meilian3, Suriguga 1,2(
), Meng Meilian3, Suriguga 1,2( )
)
- 1College of Life Science and Technology, Inner Mongolia Normal University, Hohhot 010022, Inner Mongolia, China
2Key Laboratory of Biodiversity Conservation and Sustainable Utilization for College and University of Inner Mongolia Autonomous Region, Hohhot 010022, Inner Mongolia, China
3College of Agronomy, Inner Mongolia Agricultural University, Hohhot 010020, Inner Mongolia, China
摘要:
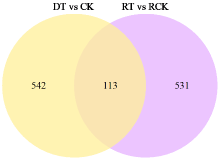
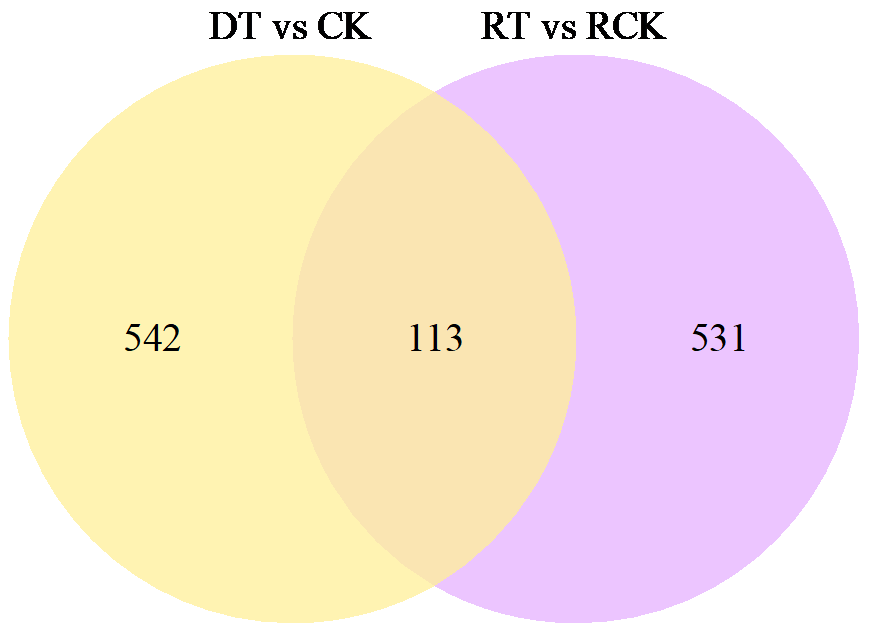
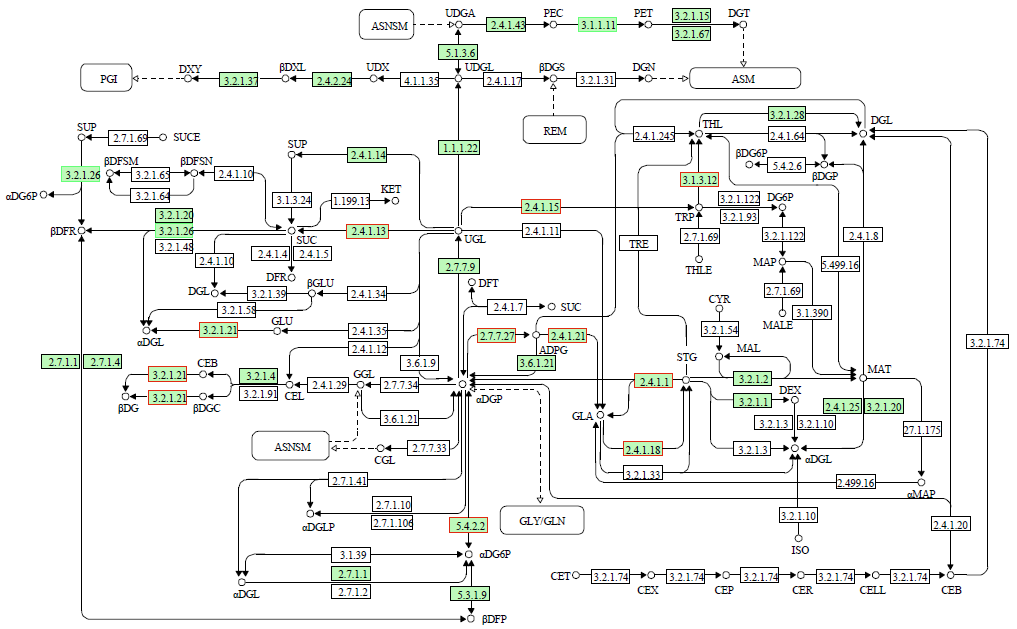
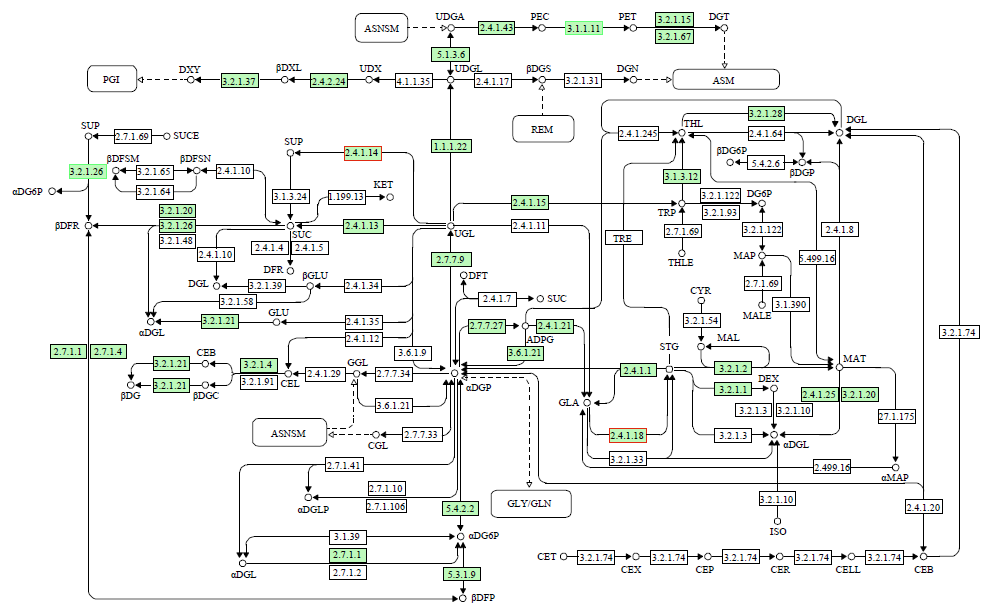
为了明确干旱胁迫对马铃薯的损伤以及复水对其保护效果,利用高通量测序技术进行了马铃薯代谢途径中基因表达研究。试验采用盆栽法,设干旱处理(40%土壤相对含水量,DT)和干旱对照(保持土壤相对含水量为70%并持续14d,CK)、复水处理(土壤相对含水量40%维持14d后恢复至土壤相对含水量的70%,RT)和复水对照(土壤相对含水量保持在70%并维持21d,RCK),研究干旱胁迫与复水处理对马铃薯体内淀粉及糖代谢途径的影响。DT与CK处理相比,有655个基因表达发生显著变化,其中与淀粉及糖代谢途径相关的有13个基因,上调表达的有9个,下调表达的有4个,涉及的酶有11个;RT与RCK处理相比,差异表达基因有644个,与淀粉及糖代谢途径相关的有5个,上调表达的有3个,下调表达的有2个,涉及的酶有4个。干旱胁迫后,淀粉及糖代谢途径中13个基因表达发生变化,涉及代谢途径中11个酶,功能与糖基水解酶、蔗糖合酶、糖基转移酶、淀粉合酶和果胶酯酶相关。复水后,有5个基因表达仍呈显著差异,影响4个酶,发生差异表达的基因数和涉及酶的数量分别减少了8和7个,下降幅度较为明显。虽然不是完全修复,但整体修复效果较好,补偿效应显著。
| [1] | 梁淑敏, 王颖, 潘哲超, 等. 不同栽培模式的土壤水热效应对马铃薯产量及结薯规律的影响. 作物杂志, 2018(3):90-96. |
| [2] | 魏延安. 世界马铃薯产业发展现状及特点. 世界农业, 2005(3):29-32. |
| [3] | 李勤志, 冯中朝. 中国马铃薯生产的经济分析. 广州: 暨南大学出版社, 2009. |
| [4] |
Anithakumari A M, Nataraja K, Visser R F C, et al. Genetic dissection of drought tolerance and recovery potential by quantitative trait locus mapping of a diploid potato population. Molecular Breeding, 2012, 30(3):1413-1429.
pmid: 23024597 |
| [5] |
Levy D. Tuber yield and tuber quality of several potato cultivars as affected by seasonal high temperatures and by water deficit in a semi-arid environment. Potato Research, 1986, 29(1):95-107.
doi: 10.1007/BF02361984 |
| [6] |
Dong S Y, Diane M, Beckles. Dynamic changes in the starch- sugar interconversion within plant source and sink tissues promote a better abiotic stress response. Journal of Plant Physiology, 2019, 234/235:80-93.
doi: 10.1016/j.jplph.2019.01.007 |
| [7] |
Ball S, Colleoni C, Cenci U, et al. The evolution of glycogen and starch metabolism in eukaryotes gives molecular clues to understand the establishment of plastid endosymbiosis. Journal of Experimental Botany, 2011, 62(6):1775-1801.
doi: 10.1093/jxb/erq411 pmid: 21220783 |
| [8] |
Cenci U, Nitschke F, Steup M, et al. Transition from glycogen to starch metabolism in Archaeplastida. Trends Plant Science, 2014, 19(1):18-28.
doi: 10.1016/j.tplants.2013.08.004 |
| [9] |
Kaura H, Kaura K, Gill G K. Modulation of sucrose and starch metabolism by salicylic acid induces thermotolerance in spring maize. Russian Journal of Plant Physiology, 2019, 66(5):771-777.
doi: 10.1134/S102144371905008X |
| [10] |
Hare P D, Cress W A, Van Staden J. Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environment, 1998, 21(6):535-553.
doi: 10.1046/j.1365-3040.1998.00309.x |
| [11] | Krasavina M S, Burmistrova N A, Raldugina G N. The role of carbohydrates in plant resistance to abiotic stresses. San Diego: Academic Press, 2014. |
| [12] | Thomashow M F. Plant cold acclimation:freezing tolerance genes and regulatory mechanisms. Annual Review of Plant Biology, 1999, 50:571-599. |
| [13] |
Wanner L A, Junttila O. Cold-induced freezing tolerance in Arabidopsis. Plant Physiology, 1999, 120(2):391-400.
doi: 10.1104/pp.120.2.391 pmid: 10364390 |
| [14] |
MacNeill G J, Mehrpouyan S, Minow M A A, et al. Starch as a source, starch as a sink:the bifunctional role of starch in carbon allocation. Journal of Experimental Botany, 2017, 68(16):4433-4453.
doi: 10.1093/jxb/erx291 pmid: 28981786 |
| [15] |
Baroja-Fernández E, José Muñoz F, Montero M, et al. Enhancing sucrose synthase activity in transgenic potato (Solanum tuberosum L.) tubers results in increased levels of starch, ADP glucose and UDP glucose and total yield. Plant Cell Physiology, 2009, 50(9):1651-1662.
doi: 10.1093/pcp/pcp108 |
| [16] |
Lutfiyya L L, Xu N F, Robert L D, et al. Phylogenetic and expression analysis of sucrose phosphate synthase isozymes in plans. Journal of Plant Physiology, 2007, 164(7):923-933.
pmid: 16876912 |
| [17] |
Li J, Barojafernandez, Bahaji A, et al. Enhancing sucrose synthase activity results in increased levels of starch and ADP- glucose in maize (Zea mays L.) seed endosperms. Plant and Cell Physiology, 2013, 54(2):282-294.
doi: 10.1093/pcp/pcs180 |
| [18] | 卢合全, 沈法富, 刘凌霄, 等. 植物蔗糖合成酶功能与分子生物学研究进展. 中国农学通报, 2005, 21(7):34-37. |
| [19] | 齐红岩, 李天来, 刘海涛, 等. 番茄不同部位中糖含量和相关酶活性的研究. 园艺学报, 2005, 32(2):239-243. |
| [20] | 秦翠鲜, 桂意云, 陈忠良, 等. 植物蔗糖合成酶基因研究进展. 分子植物育种, 2018, 16(12):3907-3914. |
| [21] |
Fu H, Park W D. Sink- and vascular-associated sucrose synthase functions are encoded by different gene classes in potato. Plant Cell, 1995, 7(9):1369-1385.
doi: 10.1105/tpc.7.9.1369 pmid: 8589622 |
| [22] |
Yang J C, Zhang J H, Wang Z Q, et al. Activities of key enzymes in sucrose-to-starch conversion in wheat grains subjected to water deficit during grain filling. Plant Physiology, 2004, 135(3):1621-1629.
pmid: 15235118 |
| [23] |
Zhao C, Hua L N, Liu X F, et al. Sucrose synthase FaSS 1 plays an important role in the regulation of strawberry fruit ripening. Plant Growth Regulation, 2017, 81(1):175-181.
doi: 10.1007/s10725-016-0189-4 |
| [24] |
Bai W Q, Xiao Y H, Zhao J, et al. Gibberellin overproduction promotes sucrose synthase expression and secondary cell wall deposition in cotton fibers. PLoS ONE, 2014, 9(5):e96537.
doi: 10.1371/journal.pone.0096537 |
| [25] |
Yu W P. Complete structures of three rice sucrose synthase isogenes and differential regulation of their expressions. Bioscience Biotechnology and Biochemistry, 1996, 60(2):233-239.
pmid: 9063969 |
| [26] | Gordon A J, Minchin F R, James C L, et al. Sucrose synthase in legume nodules is essential for nitrogen fixation. Plant Physiology, 1999, 129(3):867-878. |
| [27] | Minoru K, Michihiro A, Susumu G, et al. KEGG for linking genomes to life and the environment. Nucleic Acids Research, 2008, 36(1):480-484. |
| [28] | Gong L. Transcriptome profiling of the potato (Solanum tuberosum L.) plant under drought stress and water-stimulus conditions. PLoS ONE, 2015, 10(5):1-20. |
| [29] | 刘素军, 蒙美莲, 陈有君. 干旱胁迫及复水对马铃薯类黄酮合成途径中关键酶及基因表达的影响. 植物生理学报, 2018, 54(1):81-91. |
| [30] |
Pomper K W, Breen P J. Levels of apoplastic solutes in developing strawberry fruit. Journal of Experimental Botany, 1995, 46(288):743-752.
doi: 10.1093/jxb/46.7.743 |
| [31] |
Viol A R, Romberts A G, Haupt S, et al. Tuberization in potato involves a switch from apoplastic to symplastic phloem unloading. Plant Cell, 2001, 13(2):385-398.
pmid: 11226192 |
| [1] | 付梓平, 范昱, 赖弟利, 张凯旋, 朱剑锋, 李基光, 周美亮, 王俊珍. 脱支和反复湿热处理对苦荞抗性淀粉含量和理化特性的影响[J]. 作物杂志, 2023, (1): 52–57 |
| [2] | 吴子帅, 刘广林, 李虎, 罗群昌, 陈传华, 朱其南. 施氮量对优质常规籼稻稻米品质的影响[J]. 作物杂志, 2023, (1): 84–88 |
| [3] | 杨妍, 徐宁生, 潘哲超, 李燕山, 杨琼芬, 张磊. 多效唑和氮肥对马铃薯产量及经济效益的影响[J]. 作物杂志, 2022, (6): 139–144 |
| [4] | 徐燕, 牛俊峰, 陈利军, 王世强, 董忠民, 王喆之. 基于高通量测序技术研究栽培苍术根际土壤微生物变化[J]. 作物杂志, 2022, (5): 221–228 |
| [5] | 郑思怡, 杨晔, 宋远辉, 花芹, 林泉祥, 张海涛, 程治军. 水稻甜质胚乳突变体m5788的鉴定及基因定位[J]. 作物杂志, 2022, (4): 14–21 |
| [6] | 谢奎忠, 孙小花, 罗爱花, 柳永强, 唐德晶, 朱永永, 胡新元. 基施锌肥对长期连作马铃薯抗病性相关酶活性、土传病害和产量的影响[J]. 作物杂志, 2022, (4): 154–159 |
| [7] | 陈玉珍, 唐广彬, 马宪新, 田贵云, 于宏心, 骆璎珞, 樊明寿, 贾立国. 马铃薯块茎发育的四大调控途径[J]. 作物杂志, 2022, (4): 9–13 |
| [8] | 张小全, 贾振宇, 李举旭, 李洪臣, 王豹祥, 汪健, 石刚, 王川, 武云杰. 不同促根措施对皖南烤烟成熟期钾素代谢的影响[J]. 作物杂志, 2022, (3): 205–210 |
| [9] | 刘菊, 李广存, 段绍光, 胡军, 简银巧, 刘建刚, 金黎平, 徐建飞. 不同夜间温度处理对马铃薯试管薯及块茎形成相关基因表达的影响[J]. 作物杂志, 2022, (3): 92–98 |
| [10] | 赵锴, 晋秀娟, 孙丽丽, 闫荣岳, 卢娟, 郭峰, 史雨刚, 孙黛珍. 小麦脱植基相关基因在春性小麦叶片叶绿素降解过程中的作用分析[J]. 作物杂志, 2022, (2): 81–88 |
| [11] | 杨志楠, 黄金文, 韩凡香, 李亚伟, 马建涛, 柴守玺, 程宏波, 杨德龙, 常磊. 秸秆带状覆盖对西北雨养区马铃薯农田土壤温度及产量的影响[J]. 作物杂志, 2022, (1): 196–204 |
| [12] | 李润卿, 申勇, 朱宽宇, 王志琴, 杨建昌. 施氮量对超级稻南粳9108产量、淀粉RVA谱特征值和理化特性的影响[J]. 作物杂志, 2022, (1): 205–212 |
| [13] | 高佳, 王姣, 王松, 刘红健, 康佳, 沈弘, 王海莉, 任少勇. 生物炭基肥对马铃薯田土壤脲酶活性和产量的影响[J]. 作物杂志, 2021, (6): 134–138 |
| [14] | 罗磊, 李亚杰, 姚彦红, 李丰先, 范奕, 董爱云, 刘惠霞, 牛彩萍, 李德明. 不同小整薯规格和药剂拌种处理对旱作重茬马铃薯生长及产量的影响[J]. 作物杂志, 2021, (6): 211–216 |
| [15] | 李鑫, 金光辉, 王鹏程, 王紫雯. 马铃薯品种(系)淀粉与产量表现稳定性分析[J]. 作物杂志, 2021, (6): 51–57 |
|
||