作物杂志,2024, 第3期: 192–200 doi: 10.16035/j.issn.1001-7283.2024.03.026
灌浆期干旱胁迫对不同小麦品种的生理性状与根系生长的影响
薛鑫雨1( ), 詹文博1, 陈新宜1, 周瑞祥1, 王永霞2, 薛瑞丽1, 李华1, 汪月霞1(
), 詹文博1, 陈新宜1, 周瑞祥1, 王永霞2, 薛瑞丽1, 李华1, 汪月霞1( ), 李艳2(
), 李艳2( )
)
- 1河南农业大学生命科学学院,450002,河南郑州
2河南省农业科学院作物分子育种研究院,450002,河南郑州
Effects of Drought Stress during Grain Filling Period on Physiological and Root Characteristics of Different Wheat Cultivars
Xue Xinyu1( ), Zhan Wenbo1, Chen Xinyi1, Zhou Ruixiang1, Wang Yongxia2, Xue Ruili1, Li Hua1, Wang Yuexia1(
), Zhan Wenbo1, Chen Xinyi1, Zhou Ruixiang1, Wang Yongxia2, Xue Ruili1, Li Hua1, Wang Yuexia1( ), Li Yan2(
), Li Yan2( )
)
- 1College of Life Sciences, Henan Agricultural University, Zhengzhou 450002, Henan, China
2Institute of Crops Molecular Breeding, Henan Academy of Agricultural Sciences, Zhengzhou 450002, Henan, China
摘要:
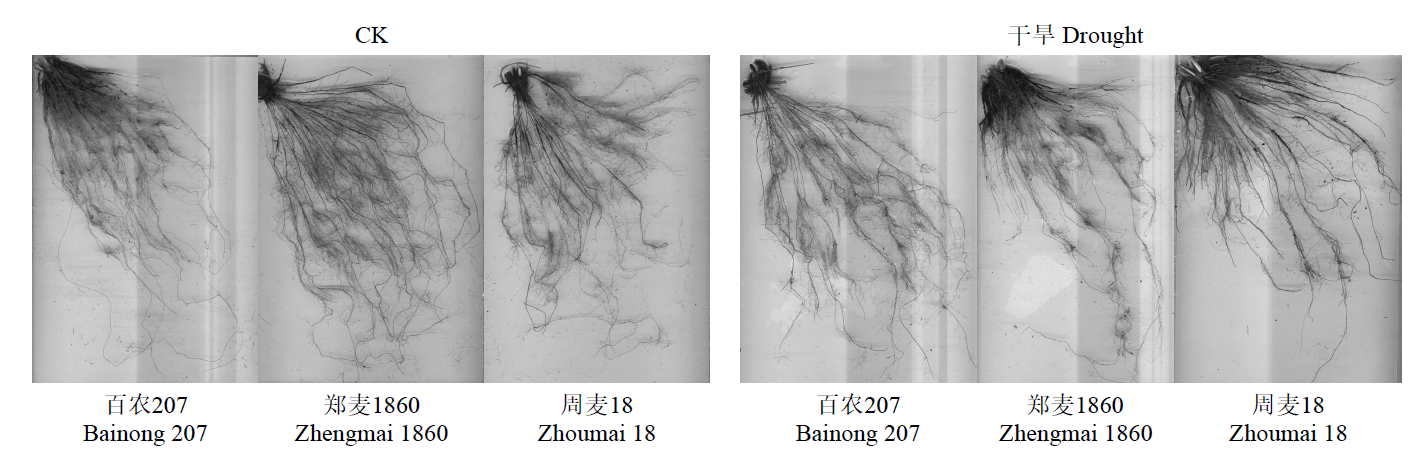
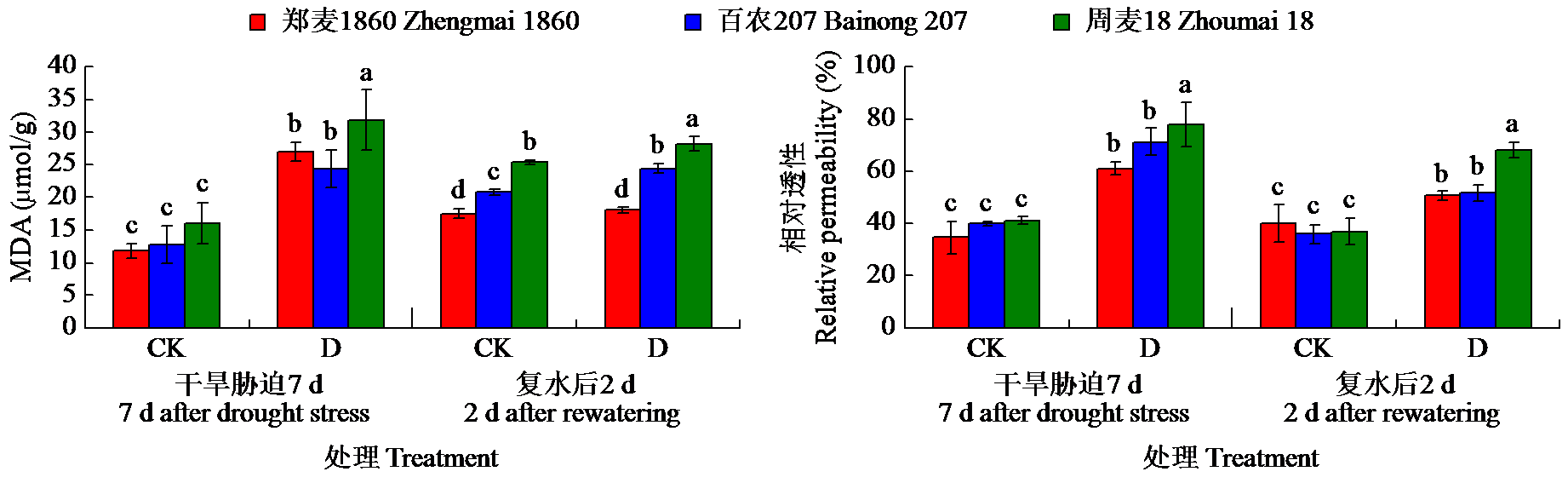
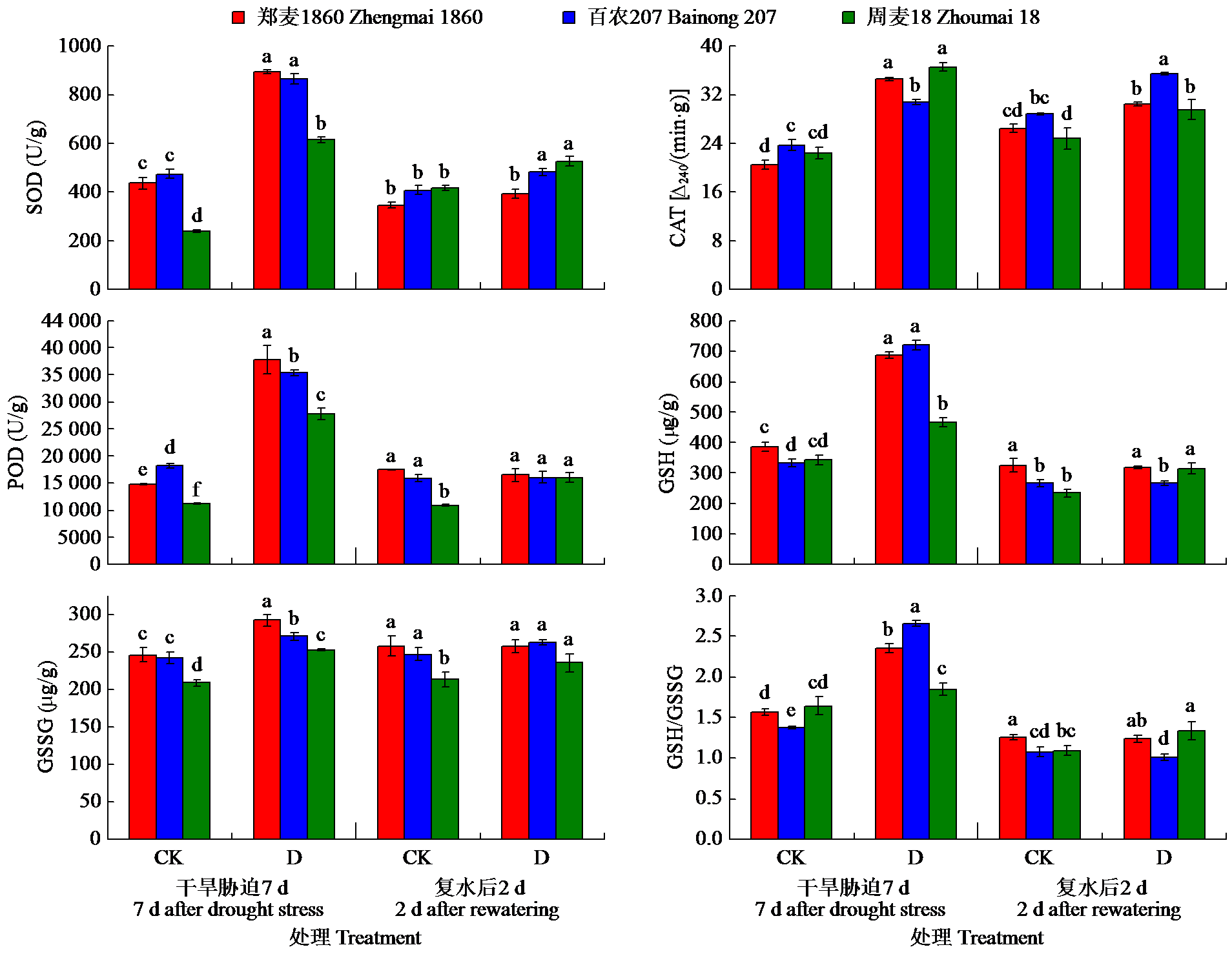
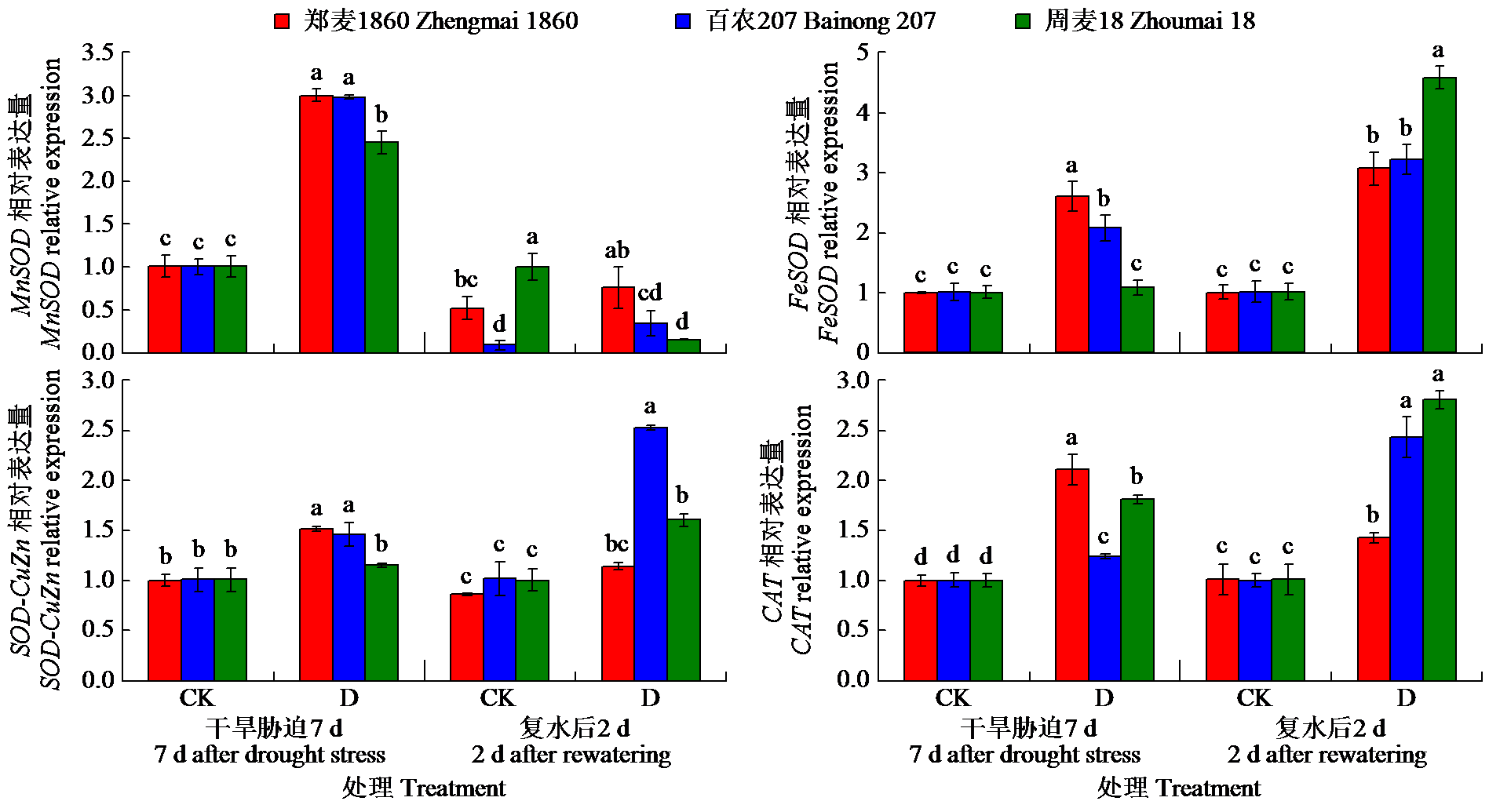
为明确灌浆期干旱胁迫对不同品种小麦生理特性和根系生长的影响,以高产高抗新品种郑麦1860和对照品种百农207、周麦18为材料,研究了灌浆期干旱处理对小麦根生长和生理性状的影响。结果表明,干旱胁迫下,郑麦1860的净光合速率(Pn)显著高于周麦18,郑麦1860与百农207的超氧化物歧化酶(SOD)、过氧化氢酶(CAT)和过氧化物酶(POD)活性提高,同时郑麦1860具有更高的根冠比、根尖数以及根表面积。与百农207和周麦18相比,郑麦1860的丙二醛含量在复水后与对照组差异最小。试验结果表明灌浆期干旱胁迫下郑麦1860能维持较高的Pn,稳定籽粒产量。通过提高SOD、CAT和POD活性从而加快谷胱甘肽再生,通过促进还原型谷胱甘肽/氧化型谷胱甘肽升高来减缓膜脂过氧化损伤。并且郑麦1860具有更强的吸水能力,在干旱胁迫下清除多余活性氧的能力更强,从而减少干旱胁迫对膜结构造成的损伤。
| [1] | 郝瑞煊, 孙敏, 任爱霞, 等. 宽幅条播冬小麦水分利用与干物质积累、品质的关系及播种密度的调控研究. 作物杂志, 2022(2):119-126. |
| [2] | Li C N, Li L, Reynolds M, et al. Recognizing the hidden half in wheat: root system attributes associated with drought tolerance. Journal of Experimental Botany, 2021, 72(14):5117-5133. |
| [3] |
Fahad S, Bajwa A A, Nazir U, et al. Crop production under drought and heat stress: plant responses and management options. Frontiers in Plant Science, 2017, 8:1147-1162.
doi: 10.3389/fpls.2017.01147 pmid: 28706531 |
| [4] | Luo Y L, Li W Q, Huang C, et al. Exogenous abscisic acid coordinating leaf senescence and transport of assimilates into wheat grains under drought stress by regulating hormones homeostasis. The Crop Journal, 2021, 9(4):901-914. |
| [5] | Su R N, Chen L, Wang Z H, et al. Differential response of cuticular wax and photosynthetic capacity by glaucous and non- glaucous wheat cultivars under mild and severe droughts. Plant Physiology and Biochemistry, 2020, 147(6):303-312. |
| [6] | Mathew I, Shimelis H, Shayanowako A I T, et al. Genome-wide association study of drought tolerance and biomass allocation in wheat. PLoS ONE, 2019, 14(12):e0225383 |
| [7] | Du H Y, Dong Q Y, Liu H P, et al. Polyamines conjugated to plasma membrane functioned in enhancing the tolerance of cucumber seedlings to osmotic stress via elevating H+-ATPase activity. Plant Physiology and Biochemistry, 2022, 170:64-74. |
| [8] |
Ebeed H T, Hassan N M, Aljarain A M. Exogenous applications of polyamines modulate drought responses in wheat through osmolytes accumulation, increasing free polyamine levels and regulation of polyamine biosynthetic genes. Plant Physiology and Biochemistry, 2017, 118:438-448.
doi: S0981-9428(17)30236-X pmid: 28743037 |
| [9] |
Chen D D, Chai S C, Mcintyre C L, et al. Overexpression of a predominantly root-expressed NAC transcription factor in wheat roots enhances root length, biomass and drought tolerance. Plant Cell Reports, 2018, 37(2):225-237.
doi: 10.1007/s00299-017-2224-y pmid: 29079898 |
| [10] | Hu C H, Zeng Q D, Tai L, et al. Interaction between TaNOX7 and TaCDPK 13 contributes to plant fertility and drought tolerance by regulating ROS production. Journal of Agricultural and Food Chemistry, 2020, 68(28):7333-7347. |
| [11] | Song R J, Zhang X C, Feng C J, et al. Exogenous hydrogen promotes germination and seedling establishment of barley under drought stress by mediating the ASA-GSH cycle and sugar metabolism. Journal of Plant Growth Regulation, 2023, 42:2749- 2762. |
| [12] | Abid M, Ali S, Qi L K, et al. Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Scientific Reports, 2018, 8(1):4615. |
| [13] |
Hassan N, Ebeed H, Aljaarany A. Exogenous application of spermine and putrescine mitigate adversities of drought stress in wheat by protecting membranes and chloroplast ultra-structure. Physiology and Molecular Biology of Plants, 2020, 26(2):233- 245.
doi: 10.1007/s12298-019-00744-7 pmid: 32158131 |
| [14] | Kirova E, Pecheva D, Simova-stoilova L. Drought response in winter wheat: protection from oxidative stress and mutagenesis effect. Acta Physiologiae Plantarum, 2021, 43(1):8. |
| [15] |
Lin Z, Wang Y L, Cheng L S, et al. Mutual regulation of ROS accumulation and cell autophagy in wheat roots under hypoxia stress. Plant Physiology and Biochemistry, 2021, 158:91-102.
doi: 10.1016/j.plaphy.2020.11.049 pmid: 33302125 |
| [16] |
Saed-moucheshi A, Sohrabi F, Fasihfar E, et al. Superoxide dismutase (SOD) as a selection criterion for triticale grain yield under drought stress: a comprehensive study on genomics and expression profiling, bioinformatics, heritability, and phenotypic variability. BMC Plant Biology, 2021, 21(1):148.
doi: 10.1186/s12870-021-02919-5 pmid: 33752615 |
| [17] | Aliyeva D R, Aydinli L M, Zulfugarov I S, et al. Diurnal changes of the ascorbate-glutathione cycle components in wheat genotypes exposed to drought. Functional Plant Biology, 2020, 47(11):998-1006. |
| [18] | Gasperl A, Balogh E, Boldizsár Á, et al. Comparison of light condition-dependent differences in the accumulation and subcellular localization of glutathione in arabidopsis and wheat. International Journal of Molecular Sciences, 2021, 22(2):607. |
| [19] | Hasanuzzaman M, Nahar K, Rahman A, et al. Exogenous glutathione attenuates lead-induced oxidative stress in wheat by improving antioxidant defense and physiological mechanisms. Journal of Plant Interactions, 2018, 13(1):203-212. |
| [20] | Ghahremani B, Hassannejad S, Alizadeh K, et al. Salicylic acid alleviates oxidative stress and lipid peroxidation caused by clopyralid herbicide in Indian mustard plants. Acta Physiologiae Plantarum, 2022, 44(4):49. |
| [21] | 刘建新, 刘瑞瑞, 贾海燕, 等. 外源H2S对盐碱胁迫下裸燕麦幼苗生长和生理特性的影响. 麦类作物学报, 2021, 41(2):245-253. |
| [22] |
Hu L, Xie Y, Fan S J, et al. Comparative analysis of root transcriptome profiles between drought-tolerant and susceptible wheat genotypes in response to water stress. Plant Science, 2018, 272:276-293.
doi: S0168-9452(17)30854-3 pmid: 29807601 |
| [23] | Djanaguiraman M, Prasad P V V, Kumari J, et al. Root length and root lipid composition contribute to drought tolerance of winter and spring wheat. Plant and Soil, 2019, 439(1/2):57-73. |
| [24] | Hassouni K E, Alahmad S, Belkadi B, et al. Root system architecture and its association with yield under different water regimes in durum wheat. Crop Science, 2018, 58(6):2331-2346. |
| [25] | Wei C, Jiao Q J, Agathokleous E, et al. Hormetic effects of zinc on growth and antioxidant defense system of wheat plants. Science of the Total Environment, 2022, 807:150992. |
| [26] | Zhang P Y, Yuan Z, Wei L, et al. Overexpression of ZmPP2C 55 positively enhances tolerance to drought stress in transgenic maize plants. Plant Science, 2022, 314:111127. |
| [27] |
Rehman H U, Alharby H F, Bamagoos A A, et al. Sequenced application of glutathione as an antioxidant with an organic biostimulant improves physiological and metabolic adaptation to salinity in wheat. Plant Physiology and Biochemistry, 2021, 158:43-52.
doi: 10.1016/j.plaphy.2020.11.041 pmid: 33296845 |
| [28] | Shan C J, Zhang S L, Ou X Q. The roles of H2S and H2O2 in regulating AsA-GSH cycle in the leaves of wheat seedlings under drought stress. Protoplasma, 2018, 255(4):1257-1262. |
| [29] | Madhu, Kaur A, Tyagi S, et al. Exploration of glutathione reductase for abiotic stress response in bread wheat (Triticum aestivum L.). Plant Cell Reports, 2022, 41(3):639-654. |
| [30] |
Kaya C, Ugurlar F, Farooq S, et al. Combined application of asparagine and thiourea improves tolerance to lead stress in wheat by modulating AsA-GSH cycle, lead detoxification and nitrogen metabolism. Plant Physiology and Biochemistry, 2022, 190:119-132.
doi: 10.1016/j.plaphy.2022.08.014 pmid: 36113307 |
| [31] | Zhao Q, Zhong M, He L, et al. Overexpression of a chrysanthemum transcription factor gene DgNAC1 improves drought tolerance in chrysanthemum. Plant Cell,Tissue and Organ Culture, 2018, 135(1):119-132. |
| [32] | Tounsi S, Jemli S, Feki K, et al. Superoxide dismutase (SOD) family in durum wheat: promising candidates for improving crop resilience. Protoplasma, 2023, 260(1):145-158. |
| [1] | 郭海斌, 张军刚, 王文文, 薛志伟, 许海涛, 冯晓曦, 王斌功, 王成业. 夏玉米光合特性、根系生长和产量对砂姜黑土深松增密的响应[J]. 作物杂志, 2024, (3): 109–118 |
| [2] | 张素瑜, 岳俊芹, 李向东, 靳海洋, 任德超, 杨明达, 邵运辉, 王汉芳, 方保停, 张德奇, 时艳华, 秦峰, 程红建. 施氮对郑麦366光合速率、花后干物质积累及产量的影响[J]. 作物杂志, 2024, (3): 127–132 |
| [3] | 吴永兵, 袁华恩, 张瑛, 陈泳纬, 阳苇丽, 何正川, 赵铭钦. 不同垄高下雪茄烟根组织结构及根系与地上部生长动态变化[J]. 作物杂志, 2024, (3): 148–155 |
| [4] | 李佳奇, 史建国, 陈启航, 常风云, 段义忠, 柴乖强, 贾磊, 陈涛. 陕北风沙草滩区覆膜对玉米根际土壤微生物群落的影响[J]. 作物杂志, 2024, (3): 238–246 |
| [5] | 卿晨, 刘正学, 李彦杰. 转录组测序分析干旱胁迫下复合微生物菌肥对玉米幼苗抗旱性的影响[J]. 作物杂志, 2024, (3): 32–39 |
| [6] | 孙辉, 赵昌平, 岳洁茹, 白秀成, 杨吉芳, 叶志杰, 张风廷. 不同生态环境和日温差对BS型小麦光温敏雄性不育系育性转换和农艺性状的影响[J]. 作物杂志, 2024, (3): 40–46 |
| [7] | 孙通, 杨玉双, 马瑞琦, 朱英杰, 常旭虹, 董志强, 赵广才. 聚糠萘合剂和乙矮合剂对小麦抗倒伏能力、产量与品质的影响[J]. 作物杂志, 2024, (2): 113–121 |
| [8] | 徐哲莉, 朱伟旗, 王立涛, 史峰, 韦志英, 王丽娜, 邱红伟, 张晓英, 李辉利. 灌水及叶面施氮对晚播小麦产量、品质及光合特性的影响[J]. 作物杂志, 2024, (2): 139–147 |
| [9] | 谢晋, 李谨成, 梁增发, 黄浩, 张玺, 高仁吉, 金保锋, 曾繁东, 卢志伟, 蔡一霞, 王维. 垄高与有机肥施用比例对烤烟根系生长及上部烟叶质量的影响[J]. 作物杂志, 2024, (2): 165–171 |
| [10] | 赵广才. 北方冬麦区小麦苗情分析及春季管理技术建议[J]. 作物杂志, 2024, (2): 255–260 |
| [11] | 张玉, 杨文静, 刘璇, 聂峰杰, 张丽, 石磊, 张国辉, 郭志乾, 巩檑. 马铃薯细胞壁蔗糖转化酶基因StCWIN1启动子克隆与表达及其在干旱胁迫下的作用分析[J]. 作物杂志, 2024, (2): 54–61 |
| [12] | 胡昊驰, 王富贵, 朱孔艳, 胡树平, 王猛, 王志刚, 孙继颖, 于晓芳, 包海柱, 高聚林. 秸秆还田年限及施磷水平对玉米根系生长和产量的影响[J]. 作物杂志, 2024, (2): 80–88 |
| [13] | 罗晓颖, 房彦飞, 胡冬平, 唐江华, 徐文修, 王怀港. 播种方式和播量对干旱区旱地小麦土壤水分利用及产量的影响[J]. 作物杂志, 2024, (2): 97–104 |
| [14] | 张玉林, 杜艺, 柴旭田, 李向义, 鲁艳, 张志浩, 曾凡江. 不同生育时期油莎豆饲草离子特性和根系养分对不同种植模式的响应[J]. 作物杂志, 2024, (1): 104–110 |
| [15] | 刘红杰, 任德超, 葛君, 张素瑜, 吕国华, 何勋. 积温和播种密度对小麦越冬前生长状况的影响[J]. 作物杂志, 2024, (1): 141–147 |
|
||