作物杂志,2018, 第6期: 76–82 doi: 10.16035/j.issn.1001-7283.2018.06.012
脱落酸对低温胁迫下玉米幼苗生长和光合特性的影响
田礼欣1,杨晔1,左师宇1,刘旋1,魏湜1,孙磊2,李晶1
- 1 东北农业大学农学院,150030,黑龙江哈尔滨
2 黑龙江省农业科学院土壤肥料与环境资源研究所,150086,黑龙江哈尔滨
Effects of Abscisic Acid on Growth and Photosynthetic Characteristics of Maize Seedlings under Low Temperature Stress
Tian Lixin1,Yang Ye1,Zuo Shiyu1,Liu Xuan1,Wei Shi1,Sun Lei2,Li Jing1
- 1 College of Agronomy, Northeast Agricultural University, Harbin 150030, Heilongjiang, China
2 Institute of Soil Fertilizer and Environment Resources, Heilongjiang Academy of Agricultural Sciences, Harbin 150086, Heilongjiang, China
摘要:
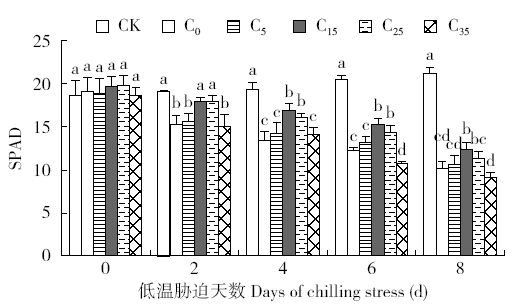
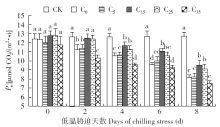
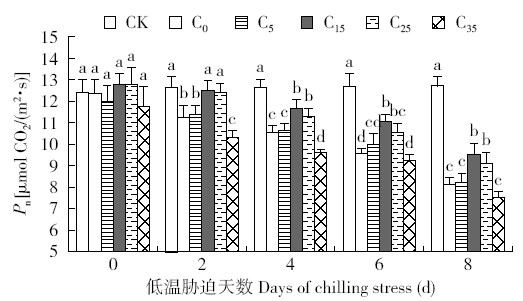
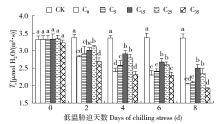
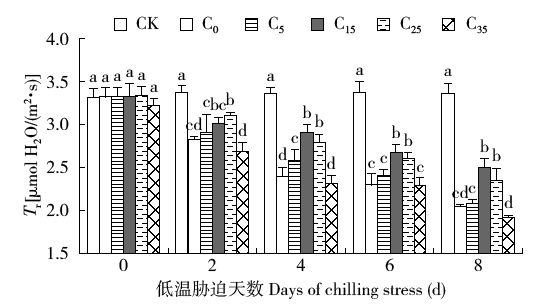
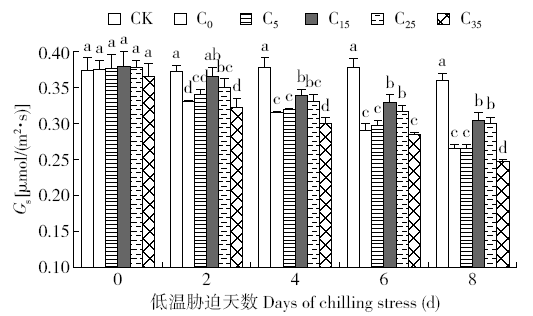
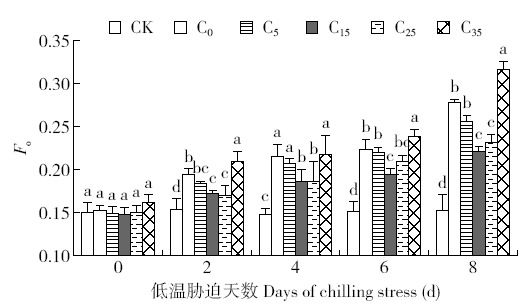
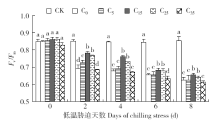
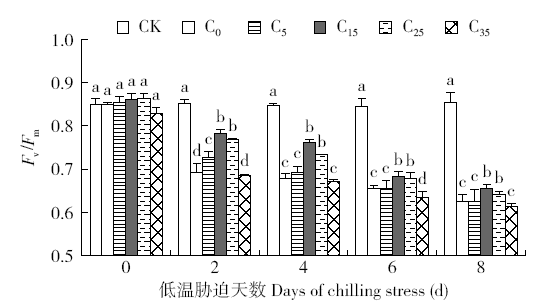
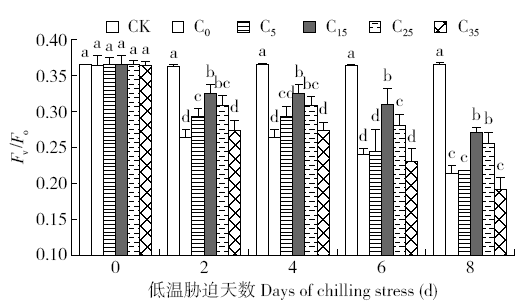
脱落酸(ABA)是作物生产中广泛应用的一种植物激素。探究ABA对低温胁迫下玉米幼苗光合特性的影响,为阐明低温胁迫下玉米幼苗叶片光合结构的防御保护机制提供理论依据。以玉米品种“久龙5号”为试验材料,采用盆栽试验,探讨了低温胁迫0、2、4、6、8d下不同浓度脱落酸对玉米幼苗生长及光合特性的影响。结果表明,低温胁迫显著抑制玉米幼苗的正常生长,导致SPAD值下降,叶片净光合速率(Pn)、蒸腾速率(Tr)、气孔导度(Gs)、PSⅡ光化学最大效率(Fv/Fm)和潜在光化学效率(Fv/Fo)水平下降,初始荧光(Fo)显著升高。外源喷施适当浓度的ABA能够显著缓解由低温胁迫导致的玉米叶片Pn、Tr和Gs下降幅度,提高Fv/Fm和Fv/Fo,降低Fo水平。由此可见,外源施加适当浓度的ABA能提高幼苗叶片的光合能力,提高PSⅡ反应中心活性,促进物质积累,提高玉米幼苗的耐冷性。同时,外源施加ABA存在一定的浓度效应,即“低促高抑”。本研究所设计浓度范围中15mg/L ABA浓度对光合抑制缓解效果最显著。
| [1] | 国家统计局. (2016-12-08)[2018-06-04]. . |
| [2] | 扈光辉 . 玉米抗冷研究进展. 黑龙江农业科学, 2009(2):150-152. |
| [3] |
Janowiak F, Luck E, Dörffling K . Chilling tolerance of maize seedlings in the field during cold periods in spring is related to chilling-induced increase in abscisic acid level. Journal of Agronomy & Crop Science, 2003,189(3):156-161.
doi: 10.1046/j.1439-037X.2003.00027.x |
| [4] |
Chen L J, Xiang H Z, Miao Y , et al. An overview of cold resistance in plants. Journal of Agronomy & Crop Science, 2014,200(4):237-245.
doi: 10.1111/jac.12082 |
| [5] |
Nguyen H T, Leipner J, Stamp P , et al. Low temperature stress in maize (Zea mays L.) induces genes involved in photosynthesis and signal transduction as studied by suppression subtractive hybridization. Plant Physiology & Biochemistry, 2009,47(2):116-122.
doi: 10.1016/j.plaphy.2008.10.010 pmid: 19042136 |
| [6] | Rivaroveda L, Périlleux C . Effects of cold temperatures on the early stages of maize (Zea mays L.). A review. Biotechnologie Agronomie Societe et Environnement, 2015,19(1):42-52. |
| [7] | 马延华, 王庆祥, 陈绍江 . 玉米耐寒性生理生化机理与分子遗传研究进展. 玉米科学, 2013,21(3):76-81,86. |
| [8] | Kosová K, Haise D, Tichá I . Photosynthetic performance of two maize genotypes as affected by chilling stress. Plant Soil Environment, 2005,51(5):206-212. |
| [9] |
Janowiak F, Dorffling K . Chilling of maize seedlings:changes in water status and abscisic acid content in ten genotypes differing in chilling tolerance. Journal of Plant Physiology, 1996,147(5):582-588.
doi: 10.1016/S0176-1617(96)80049-6 |
| [10] |
Bravo L A, Zúñiga G E, Alberdi M , et al. The role of ABA in freezing tolerance and cold acclimation in barley. Physiologia Plantarum, 1998,103(1):17-23.
doi: 10.1034/j.1399-3054.1998.1030103.x |
| [11] | Ahmed S, Higuchi H, Nawata E , et al. Effects of exogenous ABA and ethylene application and waterlogging on photosynthesis in Mungbean (Vigna radiate (L.) Wilczak). Japanese Journal of Tropical Agriculture, 2002,46(3):166-174. |
| [12] | Wang Y, Zhu M L, Yang Y Q , et al. Exogenous application of abscisic acid (ABA) enhances chilling tolerance in seedlings of Napier Grass (Pennisetum purpureum Schum.). Agricultural Science & Technology, 2017,18(3):417-423. |
| [13] |
Guo W L, Chen R G, Gong Z H , et al. Exogenous abscisic acid increases antioxidant enzymes and related gene expression in pepper (Capsicum annuum) leaves subjected to chilling stress. Genetics and Molecular Research, 2012,11(3):4063-4080.
doi: 10.4238/2012.September.10.5 pmid: 23079969 |
| [14] |
Wang G J, Miao W, Wang J Y , et al. Effects of exogenous abscisic acid on antioxidant system in weedy and cultivated rice with different chilling sensitivity under chilling stress. Journal of Agronomy & Crop Science, 2012,199(3):200-208.
doi: 10.1111/jac.12004 |
| [15] |
杨东清, 王振林, 倪英丽 , 等. 高温和外源ABA对不同持绿型小麦品种籽粒发育及内源激素含量的影响. 中国农业科学, 2014,47(11):2109-2125.
doi: 10.3864/j.issn.0578-1752.2014.11.005 |
| [16] | 任旭琴, 缪旻珉 . 低温下喷施ABA对辣椒光合特性和N、P、K的影响. 湖北农业科学, 2008,47(12):1461-1463. |
| [17] |
Karimi R, Ershadi A, Nejad A R , et al. Abscisic acid alleviates the deleterious effects of cold stress on ‘Sultana’ grapevine (Vitis vinifera L.) plants by improving the anti-oxidant activity and photosynthetic capacity of leaves. Journal of Horticultural Science & Biotechnology, 2016,91(2):1-10.
doi: 10.1080/14620316.2016.1162027 |
| [18] | 黄宇, 林智勇, 荣俊冬 , 等. 脱落酸对低温下雷公藤幼苗光合作用及叶绿素荧光的影响. 应用生态学报, 2011,22(12):3150-3156. |
| [19] |
李馨园, 杨晔, 张丽芳 , 等. 外源ABA对低温胁迫下玉米幼苗内源激素含量及Asr1基因表达的调节. 作物学报, 2017,43(1):141-148.
doi: 10.3724/SP.J.1006.2017.00141 |
| [20] |
杨猛, 魏玲, 庄文锋 , 等. 低温胁迫对玉米幼苗电导率和叶绿素荧光参数的影响. 玉米科学, 2012,20(1):90-94.
doi: 10.3969/j.issn.1005-0906.2012.01.017 |
| [21] |
Mishra A, Mishra K B, Höermiller I I , et al. Chlorophyll fluorescence emission as a reporter on cold tolerance in Arabidopsis thaliana accessions. Plant Signaling & Behavior, 2011,6(2):301-310.
doi: 10.4161/psb.6.2.15278 pmid: 3121992 |
| [22] |
Mohanty S, Grimm B, Tripathy B C . Light and dark modulation of chlorophyll biosynthetic genes in response to temperature. Planta, 2006,224(3):692-699.
doi: 10.1007/s00425-006-0248-6 |
| [23] | Yadegari L Z, Heidari R, Carapetian J . The influence of cold acclimation on proline,malondialdehyde (MDA),total protein and pigments contents in soybean (Glycine max) seedlings. Journal of Biological Sciences, 2008,3(1):74-79. |
| [24] |
Pagter M, Liu F, Jensen C R , et al. Effects of chilling temperatures and short photoperiod on PSⅡ function,sugar concentrations and xylem sap ABA concentrations in two hydrangea species. Plant Science, 2008,175(4):547-555.
doi: 10.1016/j.plantsci.2008.06.006 |
| [25] |
Kalisz A, Jezdinský A, Pokluda R , et al. Impacts of chilling on photosynthesis and chlorophyll pigment content in juvenile basil cultivars. Horticulture Environment & Biotechnology, 2016,57(4):330-339.
doi: 10.1007/s13580-016-0095-8 |
| [26] |
Zeng Y H, Zhang Y P, Xiang J , et al. Effects of chilling tolerance induced by spermidine pretreatment on antioxidative activity,endogenous hormones and ultrastructure of indica-japonica hybrid rice seedlings. Journal of Integrative Agriculture, 2015,15(2):295-308.
doi: 10.1016/S2095-3119(15)61051-6 |
| [27] | Mohabbati F, Paknejad F, Vazan S , et al. Protective effect of exogenous PGRs on chlorophyll fluorescence and membrane integrity of rice seedlings under chilling stress. Research Journal of Applied Sciences Engineering & Technology, 2013,5(1):146-153. |
| [28] |
Hendrickson L, Ball M C, Wood J T , et al. Low temperature effects on photosynthesis and growth of grapevine. Plant, Cell & Environment, 2004,27(7):795-809.
doi: 10.1111/j.1365-3040.2004.01184.x |
| [29] | Allen D J, Ort D R . Impacts of chilling temperatures on photosynthesis in warm-climate plants. Trends in Plant Science, 2001,6(1):36-42. |
| [30] |
Zhou B Y, Guo Z F, Lin L . Effects of abscisic acid application on photosynthesis and photochemistry of Stylosanthes guianensis under chilling stress. Plant Growth Regulation, 2006,48(3):195-199.
doi: 10.1007/s10725-006-0005-7 |
| [31] |
Bertamini M, Zulini L, Muthuchelian K , et al. Low night temperature effects on photosynthetic performance on two grapevine genotypes. Biologia Plantarum, 2007,51(2):381-385.
doi: 10.1007/s10535-007-0080-2 |
| [32] |
Souza T C D, Magalhães P C, Castro E M D , et al. The influence of ABA on water relation,photosynthesis parameters,and chlorophyll fluorescence under drought conditions in two maize hybrids with contrasting drought resistance. Acta Physiologiae Plantarum, 2013,35(2):515-527.
doi: 10.1007/s11738-012-1093-9 |
| [33] |
Baker N R . Chlorophyll fluorescence:a probe of photosynthesis in vivo. Annual Review of Plant Biology, 2008,59(1):89-113.
doi: 10.1146/annurev.arplant.59.032607.092759 |
| [1] | 张韶昀,李向岭,刘盼,张悦,翟大帅,尹宝重. 土壤耕作与施肥配合对玉米根系微观结构及产量的影响[J]. 作物杂志, 2018, (6): 144–148 |
| [2] | 明博,王克如,谢瑞芝,侯鹏,李少昆. 玉米子粒脱水研究与机械粒收对策[J]. 作物杂志, 2018, (6): 17–21 |
| [3] | 吴丽芳,魏晓梅,陆伟东,赵艳,吕婷. 秋水仙素诱导蚕豆及玉米根尖细胞染色体变异的研究[J]. 作物杂志, 2018, (6): 22–26 |
| [4] | 陈广周,王广福,渠建洲,司雷勇,金岩,徐淑兔,薛吉全,路海东. 不同玉米自交系子粒脱水速率及其与主要影响性状的相关分析[J]. 作物杂志, 2018, (5): 33–39 |
| [5] | 张一中,周福平,张晓娟,邵强,杨彬,柳青山. 高粱种质材料光合特性和水分利用效率鉴定及聚类分析[J]. 作物杂志, 2018, (5): 45–53 |
| [6] | 苏桂华,李春雷,苏义臣. 吉林省22份主推玉米品种区域试验评价[J]. 作物杂志, 2018, (5): 63–70 |
| [7] | 吴荣华,庄克章,刘鹏,张春艳. 鲁南地区夏玉米产量对气象因子的响应[J]. 作物杂志, 2018, (5): 104–109 |
| [8] | 李少昆,张万旭,王克如,韩冬生,杨小霞,陈永生,俞万兵,刘朝巍,张国强,柳枫贺,陈江鲁,杨京京,李璐璐,谢瑞芝,侯鹏,明博. 北疆玉米大田机械粒收质量调查[J]. 作物杂志, 2018, (5): 127–131 |
| [9] | 隋阳辉,高继平,刘彩虹,徐正进,王延波,赵海岩. 东北冷凉地区秸秆还田方式对水稻光合、干物质积累及氮素吸收的影响[J]. 作物杂志, 2018, (5): 137–143 |
| [10] | 高文俊,杨国义,高新中,玉柱,许庆方,原向阳,孙耀武. 氮磷钾肥对青贮玉米产量和品质的影响[J]. 作物杂志, 2018, (5): 144–149 |
| [11] | 张晓勇,杨友联,李树江,熊荣川,向红. 外源激素对低温胁迫下脱毒马铃薯扦插苗早衰的影响[J]. 作物杂志, 2018, (4): 95–101 |
| [12] | 樊艳丽,董会,卢柏山,史亚兴,高宁,史亚民,徐丽,席胜利,张翠芬,刘焱辉. 播期对不同糯玉米品种淀粉糊化特性的影响[J]. 作物杂志, 2018, (4): 79–83 |
| [13] | 李少昆,张万旭,王克如,俞万兵,陈永生,韩冬生,杨小霞,刘朝巍,张国强,王浥州,柳枫贺,陈江鲁,杨京京,谢瑞芝,侯鹏,明博. 北疆玉米密植高产宜粒收品种筛选[J]. 作物杂志, 2018, (4): 62–68 |
| [14] | 李红燕,王永宏,赵如浪,张文杰,明博,谢瑞芝,王克如,李璐璐,高尚,李少昆. 宁夏引/扬黄灌区玉米子粒脱水模型的构建与应用[J]. 作物杂志, 2018, (4): 149–153 |
| [15] | 朱鹏锦,庞新华,梁春,谭秦亮,严霖,周全光,欧克维. 低温胁迫对甘蔗幼苗活性氧代谢和抗氧化酶的影响[J]. 作物杂志, 2018, (4): 131–137 |
|
||