作物杂志,2021, 第5期: 28–34 doi: 10.16035/j.issn.1001-7283.2021.05.005
玉米HSP90基因家族的全基因组鉴定与分析
曹丽茹( ), 王国瑞(
), 王国瑞( ), 张新, 魏良明, 魏昕, 张前进, 邓亚洲, 王振华(
), 张新, 魏良明, 魏昕, 张前进, 邓亚洲, 王振华( ), 鲁晓民(
), 鲁晓民( )
)
- 河南省农业科学院粮食作物研究所,450002,河南郑州
Genome-Wide Identification and Analysis of HSP90 Gene Family in Maize
Cao Liru( ), Wang Guorui(
), Wang Guorui( ), Zhang Xin, Wei Liangming, Wei Xin, Zhang Qianjin, Deng Yazhou, Wang Zhenhua(
), Zhang Xin, Wei Liangming, Wei Xin, Zhang Qianjin, Deng Yazhou, Wang Zhenhua( ), Lu Xiaomin(
), Lu Xiaomin( )
)
- Grain Crop Research Institute, Henan Academy of Agricultural Sciences, Zhengzhou 450002, Henan, China
摘要:
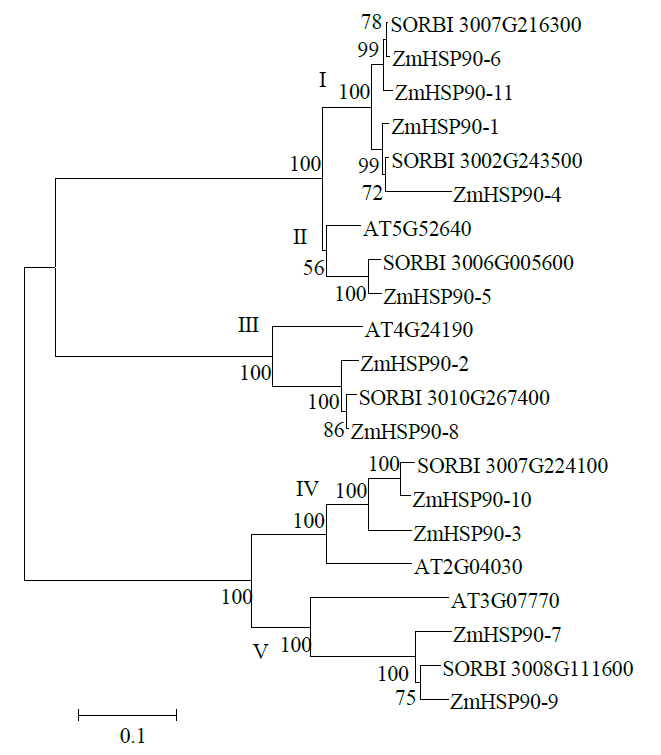
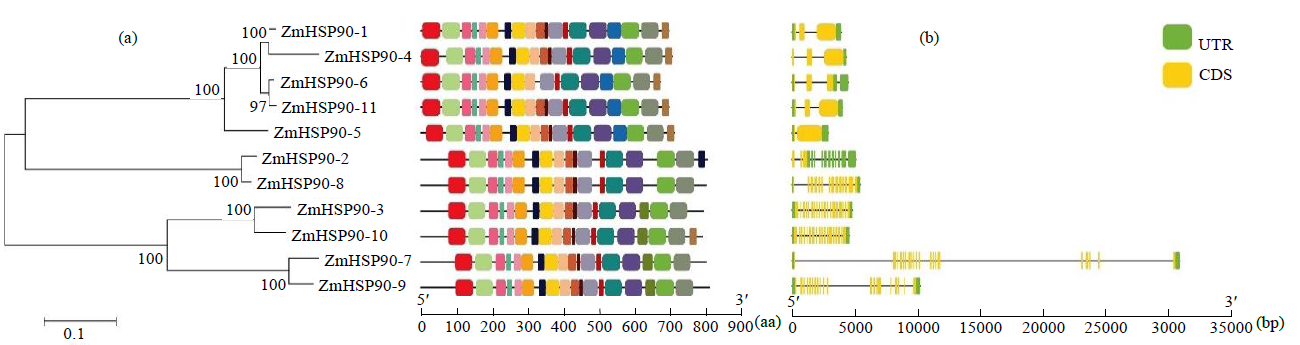
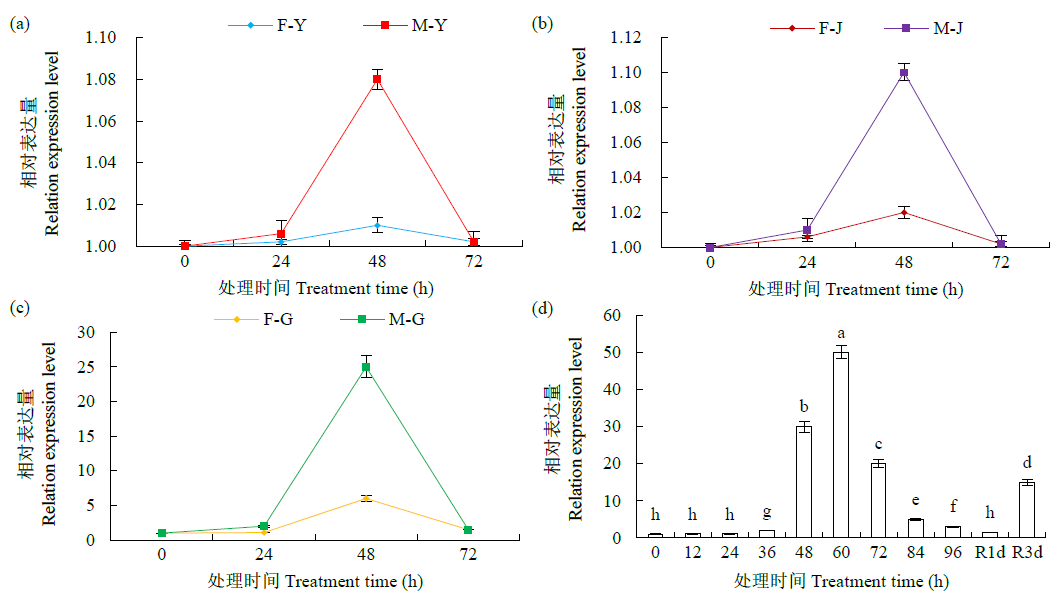
热激蛋白90(HSP90)广泛参与生物体的生长发育,且积极响应多种非生物胁迫。为全面解析玉米HSP90基因家族信息,对HSP90基因家族进化树、基因结构、保守基序(motif)、GO富集和组织表达模式等进行分析,并进一步分析ZmHSP90-1基因响应干旱胁迫的表达模式及蛋白互作关系。结果显示,从玉米全基因组水平共鉴定到11个ZmHSP90家族基因,被划分为5个亚族(I~V);相邻进化树分支上的ZmHSP90基因具有相似的motif和基因结构;GO富集分析显示,11个ZmHSP90基因均参与了蛋白质折叠过程;表达模式分析显示,11个ZmHSP90基因在不同组织或器官中均有表达,其中ZmHSP90-1基因的表达量相对较高;ZmHSP90-1基因在耐旱性强的玉米自交系中具有更高的表达量,且积极响应干旱和复水过程;蛋白互作预测显示,ZmHSP90-1可能与其互作蛋白协同响应非生物胁迫,发挥生物学功能。
| [1] | 于志晶, 尚丽霞, 蔡勤安, 等. 水稻热激蛋白基因HSP90转化大豆的研究. 大豆科学, 2016, 35(2):222-227. |
| [2] |
Wang W, Vinocur B, Shoseyov O, et al. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends in Plant Science, 2004, 9(5):244-252.
doi: 10.1016/j.tplants.2004.03.006 |
| [3] |
Sangster T A, Queitsch C. The HSP90 chaperone complex,an emerging force in plant development and phenotypic plasticity. Current Opinion in Plant Biology, 2005, 8(1):86-92.
pmid: 15653405 |
| [4] | 汤佳乐, 徐海, 苑平, 等. 植物Hsp90s与耐热性关系的研究进展. 生物技术通报, 2020, 339(10):178-184. |
| [5] |
Reddy R K, Chaudhary S, Patil P, et al. The 90 kDa heat shock protein (hsp90) is expressed throughout Brassica napus seed development and germination. Plant Science, 1998, 131(2):131-137.
doi: 10.1016/S0168-9452(97)00254-9 |
| [6] |
Wang G F, Wei X, Fan R, et al. Molecular analysis of common wheat genes encoding three types of cytosolic heat shock protein 90 (Hsp90):functional involvement of cytosolic Hsp90s in the control of wheat seedling growth and disease resistance. New Phytologist, 2011, 191(2):418-431.
doi: 10.1111/nph.2011.191.issue-2 |
| [7] |
Swindell W R, Huebner M, Weber A P. Transcriptional profiling of Arabidopsis heat shock proteins and transcription factors reveals extensive overlap between heat and non-heat stress response pathways. BMC Genomics, 2007, 8(1):125.
doi: 10.1186/1471-2164-8-125 |
| [8] |
Johnson J L, Brown C J C S, Chaperones. Plasticity of the Hsp90 chaperone machine in divergent eukaryotic organisms. Cell Stress and Chaperones, 2009, 14(1):83-94.
doi: 10.1007/s12192-008-0058-9 pmid: 18636345 |
| [9] |
Song H, Zhao R, Fan P, et al. Overexpression of AtHsp90.2,AtHsp90.5 and AtHsp90.7 in Arabidopsis thaliana enhances plant sensitivity to salt and drought stresses. Planta, 2009, 229(4):955-964.
doi: 10.1007/s00425-008-0886-y |
| [10] | Chen C, Xia R, Chen H, et al. TBtools,a toolkit for biologists integrating various HTS-data handling tools with a user-friendly interface. bioRxiv, 2018: 289660. |
| [11] | 卢云泽. 小麦灌浆期旗叶响应高温胁迫的蛋白组学与热响应关键基因HSP90的全基因组分析. 咸阳:西北农林科技大学, 2018. |
| [12] |
Chou K C, Shen H B. Cell-PLoc:a package of Web servers for predicting subcellular localization of proteins in various organisms. Nature Protocols, 2007, 3(2):153-162.
doi: 10.1038/nprot.2007.494 |
| [13] |
Meinke D W. Genome-wide identification of EMBRYO-DEFECTIVE (EMB) genes required for growth and development in Arabidopsis. New Phytologist, 2020, 226(2):306-325.
doi: 10.1111/nph.16071 pmid: 31334862 |
| [14] |
Oh S E, Yeung C, Babaei-Rad R, et al. Cosuppression of the chloroplast localized molecular chaperone HSP90.5 impairs plant development and chloroplast biogenesis in Arabidopsis. BMC Research Notes, 2014, 7(1):643.
doi: 10.1186/1756-0500-7-643 |
| [15] | Inoue H, Li M, Schnell D J, et al. An essential role for chloroplast heat shock protein 90 (Hsp90C) in protein import into chloroplasts. Proceedings of the National Academy of Sciences of the United States of America, 2013, 110(8):3173-3178. |
| [16] |
Song H, Fan P, Shi W, et al. Expression of five AtHsp90 genes in Saccharomyces cerevisiae reveals functional differences of AtHsp90s under abiotic stresses. Journal of Plant Physiology, 2010, 167(14):1172-1178.
doi: 10.1016/j.jplph.2010.03.016 |
| [17] |
Velinov V, Vaseva I, Zehirov G, et al. Overexpression of the NMig1 gene encoding a NudC domain protein enhances root growth and abiotic stress tolerance in Arabidopsis thaliana. Frontiers in Plant Science, 2020, 11:815.
doi: 10.3389/fpls.2020.00815 |
| [18] |
Buchberger A, Bukau B, Sommer T. Protein quality control in the cytosol and the endoplasmic reticulum:brothers in arms. Molecular Cell, 2010, 40(2):238-252.
doi: 10.1016/j.molcel.2010.10.001 pmid: 20965419 |
| [19] | 朱健康, 倪建平. 植物非生物胁迫信号转导及应答. 中国稻米, 2016, 22(6):52-60. |
| [20] |
Zhang J, Li J, Liu B, et al. Genome-wide analysis of the Populus Hsp90 gene family reveals differential expression patterns,localization,and heat stress responses. BMC Genomics, 2013, 14(1):532.
doi: 10.1186/1471-2164-14-532 |
| [21] | 刘玲玲. 玉米耐旱相关基因ZmHSP90-1和ZmMYB-R1的序列特征分析与功能验证. 哈尔滨:东北农业大学, 2012. |
| [22] | 肖化兴, 王立丰, 王萌. 橡胶树逆境胁迫响应基因HbHSP90.1的结构与功能分析. 分子植物育种, 2019, 17(16):5230-5237. |
| [23] | 伍玲. 番木瓜SGT1和RAR1的功能验证及WRKY转录因子的研究. 武汉:华中农业大学, 2013. |
| [24] | 王萍. 棉花中一个新的Parvulin类型肽酰脯氨酰顺反异构酶的功能分析. 泰安:山东农业大学, 2012. |
| [25] |
Richter K, Walter S, Buchner J. The Co-chaperone Sba1 connects the ATPase reaction of Hsp90 to the progression of the chaperone cycle. Journal of Molecular Biology, 2004, 342(5):1403-1413.
pmid: 15364569 |
| [26] |
Zhang Z, Zhang S, Zhang Y, et al. Arabidopsis floral initiator SKB1 confers high salt tolerance by regulating transcription and pre-mRNA splicing through altering histone H4R3 and small nuclear ribonucleoprotein LSM4 methylation. The Plant Cell, 2011, 23(1):396-411.
doi: 10.1105/tpc.110.081356 |
| [27] |
Zhang J, Clatterbuck R E, Rigamonti D, et al. Interaction between krit1 and icap1α infers perturbation of integrin β1-mediated angiogenesis in the pathogenesis of cerebral cavernous malformation. Human Molecular Genetics, 2001, 10(25):2953-2960.
pmid: 11741838 |
| [28] | 吴利民. 拟南芥和水稻锚蛋白(ANKP)基因家族初步研究. 长沙:湖南师范大学, 2006. |
| [1] | 刘威, 周剑雄, 谢媛圆, 张旭, 熊又升, 徐祥玉, 袁家富, 熊汉锋. 氮肥一次性基施对夏播鲜食甜玉米产量、品质和氮素利用效率的影响[J]. 作物杂志, 2021, (5): 134–139 |
| [2] | 张艳茹, 杨子和, 杨荣, 韩建, 焦金龙, 赵丽, 吴元奇. 对热带玉米种质群体控制双亲混合选择适应性的评价[J]. 作物杂志, 2021, (5): 14–19 |
| [3] | 裴志超, 周继华, 徐向东, 兰宏亮, 王俊英, 郎书文, 张伟强. 干旱处理对不同玉米品种叶片光合速率和抗氧化特性及产量的影响[J]. 作物杂志, 2021, (5): 95–100 |
| [4] | 王庆彬, 聂振田, 卢洁春, 彭春娥, 张民, 孟慧, 刘治国, 耿全政. 宛氏拟青霉提取物对夏玉米产量及氮素利用的影响[J]. 作物杂志, 2021, (4): 166–171 |
| [5] | 冯艳飞, 杨威, 任国鑫, 邓杰, 李文龙, 高树仁. 黑龙江省部分玉米杂交种的综合评价[J]. 作物杂志, 2021, (4): 46–50 |
| [6] | 陶志强, 闫鹏, 张学鹏. 不同年代玉米品种灌浆期的光合特征对高温适应性的初步研究[J]. 作物杂志, 2021, (4): 73–79 |
| [7] | 刘天昊, 张翼飞, 王怀鹏, 杨克军, 张津松, 孙逸珊, 肖珊珊, 徐荣琼, 杜嘉瑞, 李佳宇, 彭程, 王宝生. 叶面喷施硅肥对寒地玉米干物质积累分配及产量品质的调控效应[J]. 作物杂志, 2021, (4): 112–117 |
| [8] | 梁茜, 吴清山, 葛均筑, 吴锡冬, 杨永安, 侯海鹏, 张垚, 马志琪. 播期对华北平原雨养夏玉米产量形成与资源利用效率的影响[J]. 作物杂志, 2021, (4): 136–143 |
| [9] | 高鹏, 郭美俊, 杨雪芳, 董淑琦, 温银元, 郭平毅, 原向阳. 谷子和玉米叶片光合荧光参数对烟嘧磺隆胁迫的响应差异[J]. 作物杂志, 2021, (3): 70–77 |
| [10] | 刘剑钊, 袁静超, 梁尧, 贺宇, 张水梅, 史海鹏, 蔡红光, 任军. 玉米秸秆全量深翻还田地力提升技术模式实证及效益分析[J]. 作物杂志, 2021, (2): 135–139 |
| [11] | 李忠南, 王越人, 邬生辉, 刘励蔚, 曲海涛, 孙振宇, 李光发. 玉米单倍体自然加倍花粉结实力遗传的初步研究[J]. 作物杂志, 2021, (2): 57–61 |
| [12] | 张学鹏, 李腾, 王彪, 刘晴, 刘涵瑜, 陶志强, 隋鹏. 玉米叶片“源”的高温胁迫阈值研究[J]. 作物杂志, 2021, (2): 62–70 |
| [13] | 李瑞杰, 闫鹏, 王庆燕, 许艳丽, 卢霖, 董志强, 张凤路. 5-氨基乙酰丙酸和乙烯利对东北春玉米功能叶光合生理特性和产量的影响[J]. 作物杂志, 2021, (1): 135–142 |
| [14] | 刘佳敏, 汪洋, 褚旭, 齐欣, 王慢慢, 赵亚南, 叶优良, 黄玉芳. 种植密度和施氮量对小麦-玉米轮作体系下周年产量及氮肥利用率的影响[J]. 作物杂志, 2021, (1): 143–149 |
| [15] | 刘艳, 宫亮, 邢月华, 包红静. 基于正交设计的玉米有机无机培肥模式优化研究[J]. 作物杂志, 2021, (1): 168–174 |
|
||