作物杂志,2017, 第1期: 127–134 doi: 10.16035/j.issn.1001-7283.2017.01.023
不同水分条件对玉米根际微生物群落的影响
冯帅1,2,刘小利1,2,吴小丽1,2,江世杰2,周正富2,张维2,陈明2,王劲1,2,柯秀彬2
- 1西南科技大学生命科学与工程学院,621010,四川绵阳
2中国农业科学院生物技术研究所,100081,北京
Effects of Water Conditions on Microbial Community in Maize Rhizosphere
Feng Shuai1,2,Liu Xiaoli1,2,Wu Xiaoli1,2,Jiang Shijie2,Zhou Zhengfu2,Zhang Wei2,Chen Ming2,Wang Jin1,2,Ke Xiubin2
- 1Life Science and Engineering College,Southwest University of Science and Technology,Mianyang 621010,Sichuan
2Biotechnology Research Institute,Chinese Academy of Agriculture Science,Beijing 100081,China
摘要:
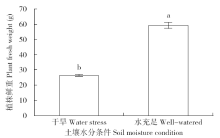
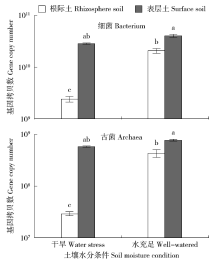
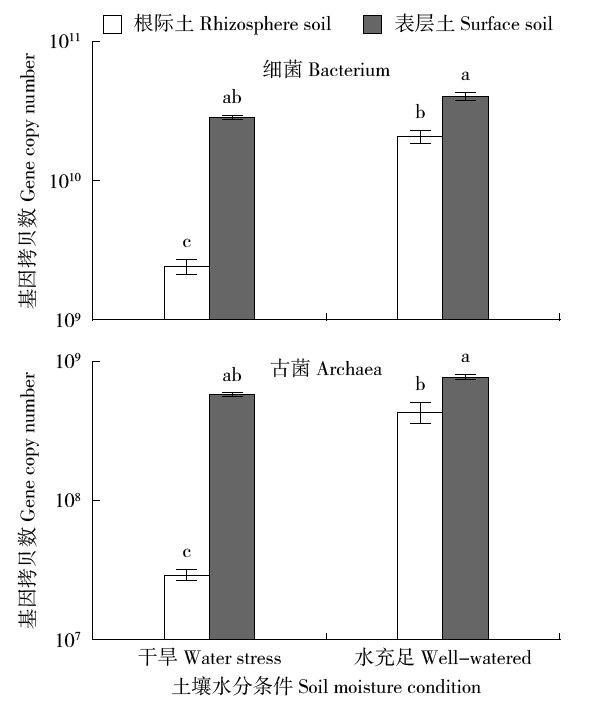
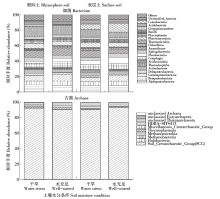
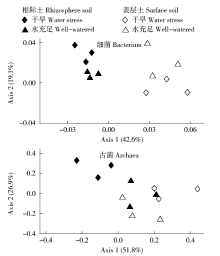
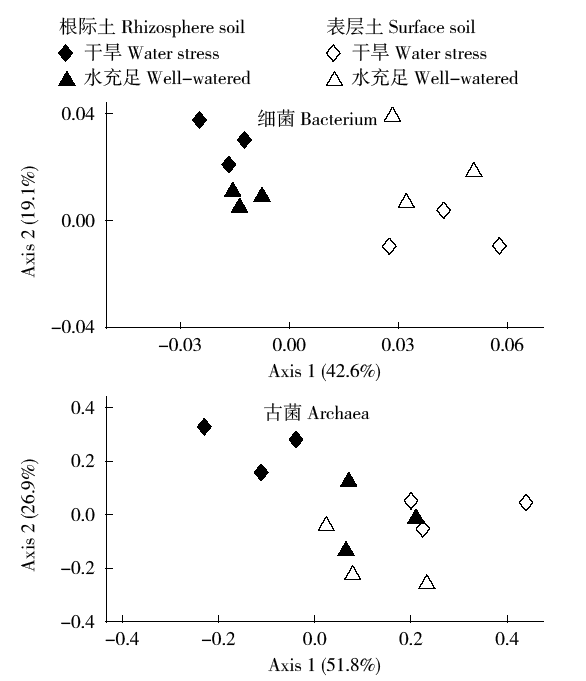
通过玉米盆栽试验研究不同水分条件下(土壤孔隙含水量20%和80%)土壤微生物群落结构及种群数量的变化。采集玉米生长第60天时的根际土和表层土,通过MiSeq Illumina高通量测序技术扩增细菌和古菌16S rRNA基因分析群落多样性,同时通过绝对定量PCR扩增细菌和古菌16S rRNA基因分析其种群数量。结果表明:高通量测序共得到41 546个细菌OTUs和6 921个古菌OTUs。土壤中细菌的多样性丰富,优势菌群为变形菌(Proteobacteria)和酸杆菌(Acidobacteria),而古菌的群落组成单一,优势菌群为奇古菌(Thaumarchaeota)。非度量多维尺度(NMDS)分析表明不同土壤区域中细菌群落结构具有明显差异,而古菌群落结构受水分和土壤区域的影响不明显。定量分析结果显示细菌和古菌数量受土壤区域和水分条件的影响显著。表层土中细菌和古菌数量均显著高于根际土。根际土中细菌和古菌数量在水分充足条件下均显著高于干旱条件,表层土中细菌和古菌数量也表现出相同趋势。本研究结果表明土壤不同区域(生态位)可能是影响土壤中微生物群落最重要的因素。
| [1] |
Bastida F, Moreno J L, Nicolás C , et al. Soil metaproteomics:a review of an emerging environmental science.Significance,methodology and perspectives. European Journal of Soil Science, 2009,60(6):845-859.
doi: 10.1111/j.1365-2389.2009.01184.x |
| [2] |
Chaudary D R, Gautam R K, Yousuf B , et al. Nutrients,microbial community structure and functional gene abundance of rhizosphere and bulk soils of halophytes. Applied Soil Ecology, 2015,91:16-26.
doi: 10.1016/j.apsoil.2015.02.003 |
| [3] |
Singh B K, Millard P, Whiteley A S , et al. Unravelling rhizosphere-microbial interactions:opportunities and limitations. Trends in Microbiology, 2004,12(8):386-393.
doi: 10.1016/j.tim.2004.06.008 |
| [4] | East R . Microbiome:Soil science comes to life. Nature, 2013,501(7468):18-19. |
| [5] |
Berendsen R L, Pieterse C M, Bakker P A . The rhizosphere microbiome and plant health. Trends in Plant Science, 2012,17(8):478-486.
doi: 10.1016/j.tplants.2012.04.001 |
| [6] | Bååth E, Berg B, Lohm U , et al. Effects of experimental acidification and liming on soil organisms and decomposition in a Scots pine forest. Pedobiologia, 1980,20:85-100. |
| [7] | Bais H P, Weir T L, Perryl L G , et al. The role of root exudates in rhizosphere interations with plants and other organisms.Annual Review of Plant Biology.Palo Alto; Annual Reviews. 2006,57:233-266. |
| [8] |
Bardgett R, Mawdsley J, Edwards S , et al. Plant species and nitrogen effects on soil biological properties of temperate upland grasslands. Functional Ecology, 1999,13(5):650-660.
doi: 10.1046/j.1365-2435.1999.00362.x |
| [9] |
Berg G, Smalla K . Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere. FEMS Microbiology Ecology, 2009,68(1):1-13.
doi: 10.1111/fem.2009.68.issue-1 |
| [10] |
Cavaglieri L, Orlando J, Etcheverry M . Rhizosphere microbial community structure at different maize plant growth stages and root locations. Microbiological Research, 2009,164(4):391-399.
doi: 10.1016/j.micres.2007.03.006 |
| [11] |
Castellanos T, Dohrmann A B, Imfeld G , et al. Search of environmental descriptors to explain the variability of the bacterial diversity from maize rhizospheres across a regional scale. European Journal of Soil Biology, 2009,45(5):383-393.
doi: 10.1016/j.ejsobi.2009.07.006 |
| [12] | Correa-galeote D, Bedmar E J, Fernández-González A J , et al. Bacterial communities in the rhizosphere of amilaceous maize (Zea mays L.) as assessed by pyrosequencing. Front Plant Science, 2016,7:1016. |
| [13] |
覃潇敏, 郑毅, 汤利 , 等. 玉米与马铃薯间作对根际微生物群落结构和多样性的影响. 作物学报, 2015,41(6):919-928.
doi: 10.3724/SP.J.1006.2015.00919 |
| [14] | 叶慧香, 崔跃原, 宋新元 , 等. 转cry1Ie基因抗虫玉米对土壤中细菌群落结构的影响. 生物安全学报, 2015,24(1):64-71. |
| [15] |
van Dijk E L, Auger H, Jaszczyszyn Y , et al. Ten years of next-generation sequencing technology. Trends in Genetics, 2014,30(9):418-426.
doi: 10.1016/j.tig.2014.07.001 |
| [16] |
Walker N J . Real-time and quantitative PCR:applications to mechanism-based toxicology. Journal of Biochemical and Molecular Toxicology, 2001,15(3):121-127.
doi: 10.1002/(ISSN)1099-0461 |
| [17] |
Collavino M M, Tripp H J, Frank I E , et al. NifH pyrosequencing reveals the potential for location-specific soil chemistry to influence N2-fixing community dynamics. Environmental Microbiology, 2014,16(10):3211-3223.
doi: 10.1111/emi.2014.16.issue-10 |
| [18] |
Chelius M, Triplett E . The Diversity of archaea and bacteria in association with the roots of Zea mays L. Microbial Ecology, 2001,41(3):252-263.
doi: 10.1007/s002480000087 |
| [19] |
Peiffer J A, Spor A, Koren O , et al. Diversity and heritability of the maize rhizosphere microbiome under field conditions. Proceedings of the Nationa Academy of Sciences of the United States of America, 2013,110(16):6548-6553.
doi: 10.1073/pnas.1302837110 |
| [20] |
Cao P, Zhang L M, Shen J P , et al. Distribution and diversity of archaea communities in selected Chinese soils. FEMS Microbiology Ecology, 2012,80(1):146-158.
doi: 10.1111/j.1574-6941.2011.01280.x |
| [21] | Atlas R M . Use of microbial diversity measurements to assess environmental stress.Current Perspectives in Microbial Ecology American Society for Microbiology,Washington,DC, 1984: 540-545. |
| [22] | Schimel J . Ecosystem consequences of microbial diversity and community structure.Arctic and Alpine Biodiversity:Patterns, Causes and Ecosystem Consequences, 1995: 239-254. |
| [23] |
Uhlířová E, Elhottova E, Tříska J , et al. Physiology and microbial community structure in soil at extreme water content. Folia Microbiologica, 2005,50(2):161-166.
doi: 10.1007/BF02931466 |
| [24] |
Valentine D L . Adaptations to energy stress dictate the ecology and evolution of the Archaea. Nature Reviews Microbiology, 2007,5(4):316-323.
doi: 10.1038/nrmicro1619 |
| [25] | Grinnell J . The niche-relationships of the California Thrasher. Auk, 1916,34(4):427-433. |
| [26] | 杨万勤, 宋光煜 . 土壤生态位及其应用. 世界科技研究与发展, 2001,23(2):21-23. |
| [27] |
Grayston S J, Wang S, Campbell C D , et al. Selective influence of plant species on microbial diversity in the rhizosphere. Soil Biology and Biochemistry, 1998,30(3):369-378.
doi: 10.1016/S0038-0717(97)00124-7 |
| [28] |
Li X, Rui J, Mao Y , et al. Dynamics of the bacterial community structure in the rhizosphere of a maize cultivar. Soil Biology and Biochemistry, 2014,68:392-401.
doi: 10.1016/j.soilbio.2013.10.017 |
| [29] |
Garbeva P, Vanveen J A, Vanelsas J D . Microbial diversity in soil:Selection of microbial populations by plant and soil type and implications for disease suppressiveness. Phytopathology, 2004,42:243-270.
doi: 10.1146/annurev.phyto.42.012604.135455 |
| [30] | Brimecombe M J, Deleij F A, Lynch J M . The effect of root exudates on rhizosphere microbial populations.The Rhizosphere,Biochemistry and Organic Substances at the Soil-plant Interface,Marcel Dekker,Inc, 2000: 95-140. |
| [31] |
Ke X, Angel R, Lu Y , et al. Niche differentiation of ammonia oxidizers and nitrite oxidizers in rice paddy soil. Environmental Microbiology, 2013,15(8):2275-2292.
doi: 10.1111/emi.2013.15.issue-8 |
| [32] |
Ke X, Lu Y, Conrad R . Different behaviour of methanogenic archaea and Thaumarchaeota in rice field microcosms. FEMS Microbiology Ecology, 2014,87(1):18-29.
doi: 10.1111/1574-6941.12188 |
| [33] |
Uroz S, Bu E M, Murat C , et al. Pyrosequencing reveals a contrasted bacterial diversity between oak rhizosphere and surrounding soil. Environmental Microbiology Reports, 2010,2(2):281-288.
doi: 10.1111/emi4.2010.2.issue-2 |
| [1] | 王春蕾,方志军,许艳蕊,卢晓平,穆春华,单凯,郝鲁江. 基于高通量测序技术分析使它隆对玉米根系内生菌多样性的影响[J]. 作物杂志, 2018, (1): 160–165 |
| [2] | 梁晋刚,张正光. 转基因作物种植对土壤生态系统影响的研究进展[J]. 作物杂志, 2017, (4): 1–6 |
| [3] | 蒋继志,王游游,王雪宁,李丽艳,万安琪,李明. 致病疫霉拮抗细菌SR13-2菌株的鉴定及对马铃薯离体组织的防病作用[J]. 作物杂志, 2017, (3): 146–150 |
| [4] | 张雪娇,石晶晶,常娜,张雅琴,齐永志,甄文超,尹宝重. 河北省冬小麦赤霉病拮抗细菌的分离与鉴定[J]. 作物杂志, 2017, (2): 157–162 |
| [5] | 刘振东,王波,薛蓓,王强锋,侯磊. 高原作物青稞根及根际内生细菌群落结构的高通量分析[J]. 作物杂志, 2016, (6): 49–52 |
| [6] | 许艳蕊,方志军,卢晓平,穆春华,范秋苹,郝鲁江. 四个玉米自交系根系内生细菌的分离和鉴定[J]. 作物杂志, 2016, (4): 112–117 |
| [7] | 姜良宇, 徐鹏飞, 吴俊江, 范素杰, 王欣, 李文滨, 张淑珍. 大豆细菌性斑点病侵染大豆品种叶片的超微结构观察[J]. 作物杂志, 2012, (3): 65–67 |
| [8] | 王秋菊, 焦峰, 崔战利, 李明贤. 光合细菌浸种对水稻种子萌发的影响[J]. 作物杂志, 2012, (3): 126–129 |
| [9] | 岑贞陆, 黄思良. 木薯抗细菌性枯萎病鉴定技术初报[J]. 作物杂志, 2008, (6): 33–35 |
| [10] | 邱清华, 邓绍云. 拮抗细菌处理种薯对马铃薯发芽、植株生长及产量的影响[J]. 作物杂志, 2008, (3): 43–45 |
| [11] | 杨水英, 李振轮, 青玲, 等. 细菌CHB101抑制油菜菌核病菌的机理研究[J]. 作物杂志, 2007, (2): 42–44 |
| [12] | 郑雅楠, 王晓鸣, 吕国忠. 玉米细菌性病害及其防治策略[J]. 作物杂志, 2006, (1): 62–65 |
| [13] | 孙桂华, 王海涌, 刘忠林. 2004年牡丹江市大豆生长中后期病虫害大发生原因分析与防治措施[J]. 作物杂志, 2005, (3): 28–29 |
| [14] | 王振民, 康波, 邓少华. 大豆新品种吉农6号[J]. 作物杂志, 1999, (3): 32–32 |
| [15] | 黄斌. 关于水稻施用生物钾肥问题的探讨[J]. 作物杂志, 1994, (4): 19–21 |
|