作物杂志,2021, 第6期: 58–61 doi: 10.16035/j.issn.1001-7283.2021.06.009
利用高密度遗传图谱发掘水稻抽穗期新位点
苏代群1( ), 陈亮1, 李锋1, 武琦1, 白君杰2, 邹德堂2, 王敬国2, 刘化龙2(
), 陈亮1, 李锋1, 武琦1, 白君杰2, 邹德堂2, 王敬国2, 刘化龙2( ), 郑洪亮2
), 郑洪亮2
- 1黑龙江省种业技术服务中心,150008,黑龙江哈尔滨
2东北农业大学寒地粮食作物种质创新与生理生态教育部重点实验室,150030,黑龙江哈尔滨
Identification of New Heading Date QTLs Using High Density Genetic Map in Rice
Su Daiqun1( ), Chen Liang1, Li Feng1, Wu Qi1, Bai Junjie2, Zou Detang2, Wang Jingguo2, Liu Hualong2(
), Chen Liang1, Li Feng1, Wu Qi1, Bai Junjie2, Zou Detang2, Wang Jingguo2, Liu Hualong2( ), Zheng Hongliang2
), Zheng Hongliang2
- 1Heilongjiang Province Seed Industry Technical Service Center, Harbin 150008, Heilongjiang, China
2Key Laboratory of Germplasm Enhancement, Physiology and Ecology of Food Crops in Cold Region,Ministry of Education, Northeast Agricultural University, Harbin 150030, Heilongjiang, China
摘要:
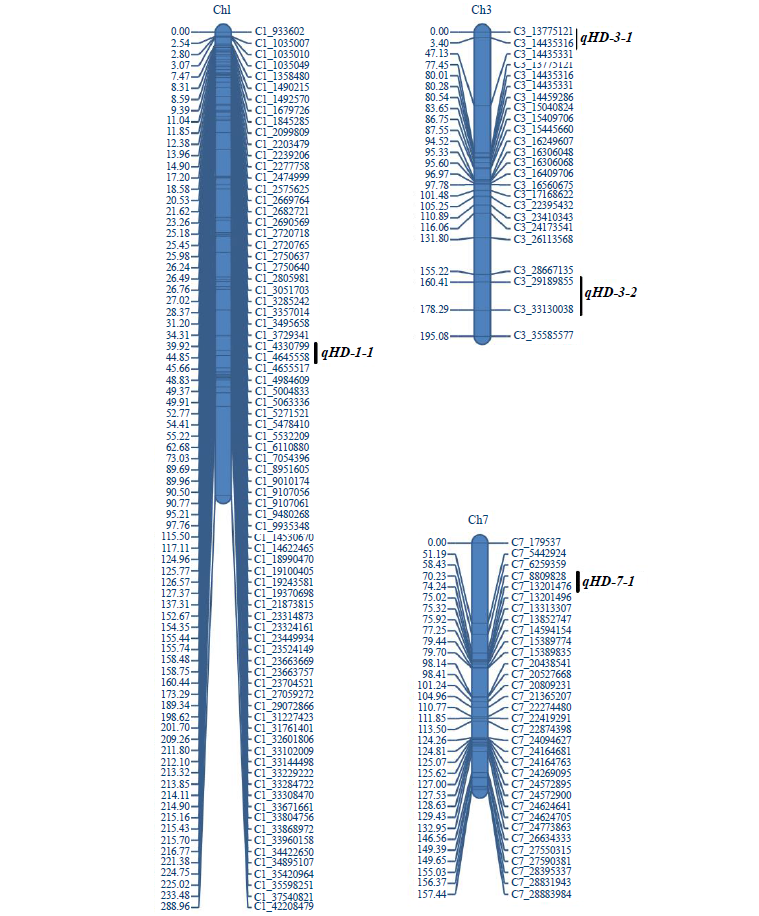
水稻抽穗期是决定水稻种植地区及其季节适应性的关键因素,发掘控制水稻抽穗期相关的新主效QTL至关重要。利用包含527个bin标记的高密度遗传连锁图谱,通过靶向测序基因型检测技术对水稻“空育131/小白粳子”衍生的RIL群体进行抽穗期基因型分析。通过对双亲和RIL群体的基本统计分析发现,双亲抽穗期呈极显著差异,表型处于RIL群体范围内,RIL群体有明显的超亲分离现象,符合正态分布。利用IciMapping 4.2软件的完备区间作图法,在水稻第1、3和7号染色体上共检测到4个QTL,其中3个QTL区间内分别含有与抽穗期相关的已知基因OsGI、Hd6和Ghd7,而qHD-3-1是控制水稻抽穗期的新位点。
| [1] | 王远征, 王晓菁, 李源, 等. 北方粳稻产量与品质性状及其相互关系分析. 作物学报, 2015, 41(6):910-918. |
| [2] |
Normile D. Reinventing rice to feed the world. Science, 2008, 321(5887):330-333.
doi: 10.1126/science.321.5887.330 pmid: 18635770 |
| [3] |
Fujino K, Sekiguchi H. Mapping of quantitative trait loci controlling heading date among rice cultivars in the northern most region of Japan. Breeding Science, 2008, 58(4):367-373.
doi: 10.1270/jsbbs.58.367 |
| [4] |
Cheng L R, Wang J, Ye G, et al. Identification of stably expressed QTL for heading date using reciprocal introgression line and recombinant inbred line populations in rice. Genetics Research, 2012, 94(5):245-253.
doi: 10.1017/S0016672312000444 |
| [5] | 郭梁, 张振华, 庄杰云. 水稻抽穗期QTL及其与产量性状遗传控制的关系. 中国水稻科学, 2012, 26(2):235-245. |
| [6] |
Kojima S, Takahashi Y, Kobayashi Y, et al. Hd3a,a rice ortholog of the Arabidopsis FT gene,promotes transition to flowering downstream of Hd1 under short-day conditions. Plant and Cell Physiology, 2002, 43(10):1096-1105.
pmid: 12407188 |
| [7] |
Zong W, Ren D, Huang M, et al. Strong photoperiod sensitivity is controlled by cooperation and competition among Hd1,Ghd7 and DTH8 in rice heading. New Phytologist, 2021, 229(3):1635-1649.
doi: 10.1111/nph.v229.3 |
| [8] |
Li J, Chu H, Zhang Y, et al. The rice HGW gene encodes a ubiquitin-associated (UBA) domain protein that regulates heading date and grain weight. PLoS ONE, 2012, 7(3):e34231.
doi: 10.1371/journal.pone.0034231 |
| [9] |
Liu X, Zhang H, Li H, et al. Fine-mapping quantitative trait loci for body weight and abdominal fat traits:effects of marker density and sample size. Poultry Science, 2008, 87(7):1314-1319.
doi: 10.3382/ps.2007-00512 pmid: 18577610 |
| [10] |
Thomson M J. High-throughput SNP genotyping to accelerate crop improvement. Plant Breeding and Biotechnology, 2014, 2(3):195-212.
doi: 10.9787/PBB.2014.2.3.195 |
| [11] | 李冬秀, 杨靖, 孙凯, 等. 基于高密度遗传图谱定位新的水稻抽穗期QTLs. 西北农林科技大学学报, 2020, 48(8):44-49. |
| [12] |
Churchill G A, Doerge R W. Empirical threshold values for quantitative trait mapping. Genetics, 1994, 138(3):963-971.
pmid: 7851788 |
| [13] |
McCouch S R. Gene nomenclature system for rice. Rice, 2008, 1(1):72-84.
doi: 10.1007/s12284-008-9004-9 |
| [14] | 黄成, 姜树坤, 刘梦红, 等. 水稻抽穗期的QTL剖析. 华北农学报, 2009, 24(3):7-9. |
| [15] |
Yu H, Xie W, Li J, et al. A whole‐genome SNP array (RICE 6 K) for genomic breeding in rice. Plant Biotechnology Journal, 2014, 12(1):28-37.
doi: 10.1111/pbi.2013.12.issue-1 |
| [16] |
Izawa T, Mihara M, Suzuki Y, et al. Os-GIGANTEA confers robust diurnal rhythms on the global transcriptome of rice in the field. The Plant Cell, 2011, 23(5):1741-1755.
doi: 10.1105/tpc.111.083238 |
| [17] | Takahashi Y, Shomura A, Sasaki T, et al. Hd6,a rice quantitative trait locus involved in photoperiod sensitivity,encodes the α subunit of protein kinase CK2. Proceedings of the National Academy of Sciences of the United States of America, 2001, 98(14):7922-7927. |
| [18] |
Xue W, Xing Y, Weng X, et al. Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nature Genetics, 2008, 40(6):761-767.
doi: 10.1038/ng.143 |
| [1] | 李旭, 付立东, 王宇, 隋鑫, 任海, 吕小红, 马畅, 杜萌, 毛艇. DEP1与NRT1.1B基因的遗传互作对水稻氮素利用的影响[J]. 作物杂志, 2021, (6): 22–27 |
| [2] | 张俊, 邓艾兴, 尚子吟, 唐志伟, 严圣吉, 张卫建. 秸秆还田下水稻丰产与甲烷减排的技术模式[J]. 作物杂志, 2021, (6): 230–235 |
| [3] | 余镁霞, 邓浩东, 谭景艾, 宋贵廷, 吴光亮, 陈利平, 刘睿琦, 邹安东, 贺浩华, 边建民. 利用染色体片段置换系定位低温影响水稻萌芽期根长和芽长QTL[J]. 作物杂志, 2021, (6): 36–45 |
| [4] | 石楠, 高志强, 胡海燕, 陈崇怡, 文双雅. 杂交稻有序机抛增密减肥处理对产量及肥料偏生产力的影响[J]. 作物杂志, 2021, (5): 128–133 |
| [5] | 唐志强, 张丽颖, 何娜, 马作斌, 赵明珠, 王昌华, 郑文静, 银永安, 王辉. 机械旱直播对水稻生育进程、光合特性及产量的影响[J]. 作物杂志, 2021, (5): 87–94 |
| [6] | 吴可, 谢慧敏, 刘文奇, 莫并茂, 韦国良, 陆献, 李壮林, 邓森霞, 韦善清, 梁和, 江立庚. 氮、磷、钾肥对南方双季稻区水稻产量及产量构成因子的影响[J]. 作物杂志, 2021, (4): 178–183 |
| [7] | 凌晨, 刘洪, 杨哲, 黄展权, 陈孟强, 饶得花, 徐振江. 双季稻栽培对水稻DUS测试标准品种数量性状表达的影响[J]. 作物杂志, 2021, (4): 18–25 |
| [8] | 张全芳, 姜明松, 陈峰, 朱文银, 周学标, 杨连群, 徐建第. 山东省水稻品种(系)的遗传多样性分析[J]. 作物杂志, 2021, (4): 26–31 |
| [9] | 王国骄, 宋鹏, 杨振中, 张文忠. 秸秆还田对水稻光合物质生产特征、稻米品质和土壤养分的影响[J]. 作物杂志, 2021, (4): 67–72 |
| [10] | 薛菁芳, 蔡永盛, 陈书强. 节水灌溉栽培模式对稻米品质和淀粉RVA谱的影响[J]. 作物杂志, 2021, (4): 86–92 |
| [11] | 杨磊, 金延迪, 刘侯俊. 铁、镉及其互作对水稻光合原初反应的影响[J]. 作物杂志, 2021, (4): 144–151 |
| [12] | 孟祥宇, 冉成, 刘宝龙, 赵哲萱, 白晶晶, 耿艳秋. 秸秆还田配施氮肥对东北黑土稻区土壤养分及水稻产量的影响[J]. 作物杂志, 2021, (3): 167–172 |
| [13] | 邢愿, 贺中华. 基于水分亏缺指数的贵州省水稻干旱特征分析[J]. 作物杂志, 2021, (2): 191–199 |
| [14] | 付景, 尹海庆, 王亚, 杨文博, 张珍, 白涛, 王越涛, 王付华, 王生轩. 不同追氮模式对河南沿黄稻区粳稻根系生长和产量的影响[J]. 作物杂志, 2021, (2): 77–86 |
| [15] | 周江, 谢宜章, 向平安. 湖南主要大田作物系统投入产出的能值分析[J]. 作物杂志, 2021, (1): 175–181 |
|
||