作物杂志,2020, 第2期: 20–27 doi: 10.16035/j.issn.1001-7283.2020.02.004
高粱硬脂酰-ACP脱氢酶基因(SbSAD)家族鉴定及不同发育阶段表达分析
赵训超,徐晶宇( ),盖胜男,魏玉磊,许晓萱,丁冬,刘梦,张今杰,邵文静
),盖胜男,魏玉磊,许晓萱,丁冬,刘梦,张今杰,邵文静
- 黑龙江八一农垦大学农学院/黑龙江省现代农业栽培技术与作物种质改良重点实验室,163319,黑龙江大庆
Identification of Stearyl -ACP Desaturase Gene (SbSAD) Family and Their Expression Analysis at Different Developmental Stages in Sorghum
Zhao Xunchao,Xu Jingyu( ),Gai Shengnan,Wei Yulei,Xu Xiaoxuan,Ding Dong,Liu Meng,Zhang Jinjie,Shao Wenjing
),Gai Shengnan,Wei Yulei,Xu Xiaoxuan,Ding Dong,Liu Meng,Zhang Jinjie,Shao Wenjing
- College of Agronomy, Heilongjiang Bayi Agricultural University/Key Laboratory of Modern Agricultural Cultivation and Crop Germplasm Improvement of Heilongjiang Province, Daqing 163319, Heilongjiang, China
摘要:
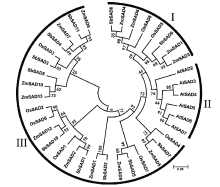
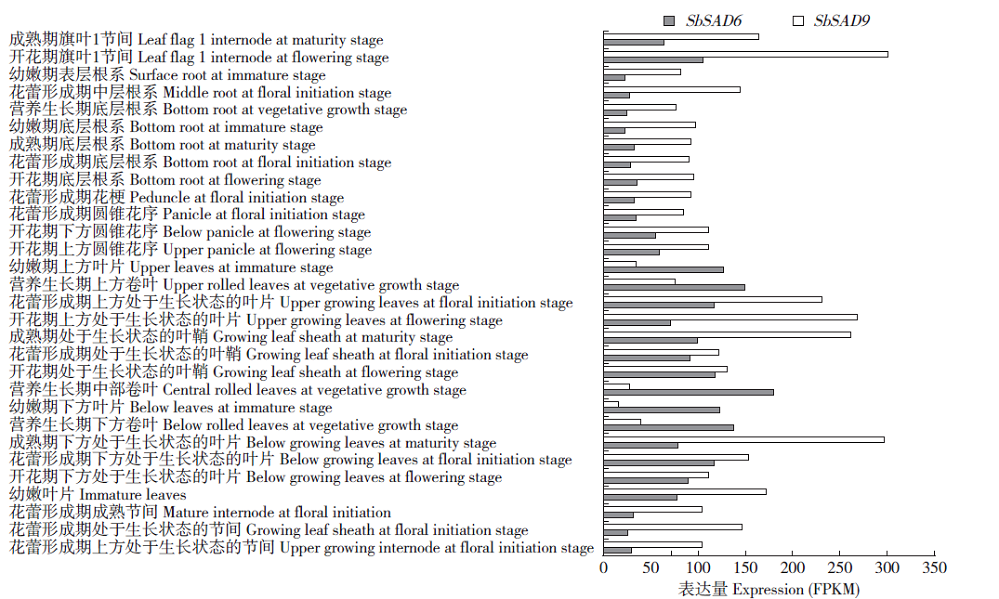
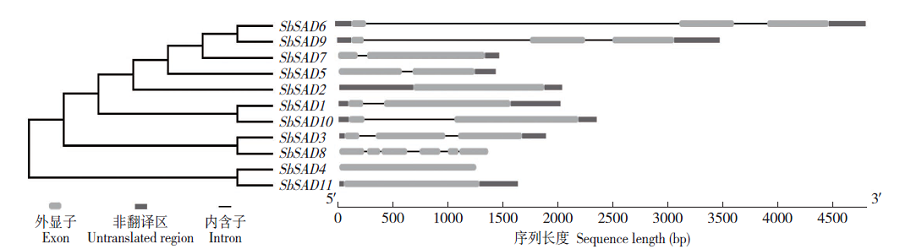
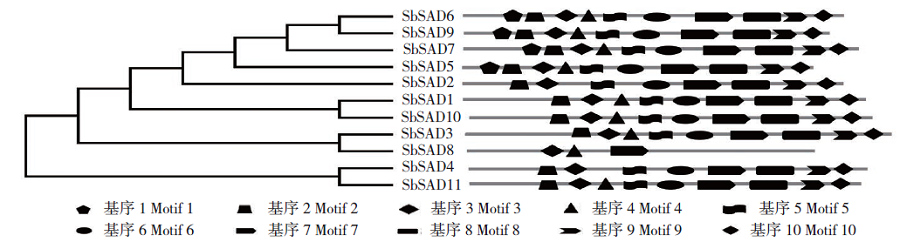
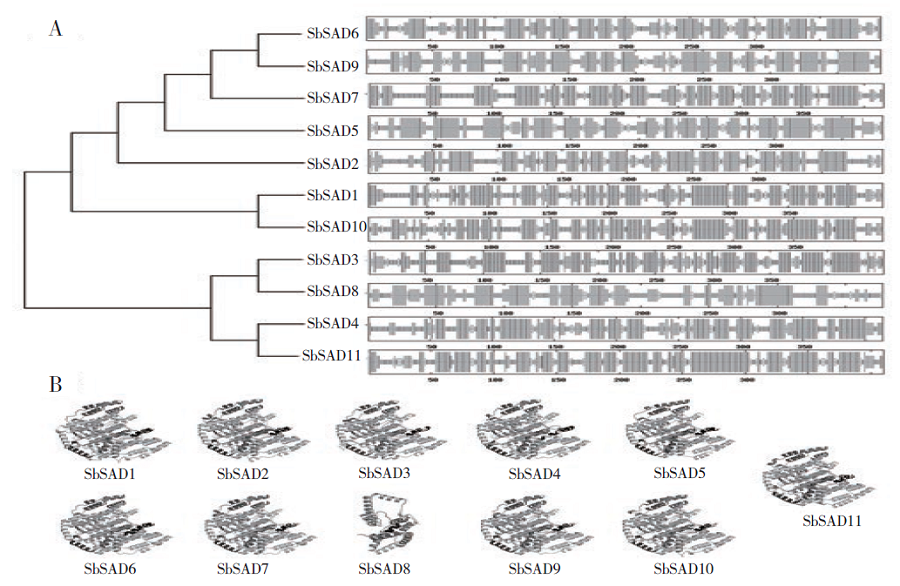
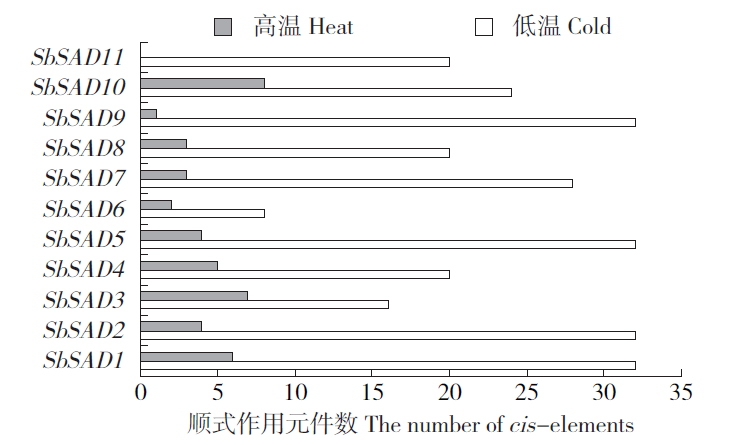
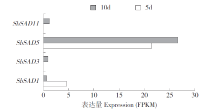
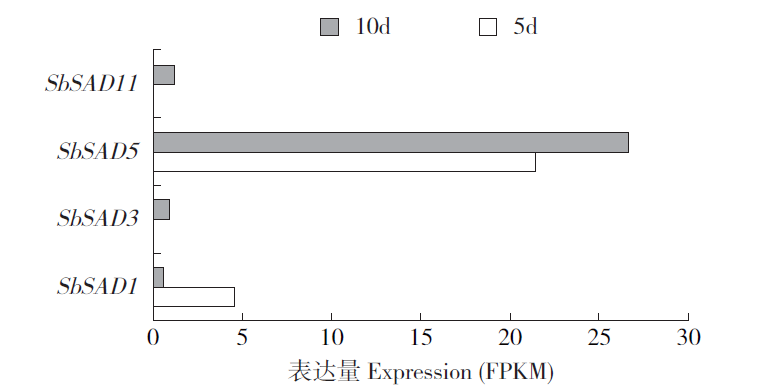
硬脂酰-ACP脱氢酶是形成不饱和脂肪酸的关键酶。利用拟南芥蛋白序列作为比对序列在NCBI和Phytozome数据库进行Blastp同源比对,共鉴定11个SbSADs基因。对SbSAD基因家族进行蛋白特性、进化关系、不同发育阶段表达、基因结构、保守基序、染色体定位、基因二级和三级结构、启动子区的顺式元件及种子发育阶段表达分析,结果显示,SbSADs基因编码区为1 134~1 290bp,其编码蛋白的氨基酸数为377~449,分子量最大为48.0kDa,最小为39.8kDa,等电点为5.26~8.76;根据系统发育树,将SbSAD基因家族分为3个亚族,其中亚族I在不同组织发育阶段表达量较高;11个SbSADs基因不均等地定位在高粱1、2、3、4、6、7和10号染色体上;SbSADs蛋白二级结构均以α-螺旋和无规则卷曲为主,三级结构预测显示,除SbSAD8外,其余SbSADs的蛋白三级结构高度相似;11个SbSADs基因的启动子区与低温胁迫相关的元件数量高度富集。SbSAD5基因在种子发育5和10d均有较高的表达量。
| [1] | 杨士春 . 甜高粱籽粒中脂肪酸含量的气相色谱分析. 中国粮油学报, 2012,27(11):100-104. |
| [2] | 张国琴, 葛玉彬, 张正英 , 等. 高粱抗旱研究综述. 甘肃农业科技, 2018,56(6):67-72. |
| [3] | O'Byrne D J, Knauft D A, Shireman R B . Low fat-monounsaturated rich diets containing high-oleic peanuts improve serum lipoprotein profiles. Lipids, 1997,32(7):687-695. |
| [4] | Carrillo C, Del M C M, Roelofs H , et al. Activation of human neutrophils by oleic acid involves the production of reactive oxygen species and a rise in cytosolic calcium concentration:a comparison with N-6 polyunsaturated fatty acids. Cellular Physiology and Biochemistry, 2011,28(2):329-338. |
| [5] | 袁蕊, 敖宗华, 丁海龙 , 等. 高粱中脂肪酸和低分子有机酸气相色谱测定. 酿酒, 2011,38(4):42-43. |
| [6] | Ohlrogge J, Browse J . Lipid biosynthesis. The Plant Cell, 1995,7(7):957-970. |
| [7] | Xuan W Y, Zhang Y, Liu Z Q , et al. Molecular cloning and expression analysis of a novel BCCP subunit gene from Aleurites moluccana. Genetics and Molecular Research, 2015,14(3):9922-9931. |
| [8] | Gonzalez-Thuillier I, Venegas-Caleron M, Sanchez R , et al. Sunflower (Helianthus annuus) fatty acid synthase complex:beta-hydroxyacyl-[acyl carrier protein] dehydratase genes. Planta, 2016,243(2):397-410. |
| [9] | Rodriguez M F, Sanchez-Garcia A, Salas J J , et al. Characterization of soluble acyl-ACP desaturases from Camelina sativa,Macadamia tetraphylla and Dolichandra unguiscati. Journal of Plant Physiology, 2015,178(15):35-42. |
| [10] | Li-Beisson Y, Shorrosh B, Beisson F , et al. Acyl-lipid metabolism. The Arabidopsis Book, 2013,11:e0161. |
| [11] | Kachroo A, Shanklin J, Whittle E , et al. The Arabidopsis stearoyl-acyl carrier protein-desaturase family and the contribution of leaf isoforms to oleic acid synthesis. Plant Molecular Biology, 2007,63(2):257-271. |
| [12] | Ruddle P N, Whetten R, Cardinal A , et al. Effect of a novel mutation in a △9-stearoyl-ACP-desaturase on soybean seed oil composition. Theoretical and Applied Genetics, 2013,126(1):241-249. |
| [13] | Jung S, Tate P L, Horn R , et al. The phylogenetic relationship of possible progenitors of the cultivated peanut. Journal of Heredity, 2003,94(4):334-340. |
| [14] | Knutzon D S, Thompson G A, Radke S E , et al. Modification of Brassica seed oil by antisense expression of a stearoyl-acyl carrier protein desaturase gene. Proceedings of the National Academy of Sciences of the United States of America, 1992,89(7):2624-2628. |
| [15] | Slocombe S P, Cummins I, Jarvis R P , et al. Nucleotide sequence and temporal regulation of a seed-specific Brassica napus cDNA encoding a stearoyl-acyl carrier protein (ACP) desaturase. Plant Molecular Biology, 1992,20(1):151-155. |
| [16] | Shanklin J, Somerville C . Stearoyl-acyl-carrier-protein desaturase from higher plants is structurally unrelated to the animal and fungal homologs. Proceedings of the National Academy of Sciences of the United States of America, 1991,88(6):2510-2514. |
| [17] | McKeon T A, Stumpf P K . Purification and characterization of the stearoyl-acyl carrier protein desaturase and the acyl-acyl carrier protein thioesterase from maturing seeds of safflower. The Journal of Biological Chemistry, 1982,257(20):12141-12147. |
| [18] | Zhang Y, Maximova S N, Guiltinan M J . Characterization of a stearoyl-acyl carrier protein desaturase gene family from chocolate tree,Theobroma cacao L. Frontiers in Plant Science, 2015,6:239-251. |
| [19] | Lightner J, Wu J, Browse J . A mutant of Arabidopsis with increased levels of stearic acid. Plant Physiology, 1994,106(4):1443-1451. |
| [20] | Osorio J, Fernándezmartínez J, Mancha M , et al. Mutant sunflowers with high concentration of saturated fatty acids in the oil. Crop Science, 1995,35(3):739-742. |
| [21] | Craig W, Lenzi P, Scotti N , et al. Transplastomic tobacco plants expressing a fatty acid desaturase gene exhibit altered fatty acid profiles and improved cold tolerance. Transgenic Research, 2008,17(5):769-782. |
| [22] | Byfield G E, Xue H, Upchurch R G . Two genes from soybean encoding soluble Δ9 stearoyl-ACP desaturases. Crop Science, 2006,46(2):840-846. |
| [23] | Murata N, Ishizaki-Nishizawa O, Higashi S , et al. Genetically engineered alteration in the chilling sensitivity of plants. Nature, 1992,356:710-713. |
| [24] | Kodama H, Horiguchi G, Nishiuchi T , et al. Fatty acid desaturation during chilling acclimation is one of the factors involved in conferring low-temperature tolerance to young tobacco leaves. Plant Physiology, 1995,107(4):1177-1185. |
| [25] | Orlova I V, Serebriiskaya T S, Popov V , et al. Transformation of tobacco with a gene for the thermophilic acyl-lipid desaturase enhances the chilling tolerance of plants. Plant and Cell Physiology, 2003,44(4):447-450. |
| [26] | Dong C G, Cao N, Zhang Z G , et al. Characterization of the fatty acid desaturase genes in cucumber:structure,phylogeny,and expression patterns. PLoS ONE, 2016,11(3):e0149917. |
| [27] | Tocher D R, Leaver M J, Hodgson P A . Recent advances in the biochemistry and molecular biology of fatty acyl desaturases. Progress in Lipid Research, 1998,37(2/3):73-117. |
| [28] | Han Y, Xu G, Du H , et al. Natural variations in stearoyl-acp desaturase genes affect the conversion of stearic to oleic acid in maize kernel. Theoretical and Applied Genetics, 2017,130(1):151-161. |
| [29] | Shang X, Cheng C, Ding J , et al. Identification of candidate genes from the SAD gene family in cotton for determination of cottonseed oil composition. Molecular Genetics and Genomics, 2017,292(1):173-186. |
| [30] | Merlo A O, Cowen N, Delate T , et al. Ribozymes targeted to stearoyl-ACP △9 desaturase mRNA produce heritable increases of stearic acid in transgenic maize leaves. Plant Cell, 1998,10:1603-1621. |
| [1] | 高杰,李青风,李晓荣,封广才,彭秋. 贵州不同年代糯高粱品种(系)干物质生产及光能利用特性差异分析[J]. 作物杂志, 2020, (1): 41–46 |
| [2] | 范昕琦,王海燕,聂萌恩,赵兴奎,张一中,杨慧勇,张晓娟,梁笃,段永红,柳青山. EMS诱变对高粱出苗及农艺性状的影响[J]. 作物杂志, 2020, (1): 47–54 |
| [3] | 宋健,晓宁,王海岗,陈凌,王君杰,刘思辰,乔治军. SiASRs家族基因的鉴定及表达分析[J]. 作物杂志, 2019, (6): 33–42 |
| [4] | 唐桃霞,王致和,施志国,常瑛,张英英,李彦荣. 不同基因型甜高粱对重金属的吸收规律研究[J]. 作物杂志, 2019, (6): 50–56 |
| [5] | 梁晓红,张瑞栋,黄敏佳,刘静,曹雄. 覆膜与施氮互作对高粱产量及水氮利用效率的影响[J]. 作物杂志, 2019, (5): 135–142 |
| [6] | 王劲松,董二伟,焦晓燕,武爱莲,白文斌,王立革,郭珺,韩雄,柳青山. 不同种植模式对高粱晋糯3号产量和养分吸收的影响[J]. 作物杂志, 2019, (5): 166–172 |
| [7] | 岳琳祺,施卫萍,郭佳晖,郭平毅,郭杰. 谷子角质合成基因对干旱胁迫的响应[J]. 作物杂志, 2019, (4): 183–190 |
| [8] | 高杰,李青风,李晓荣,封广才,彭秋. 贵州省不同年代糯高粱品种(系)农艺性状演变分析[J]. 作物杂志, 2019, (4): 17–23 |
| [9] | 李春宏,陆相龙,张培通,苏衍菁,王仪明,郭文琦,殷剑美,韩晓勇,王立,火恩杰. 防除甜高粱田杂草的除草剂筛选[J]. 作物杂志, 2018, (6): 158–161 |
| [10] | 吕亮杰,陈希勇,张业伦,刘茜,王莉梅,马乐,李辉. 小麦GASA基因家族生物信息学分析[J]. 作物杂志, 2018, (6): 58–67 |
| [11] | 张一中,周福平,张晓娟,邵强,杨彬,柳青山. 高粱种质材料光合特性和水分利用效率鉴定及聚类分析[J]. 作物杂志, 2018, (5): 45–53 |
| [12] | 张瑞栋,曹雄,岳忠孝,梁晓红,刘静,黄敏佳. 氮肥和密度对高粱产量及氮肥利用率的影响[J]. 作物杂志, 2018, (5): 110–115 |
| [13] | 张建华,郭瑞峰,曹昌林,范娜,江佰阳,李光,史丽娟,彭之东,白文斌. 几种茎叶除草剂防除高粱田杂草药效和安全性研究[J]. 作物杂志, 2018, (5): 162–166 |
| [14] | 高杰,李青风,彭秋,焦晓燕,王劲松. 不同养分配比对糯高粱物质生产及氮磷钾利用效率的影响[J]. 作物杂志, 2018, (4): 138–142 |
| [15] | 罗海斌, 蒋胜理, 黄诚梅, 曹辉庆, 邓智年, 吴凯朝, 徐林, 陆珍, 魏源文. 甘蔗ScHAK10基因克隆及表达分析[J]. 作物杂志, 2018, (4): 53–61 |
|