作物杂志,2020, 第5期: 33–40 doi: 10.16035/j.issn.1001-7283.2020.05.005
苦荞鼠李糖基转移酶FtF3GT1基因的克隆与转化毛状根研究
卢晓玲1,2( ), 何铭2, 张凯旋2, 廖志勇1(
), 何铭2, 张凯旋2, 廖志勇1( ), 周美亮2(
), 周美亮2( )
)
- 1温州大学生命与环境科学学院,325035,浙江温州
2中国农业科学院作物科学研究所,100081,北京
Study on the Cloning and Transformation of Rhamnose Transferase FtF3GT1 Gene in Tartary Buckwheat
Lu Xiaoling1,2( ), He Ming2, Zhang Kaixuan2, Liao Zhiyong1(
), He Ming2, Zhang Kaixuan2, Liao Zhiyong1( ), Zhou Meiliang2(
), Zhou Meiliang2( )
)
- 1College of Life and Environmental Science, Wenzhou University, Wenzhou 325035, Zhejiang, China
2Institute of Crop Sciences, Chinese Academy of Agricultural Sciences, Beijing 100081, China
摘要:
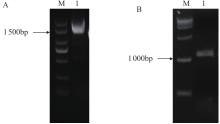
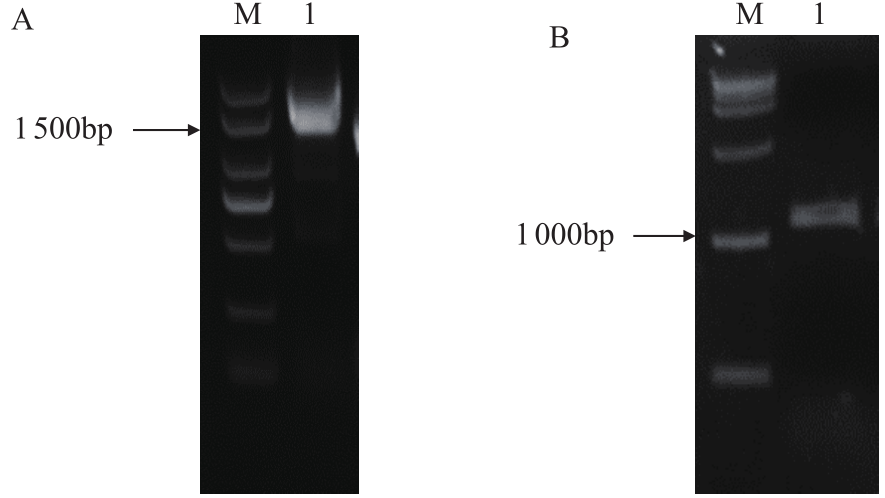
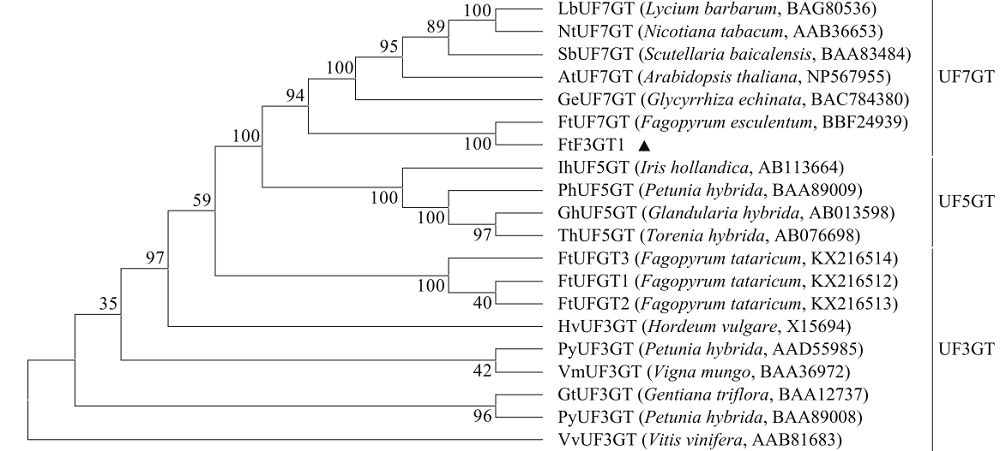
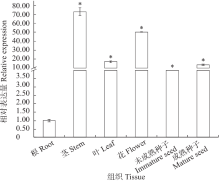
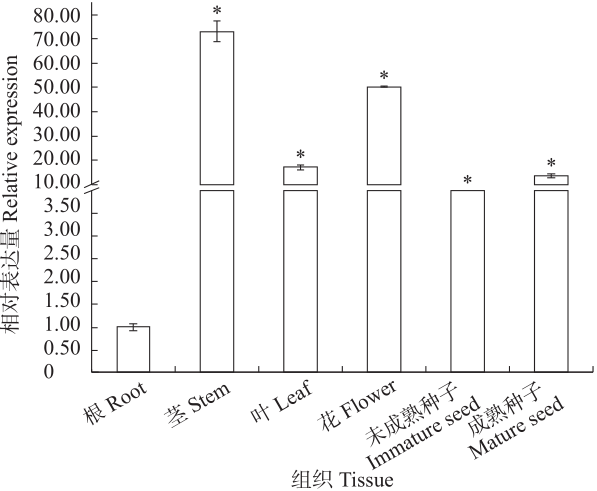
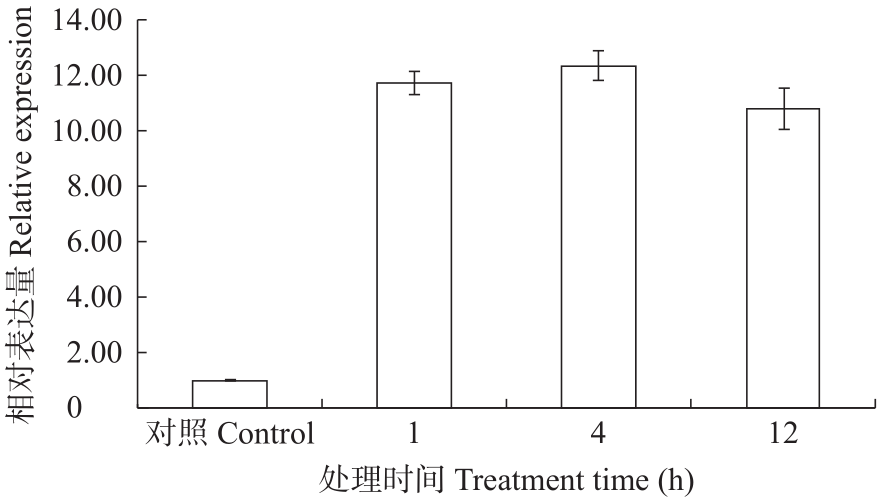
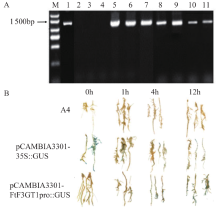
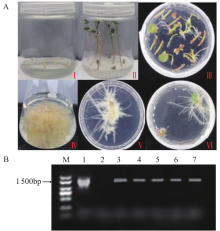
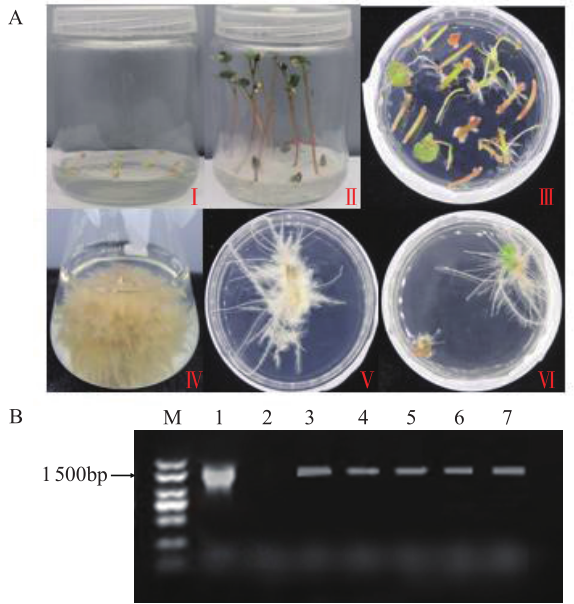
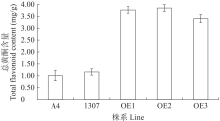
苦荞中的黄酮类化合物具有很高的药用价值和保健功能。以苦荞川荞1号为材料,克隆出FtF3GT1基因,其CDS全长为1 401bp,编码467个氨基酸。系统发育分析表明,FtF3GT1与甜荞中的FeUF7GT蛋白亲缘关系最近。组织特异性表达分析结果表明,FtF3GT1基因在各个组织中均有表达,在根部最低,在茎部最高;MeJA处理苦荞和转pCAMBIA3301:FtF3GT1pro::GUS毛状根中发现,FtF3GT1基因的表达量以及GUS的活性明显受MeJA诱导;转过表达FtF3GT1基因毛状根中,总黄酮含量明显升高,推测FtF3GT1基因可促进黄酮类物质的合成。
| [1] |
Wu J C, Lai C S, Tsai M L, et al. Chemopreventive effect of natural dietary compounds on xenobiotic-induced toxicity. Journal of Food and Drug Analysis, 2017,25(1):176-186.
pmid: 28911535 |
| [2] |
Moon Y J, Wang X, Morris M E. Dietary flavonoids:Effects on xenobiotic and carcinogen metabolism. Toxicology in Vitro, 2006,20(2):187-210.
doi: 10.1016/j.tiv.2005.06.048 pmid: 16289744 |
| [3] |
Cushnie T P T, Lamb A J. Antimicrobial activity of flavonoids. International Journal of Antimicrobial Agents, 2005,26(5):343-356.
doi: 10.1016/j.ijantimicag.2005.09.002 pmid: 16323269 |
| [4] |
Cushnie T P, Lamb A J. Recent advances in understanding the antibacterial properties of flavonoids. International Journal of Antimicrobial Agents, 2011,38(2):99-107.
doi: 10.1016/j.ijantimicag.2011.02.014 |
| [5] |
Buer C S, Imin N, Djordjevic M A. Flavonoids:new roles for old molecules. Journal of Integrative Plant Biology, 2010,52(1):98-111.
doi: 10.1111/j.1744-7909.2010.00905.x |
| [6] | 范昱, 丁梦琦, 张凯旋, 等. 荞麦种质资源概况. 植物遗传资源学报, 2019,20(4):813-828. |
| [7] |
Bai C Z, Feng M L, Hao X L, et al. Rutin,quercetin,and free amino acid analysis in buckwheat (Fagopyrum) seeds from different locations. Genetics and Molecular Research, 2015,14(4):19040-19048.
doi: 10.4238/2015.December.29.11 pmid: 26782554 |
| [8] | 李蒙. 荞麦生物活性肽的研究进展. 江西农业学报, 2018,30(12):17-21. |
| [9] |
Zhou M L, Sun Z M, Ding M Q, et al. FtSAD2 and FtJAZ1 regulate activity of the FtMYB11 transcription repressor of the phenylpropanoid pathway in Fagopyrum tataricum. New Phytologist, 2017,216(3):814-828.
doi: 10.1111/nph.14692 pmid: 28722263 |
| [10] |
Zhou M L, Memelink J. Jasmonate-responsive transcription factors regulating plant secondary metabolism. Biotechnology Advances, 2016,34(4):441-449.
doi: 10.1016/j.biotechadv.2016.02.004 pmid: 26876016 |
| [11] |
An X H, Tian Y, Chen K Q, et al. MdMYB9 and MdMYB11 are involved in the regulation of the JA-induced biosynthesis of anthocyanin and proanthocyanidin in apples. Plant & Cell Physiology, 2015,56(4):650-662.
doi: 10.1093/pcp/pcu205 pmid: 25527830 |
| [12] |
Gonzalez A, Zhao M, Leavitt J M, et al. Regulation of the anthocyanin biosynthetic pathway by the TTG1/bHLH/Myb transcriptional complex in Arabidopsis seedlings. The Plant Journal, 2008,53(5):814-827.
doi: 10.1111/j.1365-313X.2007.03373.x pmid: 18036197 |
| [13] |
Boter M, Golz J F, Gimenez-Ibanez S, et al. Filamentous flower is a direct target of JAZ3 and modulates responses to jasmonate. Plant Cell, 2015,27(11):3160-3174.
doi: 10.1105/tpc.15.00220 pmid: 26530088 |
| [14] |
Lim S H, Kim D H, Kim J K, et al. A radish basic helix-loop-helix transcription factor,RsTT8 acts a positive regulator for anthocyanin biosynthesis. Frontiers in Plant Science, 2017,8:1917-1931.
doi: 10.3389/fpls.2017.01917 pmid: 29167678 |
| [15] |
Tohge T, Watanabe M, Hoefgen R, et al. The evolution of phenylpropanoid metabolism in the green lineage. Critical Reviews in Biochemistry and Molecular Biology, 2013,48(2):123-152.
doi: 10.3109/10409238.2012.758083 |
| [16] |
He F, Mu L, Yan G L, et al. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules, 2010,15(12):9057-9091.
doi: 10.3390/molecules15129057 pmid: 21150825 |
| [17] | Du H, Huang Y B, Tang Y X. Genetic and metabolic engineering of isoflavonoid biosynthesis. Applied Microbiology and Biotech-nology, 2010,86(5):1293-1312. |
| [18] | Rio D C, Ares M, Hannon G J, et al. Purification of RNA using trizol. Cold Spring Harbor Protocols, 2010,(6):1-3. |
| [19] | 谈天斌, 卢晓玲, 张凯旋, 等. TrMYB308基因的克隆及在苦荞毛状根中的功能分析. 植物遗传资源学报, 2019,20(6):1542-1553. |
| [20] |
Zhou M L, Zhu X M, Shao J R, et al. Transcriptional response of the catharanthine biosynthesis pathway to methyl jasmonate/nitric oxide elicitation in Catharanthus roseus hairy root culture. Applied Microbiology and Biotechnology, 2010,88(3):737-750.
doi: 10.1007/s00253-010-2822-x |
| [21] | 李为喜. AlCl3分光光度法测定荞麦种质资源中黄酮的研究. 植物遗传资源学报, 2008,9(4):502-505. |
| [22] |
Nakatsuka T, Sato K, Takahashi H, et al. Cloning and characterization of the UDP-glucose:anthocyanin 5-O-glucosyltransferase gene from blue-flowered gentian. Journal of Experimental Botany, 2008,59(6):1241-1252.
doi: 10.1093/jxb/ern031 pmid: 18375606 |
| [23] |
Hsu Y H, Tagami T, Matsunaga K, et al. Functional characterization of UDP-rhamnose-dependent rhamnosyltransferase involved in anthocyanin modification,a key enzyme determining blue coloration in Lobelia erinus. The Plant Journal, 2017,89(2):325-337.
doi: 10.1111/tpj.13387 pmid: 27696560 |
| [24] |
Miller K D, Guyon V, Evans J N S, et al. Purification,cloning,and heterologous expression of a catalytically efficient flavonol 3-O-galactosyltransferase expressed in the male gametophyte of Petunia hybrida. The Journal of Biological Chemistry, 1999,274(48):34011-34019.
doi: 10.1074/jbc.274.48.34011 pmid: 10567367 |
| [25] |
Li X J, Zhang J Q, Wu Z C, et al. Functional characterization of a glucosyltransferase gene,LcUFGT1,involved in the formation of cyanidin glucoside in the pericarp of Litchi chinensis. Physiologia Plantarum, 2016,156(2):139-149.
doi: 10.1111/ppl.12391 pmid: 26419221 |
| [26] | Campell J A, Davies G J, Bulone V, et al. A classification of nucleotide-diphospho-sugar glycosyltransferases based on amino acid sequence similarities. Biochemistry, 1998,329(3):719. |
| [27] | 胡滢滢, 薛飞燕, 杨明峰, 等. 高山红景天UGT72B14G2基因克隆及其原核表达. 北京农学院学报, 2016,31(4):1-5. |
| [28] |
Koja E, Ohata S, Maruyama Y, et al. Identification and characterization of a rhamnosyltransferase involved in rutin biosynthesis in Fagopyrum esculentum (common buckwheat). Bioscience,Biotechnology, and Biochemistry, 2018,82(10):1790-1802.
doi: 10.1080/09168451.2018.1491286 pmid: 29972345 |
| [29] |
Perez A C, Goossens A. Jasmonate signalling:a copycat of auxin signalling? Plant, Cell and Environment, 2013,36(12):2071-2084.
doi: 10.1111/pce.12121 pmid: 23611666 |
| [30] |
Memelink J, Verpoorte R, Jan W K. ORCAnization of jasmonate-responsive gene expression in alkaloid metabolism. Trends in Plant Science, 2001,6(5):212-219.
doi: 10.1016/s1360-1385(01)01924-0 pmid: 11335174 |
| [31] |
Misra P, Pandey A, Tiwari M, et al. Modulation of transcriptome and metabolome of tobacco by Arabidopsis transcription factor,AtMYB12,leads to insect resistance. Plant Physiology, 2010,152(4):2258-2268.
doi: 10.1104/pp.109.150979 pmid: 20190095 |
| [32] |
Zhang K, Logacheva M D, Meng Y, et al. Jasmonate-responsive MYB factors spatially repress rutin biosynthesis in Fagopyrum tataricum. Journal of Experimental Botany, 2018,69(8):1955-1966.
doi: 10.1093/jxb/ery032 pmid: 29394372 |
| [33] |
Zhang D, Jiang C L, Huang C H, et al. The light-induced transcription factor FtMYB116 promotes accumulation of rutin in Fagopyrum tataricum. Plant, Cell and Environment, 2018,42:1340-1351.
doi: 10.1111/pce.13470 pmid: 30375656 |
| [1] | 杨学乐, 张璐, 李志清, 何录秋. 苦荞种质资源表型性状的遗传多样性分析[J]. 作物杂志, 2020, (5): 53–58 |
| [2] | 李春花, 黄金亮, 尹桂芳, 王艳青, 卢文洁, 孙道旺, 王春龙, 郭来春, 洪波, 任长忠, 王莉花. 苦荞粒形相关性状的遗传分析[J]. 作物杂志, 2020, (3): 42–46 |
| [3] | 马成瑞,向达兵,万燕,欧阳建勇,宋月,唐正松,刘建英,赵钢. 不同苦荞品种花和籽粒空间分布特征及差异分析[J]. 作物杂志, 2020, (1): 35–40 |
| [4] | 杨甜,张永清,董馥慧,马星星,薛小娇. 不同水分条件下不同抗旱性苦荞根系生长规律研究[J]. 作物杂志, 2019, (6): 76–82 |
| [5] | 宋丽芳,冯美臣,张美俊,肖璐洁,王超,杨武德,宋晓彦. 外源硒对苦荞生长发育及子粒硒含量的影响[J]. 作物杂志, 2019, (3): 150–154 |
| [6] | 马名川,刘龙龙,张丽君,崔林,周建萍. EMS诱变刺荞的形态突变体鉴定与分析[J]. 作物杂志, 2019, (3): 37–41 |
| [7] | 崔娅松, 王艳, 杨丽娟, 吴朝昕, 周飘, 冉盼, 陈庆富. 米苦荞果壳率及其相关性状的遗传研究[J]. 作物杂志, 2019, (2): 51–60 |
| [8] | 赵鑫,陈少锋,王慧,刘三才,杨修仕,张宝林. 晋北地区不同苦荞品种产量和品质研究[J]. 作物杂志, 2018, (5): 27–32 |
| [9] | 罗海斌, 蒋胜理, 黄诚梅, 曹辉庆, 邓智年, 吴凯朝, 徐林, 陆珍, 魏源文. 甘蔗ScHAK10基因克隆及表达分析[J]. 作物杂志, 2018, (4): 53–61 |
| [10] | 范昱,王红力,何凤,赖弟利,王佳俊,宋月,向达兵. 后熟对苦荞子粒营养品质的影响[J]. 作物杂志, 2018, (1): 96–101 |
| [11] | 宋月,向达兵,黄后兵,范昱,韦爽,张赛. 苦荞品种抗倒性鉴定及评价方法的研究[J]. 作物杂志, 2017, (6): 65–71 |
| [12] | 杨修仕,郭忠贤,郭慧敏,王慧,刘三才. 播期和播量对荞麦产量及主要品质的影响[J]. 作物杂志, 2017, (1): 88–93 |
| [13] | 韩赞平,陈彦惠,郭书磊,祖小峰,王顺喜,赵西拥. 玉米抗逆基因ZmqLTG3-1的克隆及功能分析[J]. 作物杂志, 2016, (4): 47–55 |
| [14] | 袁红梅,郭文栋,赵丽娟,于莹,吴建忠,程莉莉,赵东升,康庆华,黄文功,姚玉波,宋喜霞,姜卫东,刘岩,马廷芬,吴广文,关凤芝. 亚麻糖基转移酶基因LuUGT72E1的克隆与表达分析[J]. 作物杂志, 2016, (4): 62–67 |
| [15] | 吴鹏, 郭茜茜, 武涛, 等. 黄瓜ABC转运蛋白基因(abca19)的克隆及其对农药霜霉威胁迫的响应[J]. 作物杂志, 2015, (3): 45–51 |
|
||