作物杂志,2022, 第3期: 92–98 doi: 10.16035/j.issn.1001-7283.2022.03.013
不同夜间温度处理对马铃薯试管薯及块茎形成相关基因表达的影响
刘菊( ), 李广存, 段绍光, 胡军, 简银巧, 刘建刚, 金黎平, 徐建飞(
), 李广存, 段绍光, 胡军, 简银巧, 刘建刚, 金黎平, 徐建飞( )
)
- 中国农业科学院蔬菜花卉研究所/农业农村部薯类作物生物学和遗传育种重点实验室,100081,北京
The Effects of Different Night Temperature Treatments on in vitro Tuberization and Related-Genes Expression in Potato
Liu Ju( ), Li Guangcun, Duan Shaoguang, Hu Jun, Jian Yinqiao, Liu Jiangang, Jin Liping, Xu Jianfei(
), Li Guangcun, Duan Shaoguang, Hu Jun, Jian Yinqiao, Liu Jiangang, Jin Liping, Xu Jianfei( )
)
- Institute of Vegetables and Flowers, Chinese Academy of Agricultural Sciences/Key Laboratory of Biology and Genetic Improvement of Tuber and Root Crops, Ministry of Agriculture and Rural Affairs, Beijing 100081, China
摘要:
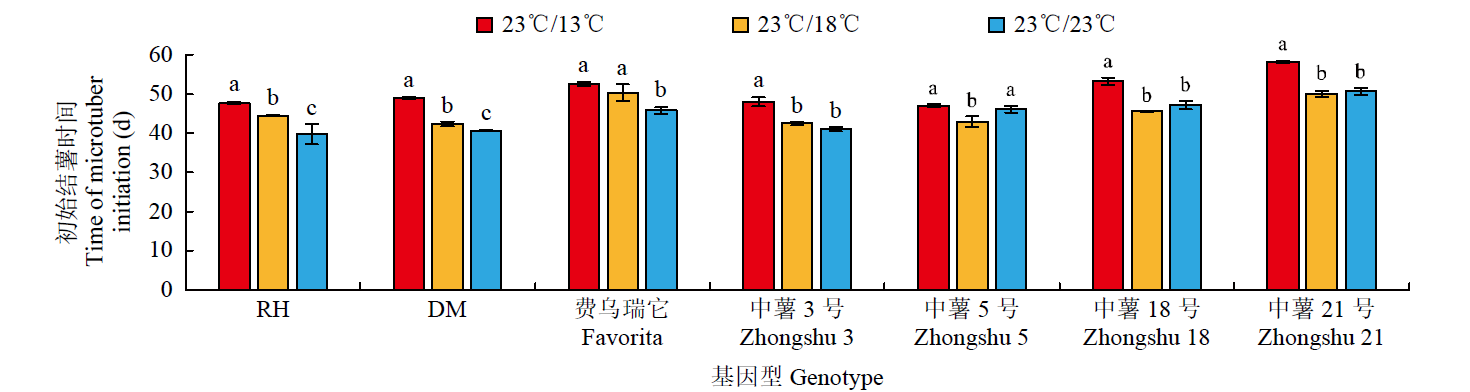
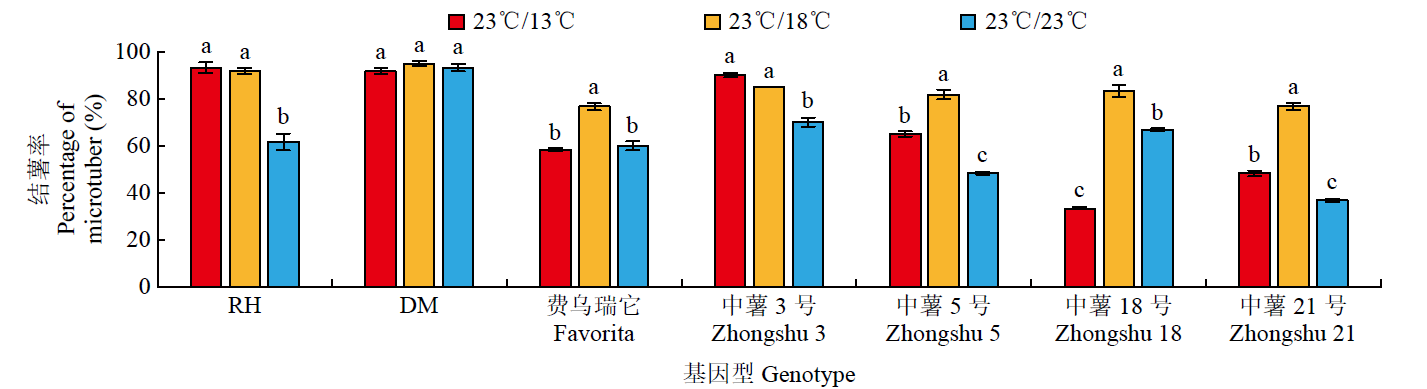
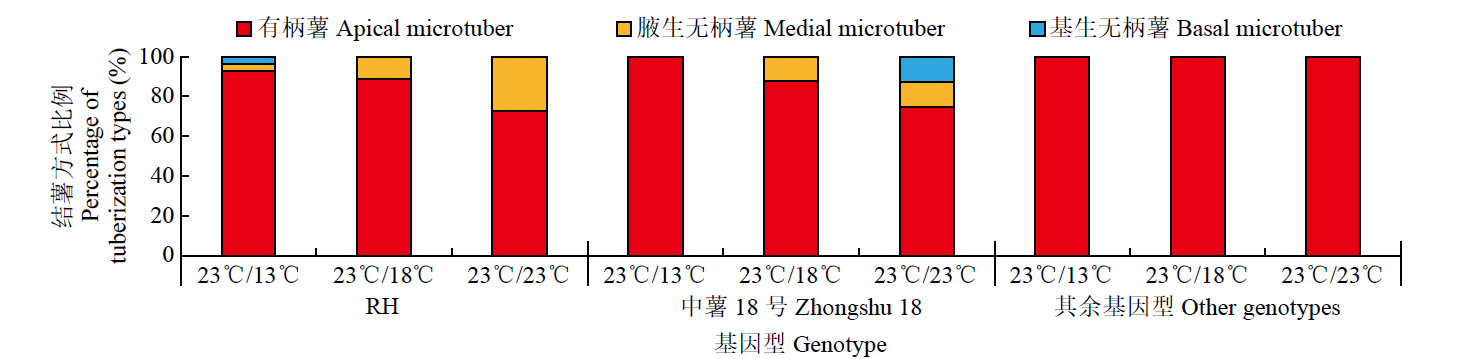
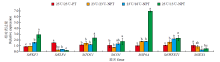
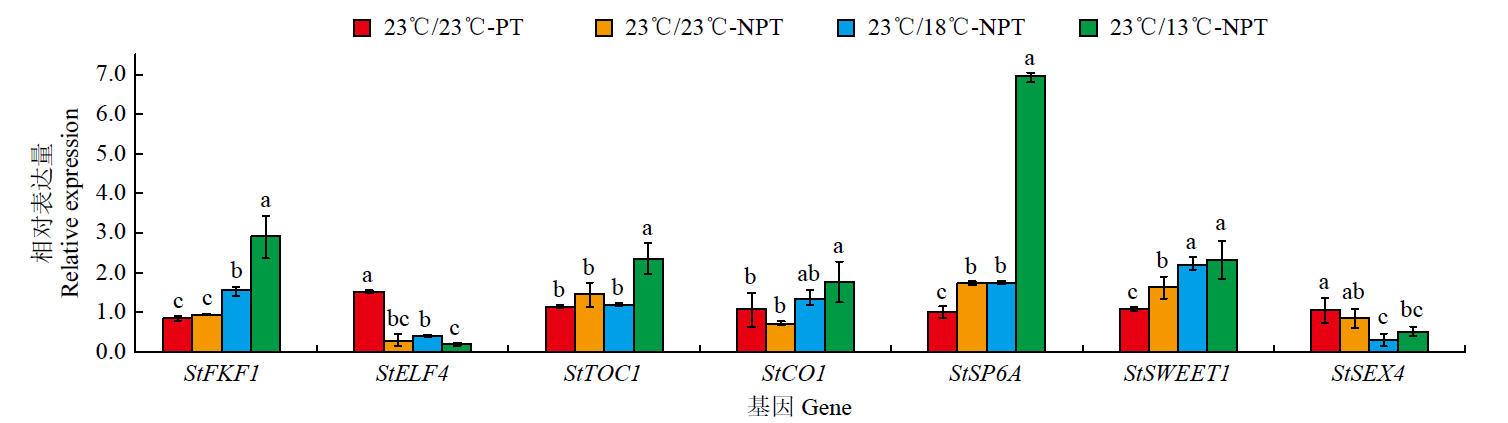
为探究不同夜间温度处理对马铃薯试管薯形成的影响,以7个马铃薯品种或材料的试管苗为试材,在昼/夜温度分别为23℃/13℃、23℃/18℃和23℃/23℃条件下进行试管薯的诱导培养,调查记录试管薯初始结薯时间、结薯率和结薯方式变化,并分析不同夜间温度处理下试管薯初始形成植株及同期未形成试管薯植株参与块茎形成相关基因的表达量变化。结果表明,对于大多数基因型,与其他处理相比,23℃/13℃处理显著延长试管薯初始结薯时间,23℃/23℃处理降低结薯率,而23℃/18℃处理结薯较早且试管薯结薯率平均达84.3%,最利于试管薯形成。夜间温度可通过影响生物钟、糖代谢和光周期通路中StFKF1、StCO1和StSWEET1等相关基因的表达而调控块茎形成。
| [1] | 徐建飞, 金黎平. 马铃薯遗传育种研究:现状与展望. 中国农业科学, 2017, 50(6):990-1015. |
| [2] |
Levy D, Veilleux R E. Adaptation of potato to high temperatures and salinity-a review. American Journal of Potato Research, 2007, 84(6):487-506.
doi: 10.1007/BF02987885 |
| [3] |
Mozos A T, Morris W L, Ducreux L J M, et al. Engineering heat tolerance in potato by temperature-dependent expression of a specific allele of HEAT-SHOCK COGNATE 70. Plant Biotechnology Journal, 2018, 16(1):197-207.
doi: 10.1111/pbi.12760 |
| [4] |
Sillmann J, Kharin V V, Zwiers F W, et al. Climate extremes indices in the CMIP5 multimodel ensemble:Part 2. future climate projections. Journal of Geophysical Research:Atmospheres, 2013, 118(6):2473-2493.
doi: 10.1002/jgrd.50188 |
| [5] |
Gregory L E. Some factors for tuberization in the potato plant. American Journal of Botany, 1956, 43(4):281-288.
doi: 10.1002/j.1537-2197.1956.tb10492.x |
| [6] |
Slater J W. The effect of night temperature on tuber initiation of the potato. European Potato Journal, 1968, 11(1):14-22.
doi: 10.1007/BF02365158 |
| [7] |
Singh B, Kukreja S, Goutam U. Impact of heat stress on potato (Solanum tuberosum L.):present scenario and future opportunities. The Journal of Horticultural Science and Biotechnology, 2020, 95(4):407-424.
doi: 10.1080/14620316.2019.1700173 |
| [8] |
Muthoni J, Kabira J N. Potato production in the hot tropical areas of Africa:progress made in breeding for heat tolerance. Journal of Agricultural Science, 2015, 7(9):220-227.
doi: 10.5539/jas.v7n12p220 |
| [9] |
Kim Y U, Lee B W. Differential mechanisms of potato yield loss induced by high day and night temperatures during tuber initiation and bulking:photosynthesis and tuber growth. Frontiers in Plant Science, 2019, 10:300.
doi: 10.3389/fpls.2019.00300 |
| [10] |
Jackson S D. Multiple signaling pathways control tuber induction in potato. Plant Physiology, 1999, 119(1):1-8.
doi: 10.1104/pp.119.1.1 |
| [11] | Navarro C, Abelenda J A E, Cue´llar C A, et al. Control of flowering and storage organ formation in potato by FLOWERING LOCUS T. Nature:International Weekly Journal of Science, 2011, 478(7367):119-122. |
| [12] |
Lehretz G G, Sonnewald S, Hornyik C, et al. Post-transcriptional regulation of FLOWERING LOCUS T modulates heat-dependent source-sink development in potato. Current Biology, 2019, 29(10):1614-1624.
doi: S0960-9822(19)30425-7 pmid: 31056391 |
| [13] |
Morris W L, Ducreux L J M, Morris J, et al. Identification of TIMING OF CAB EXPRESSION 1 as a temperature-sensitive negative regulator of tuberization in potato. Journal of Experimental Botany, 2019, 70(20):5703-5714.
doi: 10.1093/jxb/erz336 pmid: 31328229 |
| [14] | Hastilestari B R. Molecular analysis of potato (Solanum tuberosum) responses to increased temperatures. Nuremberg:Friedrich Alexander University, 2019. |
| [15] | 张小川, 颉瑞霞, 王效瑜, 等. 试管薯诱导期温度对‘宁薯14号’结薯率的效果. 中国马铃薯, 2017, 31(3):134-137. |
| [16] | 周俊. 马铃薯(Solanum tuberosum L.)试管块茎形成的QTL定位及遗传分析. 武汉:华中农业大学, 2014. |
| [17] |
Pantelić D, Dragićević I Č,Rudić J, et al. Effects of high temperature on in vitro tuberization and accumulation of stress-responsive proteins in potato. Horticulture,Environment,and Biotechnology, 2018, 59(3):315-324.
doi: 10.1007/s13580-018-0043-x |
| [18] |
Shah F, Nie L X, Cui K H, et al. Rice grain yield and component responses to near 2°C of warming. Field Crops Research, 2014, 157:98-110.
doi: 10.1016/j.fcr.2013.12.014 |
| [19] |
Fang S B, Cammarano D, Zhou G S, et al. Effects of increased day and night temperature with supplemental infrared heating on winter wheat growth in North China. European Journal of Agronomy, 2015, 64:67-77.
doi: 10.1016/j.eja.2014.12.012 |
| [20] | 梁俊梅, 贾立国, 秦永林, 等. 马铃薯试管薯形成因素的研究进展. 北方农业学报, 2016, 44(4):105-108. |
| [21] | 赵佐敏. 马铃薯组培中不同因素对诱导试管薯的影响. 中国马铃薯, 2005, 19(5):26-28. |
| [22] | 张武, 齐恩芳, 王一航, 等. 马铃薯试管薯诱导集成优化研究. 长江蔬菜, 2008(12):31-33. |
| [23] | 邱甜, 牛力立, 朱江, 等. 蔗糖浓度、温度及光照强度对试管薯诱导的影响. 安顺学院学报, 2020, 22(5):121-123,135. |
| [24] | 蒋从莲, 郭华春. 不同外源激素和培养温度对马铃薯试管薯形成的影响. 云南农业大学学报, 2007, 22(6):824-828. |
| [25] |
Gould P D, Locke J C, Larue C, et al. The molecular basis of temperature compensation in the Arabidopsis circadian clock. The Plant Cell, 2006, 18(5):1177-1187.
doi: 10.1105/tpc.105.039990 |
| [26] |
Hancock R D, Morris W L, Ducreux L J, et al. Physiological,biochemical and molecular responses of the potato (Solanum tuberosum L.) plant to moderately elevated temperature. Plant,Cell,Environment, 2014, 37(2):439-450.
doi: 10.1111/pce.12168 |
| [27] |
Abelenda J A, Bergonzi S, Oortwijn M, et al. Source-sink regulation is mediated by interaction of an FT homolog with a SWEET protein in potato. Current Biology, 2019, 29(7):1178-1186.
doi: S0960-9822(19)30157-5 pmid: 30905604 |
| [28] | 刘洋, 林希昊, 姚艳丽, 等. 高等植物蔗糖代谢研究进展. 中国农学通报, 2012, 28(6):145-152. |
| [29] |
Putterill J, Robson F, Lee K, et al. The CONSTANS gene of Arabidopsis promotes flowering and encodes a protein showing similarities to zinc finger transcription factors. Cell, 1995, 80(6):847-857.
pmid: 7697715 |
| [30] |
González-Schain N D, Díaz-Mendoza M, Żurczak M, et al. Potato CONSTANS is involved in photoperiodic tuberization in a graft‐transmissible manner. The Plant Journal, 2012, 70(4):678-690.
doi: 10.1111/j.1365-313X.2012.04909.x pmid: 22260207 |
| [31] |
Singh A, Siddappa S, Bhardwaj V, et al. Expression profiling of potato cultivars with contrasting tuberization at elevated temperature using microarray analysis. Plant Physiology and Biochemistry, 2015, 97:108-116.
doi: 10.1016/j.plaphy.2015.09.014 |
| [32] |
Hastilestari B R, Lorenz J, Reid S, et al. Deciphering source and sink responses of potato plants (Solanum tuberosum L.) to elevated temperatures. Plant Cell Environment, 2018, 41(11):2600-2616.
doi: 10.1111/pce.13366 |
| [1] | 谢奎忠, 孙小花, 罗爱花, 柳永强, 唐德晶, 朱永永, 胡新元. 基施锌肥对长期连作马铃薯抗病性相关酶活性、土传病害和产量的影响[J]. 作物杂志, 2022, (4): 154–159 |
| [2] | 陈玉珍, 唐广彬, 马宪新, 田贵云, 于宏心, 骆璎珞, 樊明寿, 贾立国. 马铃薯块茎发育的四大调控途径[J]. 作物杂志, 2022, (4): 9–13 |
| [3] | 张小全, 贾振宇, 李举旭, 李洪臣, 王豹祥, 汪健, 石刚, 王川, 武云杰. 不同促根措施对皖南烤烟成熟期钾素代谢的影响[J]. 作物杂志, 2022, (3): 205–210 |
| [4] | 赵锴, 晋秀娟, 孙丽丽, 闫荣岳, 卢娟, 郭峰, 史雨刚, 孙黛珍. 小麦脱植基相关基因在春性小麦叶片叶绿素降解过程中的作用分析[J]. 作物杂志, 2022, (2): 81–88 |
| [5] | 杨志楠, 黄金文, 韩凡香, 李亚伟, 马建涛, 柴守玺, 程宏波, 杨德龙, 常磊. 秸秆带状覆盖对西北雨养区马铃薯农田土壤温度及产量的影响[J]. 作物杂志, 2022, (1): 196–204 |
| [6] | 高佳, 王姣, 王松, 刘红健, 康佳, 沈弘, 王海莉, 任少勇. 生物炭基肥对马铃薯田土壤脲酶活性和产量的影响[J]. 作物杂志, 2021, (6): 134–138 |
| [7] | 罗磊, 李亚杰, 姚彦红, 李丰先, 范奕, 董爱云, 刘惠霞, 牛彩萍, 李德明. 不同小整薯规格和药剂拌种处理对旱作重茬马铃薯生长及产量的影响[J]. 作物杂志, 2021, (6): 211–216 |
| [8] | 李鑫, 金光辉, 王鹏程, 王紫雯. 马铃薯品种(系)淀粉与产量表现稳定性分析[J]. 作物杂志, 2021, (6): 51–57 |
| [9] | 张微, 李志新, 赵雪, 张金鹏, 付春江, 于倩倩, 刘卫平. 双重检测马铃薯X和Y病毒试纸条制备技术研究[J]. 作物杂志, 2021, (6): 62–66 |
| [10] | 龚林涛, 苏秀娟, 廖燕, 克热木汗·吾斯曼, 周迪, 尹松松. 薰衣草芳樟醇合酶基因的克隆、表达及酶活性检测[J]. 作物杂志, 2021, (6): 78–87 |
| [11] | 娄树宝, 李凤云, 田国奎, 王海艳, 田振东, 王立春, 刘喜才, 王辉. 马铃薯种质资源晚疫病抗性评价及分子标记辅助筛选[J]. 作物杂志, 2021, (4): 196–201 |
| [12] | 杨平, 陈昱利, 巩法江, 毕海滨, 高明慧. 马铃薯块茎膨大特性及其与单薯鲜重之间的相关性[J]. 作物杂志, 2021, (2): 130–134 |
| [13] | 邱甜, 牛力立, 朱江, 蔡甫格, 王庆伟. 3种生长调节剂对马铃薯试管苗生长的影响[J]. 作物杂志, 2021, (2): 160–164 |
| [14] | 王通, 赵孝东, 甄萍萍, 陈静, 陈明娜, 陈娜, 潘丽娟, 王冕, 许静, 禹山林, 迟晓元, 张建成. 花生TCP转录因子的全基因组鉴定及组织表达特性分析[J]. 作物杂志, 2021, (2): 35–44 |
| [15] | 段惠敏, 卢潇, 周晓洁, 李高峰, 文国宏, 王玉萍, 程李香, 张峰. 马铃薯叶型和种植密度对产量组分的影响[J]. 作物杂志, 2021, (1): 160–167 |
|
||