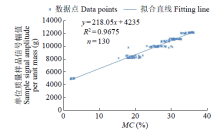
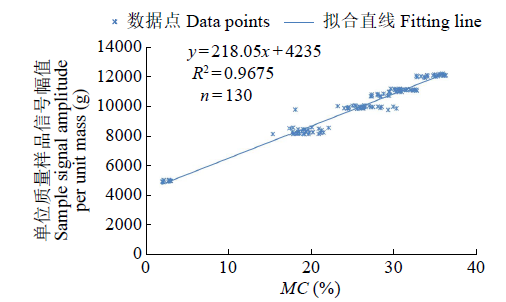
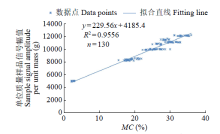
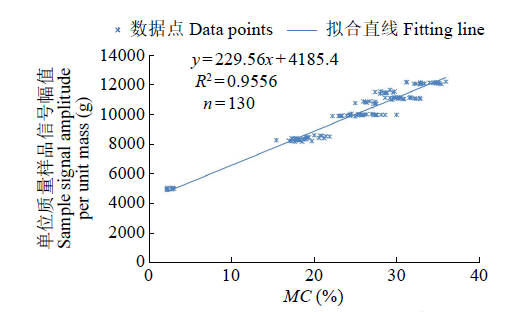
Crops ›› 2023, Vol. 39 ›› Issue (4): 253-259.doi: 10.16035/j.issn.1001-7283.2023.04.036
Previous Articles Next Articles
Research on the Moisture Content Variation and Influence to Rice Seed Germination under Salt and Alkali Stress by Low Field NMR
Yang Hongwei1,2( ), Zhang Liying3, Li Xiaohui1,2(
), Zhang Liying3, Li Xiaohui1,2( )
)
- 1College of Information and Electrical Engineering, Shenyang Agricultural University, Shenyang 110866, Liaoning, China
2Liaoning Engineering Research Center for Information Technology in Agriculture, Shenyang 110866, Liaoning, China
3Liaoning Rice Research Institute, Shenyang 110161, Liaoning, China
| [1] | 王海莲, 张华文, 刘宾, 等. 低度盐胁迫下高粱苗期相关性状的QTL定位. 分子植物育种, 2017, 15(2):604-610. |
| [2] |
Han L, Liu H, Yu S, et al. Potential application of oat for phytoremediation of salt ions in coastal saline-alkali soil. Ecological Engineering, 2013, 61:274-281.
doi: 10.1016/j.ecoleng.2013.09.034 |
| [3] |
Alvarez-Acosta C, Marrero-Dominguez A, Gallo-Llobet L, et al. Physiological response of selected avocados (Persea americana) subjected to NaCl and NaHCO3 stress. Scientia Horticulturae, 2018, 237:81-88.
doi: 10.1016/j.scienta.2018.04.010 |
| [4] |
Yan W, Gang Q J, Yin N H, et al. Effects of salt, alkali and salt- alkali mixed stresses on seed germination of the halophyte Salsola ferganica (Chenopodiaceae). Acta Ecologica Sinica, 2013, 33(6):354-360.
doi: 10.1016/j.chnaes.2013.09.010 |
| [5] |
Fan X D, Wang J Q, Yang N, et al. Gene expression profiling of soybean leaves and roots under salt, saline-alkali and drought stress by high-throughput Illumina sequencing. Gene, 2013, 512(2):392-402.
doi: 10.1016/j.gene.2012.09.100 |
| [6] |
Lin J, Yu D, Shi Y, et al. Salt-alkali tolerance during germination and establishment of Leymus chinensis in the Songnen Grassland of China. Ecological Engineering, 2016, 95:763-769.
doi: 10.1016/j.ecoleng.2016.07.011 |
| [7] | Wang F, Xiao H L, Peng X M, et al. Effects of salt and alkali stress on Reaumuria soongorica germination. Sciences in Cold and Arid Regions, 2017, 9(2):158-166. |
| [8] |
Guo R, Zhou J, Ren G X, et al. Physiological responses of linseed seedlings to iso osmotic polyethylene glycol, salt, and alkali stresses. Agronomy Journal, 2013, 105(3):764-772.
doi: 10.2134/agronj2012.0442 |
| [9] | 李双男, 郭慧娟, 王晶, 等. 不同盐碱胁迫对棉花种子萌发的影响. 种子, 2018, 37(1):38-45. |
| [10] | 刘建新, 王金成, 王瑞娟, 等. 盐、碱胁迫对燕麦幼苗光合作用的影响. 干旱地区农业研究, 2015, 33(6):155-160. |
| [11] |
Gong B, Wen D, Bloszies S, et al. Comparative effects of NaCl and NaHCO3 stresses on respiratory metabolism, antioxidant system, nutritional status,and organic acid metabolism in tomato roots. Acta Physiologiae Plantarum, 2014, 36(8):2167-2181.
doi: 10.1007/s11738-014-1593-x |
| [12] |
Gong B, Dan W, Vandenlangenberg K, et al. Comparative effects of NaCl and NaHCO3 stress on photosynthetic parameters, nutrient metabolism, and the antioxidant system in tomato leaves. Scientia Horticulturae, 2013, 157(3):1-12.
doi: 10.1016/j.scienta.2013.03.032 |
| [13] | 季平, 张鹏, 徐克章, 等. 不同类型盐碱胁迫对大豆植株生长性状和产量的影响. 大豆科学, 2013, 32(4):477-481. |
| [14] |
Lin J, Shao S, Wang Y, et al. Germination responses of the halophyte Chloris virgata to temperature and reduced water potential caused by salinity, alkalinity and drought stress. Grass and Forage Science, 2016, 71(3):507-514.
doi: 10.1111/gfs.2016.71.issue-3 |
| [15] | 张彦妮, 李博, 何淼. 盐胁迫对大花飞燕草种子萌发的影响. 草业科学, 2012, 29(8):1235-1239. |
| [16] |
Zhang H, Zhang G, Lü X, et al. Salt tolerance during seed germination and early seedling stages of 12 halophytes. Plant and Soil, 2015, 388(1/2):229-241.
doi: 10.1007/s11104-014-2322-3 |
| [17] |
He L, Lin B, Hong Y, et al. Assessing the moisture migration during microwave drying of coal using low-field nuclear magnetic resonance. Drying Technology, 2017, 36(5):1-11.
doi: 10.1080/07373937.2017.1326130 |
| [18] | 要世瑾, 杜光源, 牟红梅, 等. 基于核磁共振技术检测小麦植株水分分布和变化规律. 农业工程学报, 2014, 30(24):177- 186. |
| [19] | 牟红梅, 何建强, 邢建军, 等. 小麦灌浆过程籽粒水分变化的核磁共振检测. 农业工程学报, 2016, 32(8):98-104. |
| [20] |
Qiu Q Z, Wei L, Hao K L, et al. Low-field nuclear magnetic resonance for online determination of water content during sausage fermentation. Journal of Food Engineering, 2017, 212:291-297.
doi: 10.1016/j.jfoodeng.2017.05.021 |
| [21] |
Sandnes R, Simon S, Sjöblom J, et al. Optimization and validation of low field nuclear magnetic resonance sequences to determine low water contents and water profiles in W/O emulsions. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2014, 441(3):441-448.
doi: 10.1016/j.colsurfa.2013.09.030 |
| [1] | Zhang Yemeng, Lu Kexin, Mao Wenyu, Zhang Xiaoyan, Li Wenjing, Yang Xiu. Identification and Expression Analysis of LEA Gene Family in Chenopodium quinoa Willd. under Salt Stress [J]. Crops, 2025, 41(6): 100-111. |
| [2] | Guo Longyu, Gao Xinmei, Fu Ying, Zhang Mingwei, Wang Ying, Li Wurijimusi, Wurilige , Wang Yingjie, Wang Jingyu, Yang Fengting, Gu Yanru, Quan Yu. Effect of Immune Inducer ZNC on Seedling Growth and Physiological Response of Oat under Saline-Alkali Stress [J]. Crops, 2025, 41(6): 203-209. |
| [3] | Chen Yi, Chen Xiao, Zhang Maoxing, Li Jing, Chang Jingjing, Li Jiawei, Lin Haiqing, Chen Xingping, Deng Xiaoliang, Xie Dasen, Guo Shaolong, Shen Zhongdeng, Zhang Baige. Response of Different Wax Gourd Varieties to Low-Salt Stress and Comprehensive Evaluation of Their Salt Tolerance [J]. Crops, 2025, 41(6): 58-66. |
| [4] | Teng Wen, Ye Fan, Zhou Zhou, Wang Yule, Liu Lijun. Effects of Wheat and Rapeseed Straw Returning on Yield and Quality of Rice under Salt Stress [J]. Crops, 2025, 41(5): 11-18. |
| [5] | Xu Mingli, Wu Baichen, Liu Chang, Gao Xinhan, Yin Jiaqi, He Xue, Liu Ying, Yin Zequn, Miao Xingfen. Effect of Melatonin Soaking on Foxtail Millet Germination under Saline-Alkali Stress [J]. Crops, 2025, 41(5): 42-46. |
| [6] | Yan Jingrong, Pang Chunhua, Zhang Yongqing, Wu Yueyue, Hou Yuchen, Wang Jiaqi, Qiao Man. Effects of Desulfurized Gypsum and Humic Acid Interaction on Soil and Quinoa Growth in Saline-Alkali Land [J]. Crops, 2025, 41(5): 47-53. |
| [7] | Zhang Jindong, Wang Cheng, Lu Huan, Zeng Lingling, Zhang Gongliang, Sun Haoyue, Liu Yue, Yang Helin, Hou Xiaomin. Response of Mung Bean Genotypes to Exogenous Brassinosteroids under Saline-Alkali Stress [J]. Crops, 2025, 41(5): 54-60. |
| [8] | Chen Ping, Luo Yuanyuan, Wang Juan, Sun Quan, Ma Lingfang, Ma Wenli, Xie Jingbo. Response and Tolerance Evaluation of Oat to Mixed Saline-Alkali Stress at Germination Stage [J]. Crops, 2025, 41(5): 61-66. |
| [9] | Wang Yanwei, Wu Junxi, Wang Yan, Mu Tao, Langzhuoma , Miao Yanjun. Effects of Saline-Alkali Stress on Seed Germination of Urtica dioica L. [J]. Crops, 2025, 41(5): 67-73. |
| [10] | Zhao Zhou, Zhang Li, Gao Xinlei, Qiu Hongyu. Effects of Compound Saline-Alkali Stress on Growth and Metabolism of Oat [J]. Crops, 2025, 41(5): 74-85. |
| [11] | Jing Maoya, Zhang Ziyu, Zhang Meng, He Jiamin, Yan Fanfan, Gao Yanmei, Zhang Yongqing. Effects of Seed Soaking with Salicylic Acid on Seed Germination and Seedling Growth of Quinoa under Salt Stress [J]. Crops, 2025, 41(1): 194-201. |
| [12] | Li Feng, Gao Hongyun, Zhang Chong, Zhang Baoying, Ma Jianfu, Guo Na, Bai Wei, Fang Aiguo, Yang Zhimin, Li Yuan. Effects of Salt Stress on Growth and Physiological Indexes of Oat [J]. Crops, 2024, 40(6): 140-146. |
| [13] | E Lifeng, Xu Jinchong, Chen Xiubin, Quan Jianhua, Hua Jun, Yin Lijuan, Wang Shunqi, Zhao Wenqin. Effects of Exogenous Silicon on Seed Germination and Physiological Characteristics of Brassica pekinensis under Salt Stress [J]. Crops, 2024, 40(6): 212-217. |
| [14] | Hou Yuchen, Pang Chunhua, Zhang Yongqing, Kang Shuyu, Wu Yueyue, Yan Jingrong, Wang Jiaqi. Effects of Biochar and Nitrogen Fertilizer on the Physiological Growth Characteristics of Quinoa Seedlings under Saline Alkali Stress [J]. Crops, 2024, 40(4): 240-246. |
| [15] | Gu Huaiying, Hu Shiqin, Zhao Qing, Liu Changhua, Meng Lijun. The Progress on Enhancing Salt Tolerance of Rice by Rhizosphere Microorganisms [J]. Crops, 2024, 40(4): 8-13. |
|
||