作物杂志,2023, 第4期: 253–259 doi: 10.16035/j.issn.1001-7283.2023.04.036
盐、碱胁迫下水稻种子萌发过程水分含量变化及对种子发芽影响的低场核磁检测研究
- 1沈阳农业大学信息与电气工程学院,110866,辽宁沈阳
2辽宁省农业信息化工程技术研究中心,110866,辽宁沈阳
3辽宁省水稻研究所,110161,辽宁沈阳
Research on the Moisture Content Variation and Influence to Rice Seed Germination under Salt and Alkali Stress by Low Field NMR
Yang Hongwei1,2( ), Zhang Liying3, Li Xiaohui1,2(
), Zhang Liying3, Li Xiaohui1,2( )
)
- 1College of Information and Electrical Engineering, Shenyang Agricultural University, Shenyang 110866, Liaoning, China
2Liaoning Engineering Research Center for Information Technology in Agriculture, Shenyang 110866, Liaoning, China
3Liaoning Rice Research Institute, Shenyang 110161, Liaoning, China
摘要:
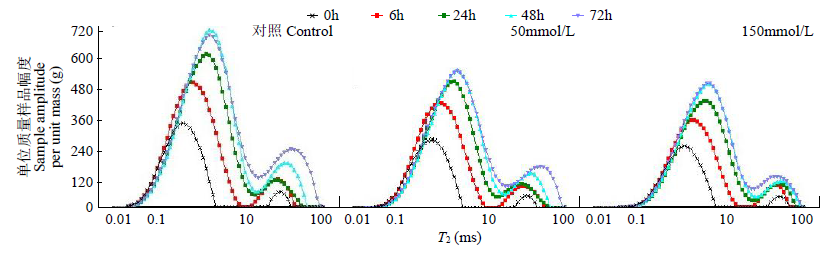
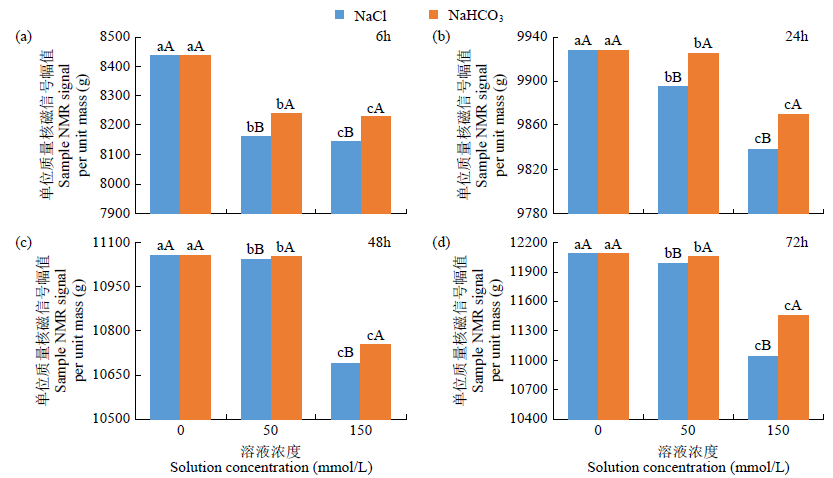
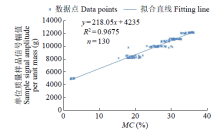
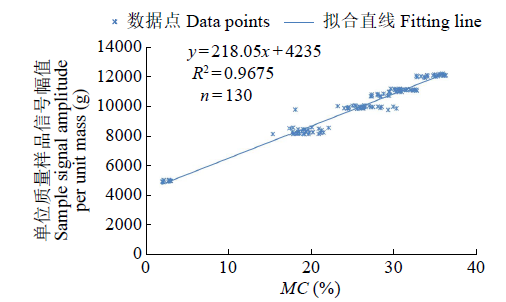
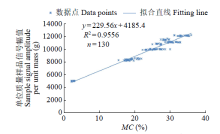
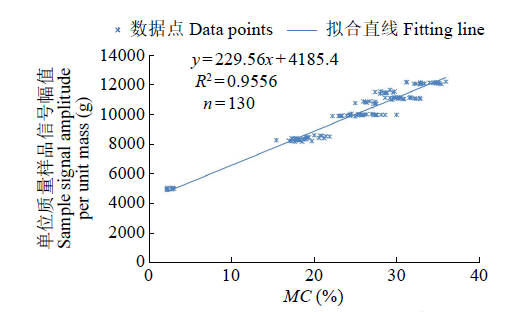
为研究盐、碱胁迫下水稻种子萌发过程水分含量变化及对种子发芽的影响,以及利用低场核磁检测技术测定种子含水量的可行性,利用光照培养箱进行培养皿种子发芽试验,分析了在蒸馏水(对照处理)、浓度为50、150mmol/L NaCl和NaHCO3胁迫处理下,盐粳48和辽星1种子萌发72h过程中核磁信号幅值变化及各处理对发芽指标的影响,确定单位质量水稻种子样品核磁信号幅值与湿基含水率的回归函数关系。结果表明,与对照处理相比,萌发6、24、48、72h后,2个品种水稻种子核磁信号幅值平均分别增长了65%、95%、115%和135%以上。萌发相同时间后,相比对照处理,NaCl和NaHCO3胁迫处理下核磁信号幅值平均降低了5%以上,发芽指数平均降低了4.5%以上,说明水稻种子萌发过程水分含量与种子发芽指数呈正相关。不同浓度NaCl和NaHCO3胁迫下,单位质量水稻种子核磁信号幅值和湿基含水率之间均具有一致的线性关系,R2均大于0.95。说明利用核磁共振技术测定盐、碱胁迫下水稻种子的水分含量是合理可靠的。
| [1] | 王海莲, 张华文, 刘宾, 等. 低度盐胁迫下高粱苗期相关性状的QTL定位. 分子植物育种, 2017, 15(2):604-610. |
| [2] |
Han L, Liu H, Yu S, et al. Potential application of oat for phytoremediation of salt ions in coastal saline-alkali soil. Ecological Engineering, 2013, 61:274-281.
doi: 10.1016/j.ecoleng.2013.09.034 |
| [3] |
Alvarez-Acosta C, Marrero-Dominguez A, Gallo-Llobet L, et al. Physiological response of selected avocados (Persea americana) subjected to NaCl and NaHCO3 stress. Scientia Horticulturae, 2018, 237:81-88.
doi: 10.1016/j.scienta.2018.04.010 |
| [4] |
Yan W, Gang Q J, Yin N H, et al. Effects of salt, alkali and salt- alkali mixed stresses on seed germination of the halophyte Salsola ferganica (Chenopodiaceae). Acta Ecologica Sinica, 2013, 33(6):354-360.
doi: 10.1016/j.chnaes.2013.09.010 |
| [5] |
Fan X D, Wang J Q, Yang N, et al. Gene expression profiling of soybean leaves and roots under salt, saline-alkali and drought stress by high-throughput Illumina sequencing. Gene, 2013, 512(2):392-402.
doi: 10.1016/j.gene.2012.09.100 |
| [6] |
Lin J, Yu D, Shi Y, et al. Salt-alkali tolerance during germination and establishment of Leymus chinensis in the Songnen Grassland of China. Ecological Engineering, 2016, 95:763-769.
doi: 10.1016/j.ecoleng.2016.07.011 |
| [7] | Wang F, Xiao H L, Peng X M, et al. Effects of salt and alkali stress on Reaumuria soongorica germination. Sciences in Cold and Arid Regions, 2017, 9(2):158-166. |
| [8] |
Guo R, Zhou J, Ren G X, et al. Physiological responses of linseed seedlings to iso osmotic polyethylene glycol, salt, and alkali stresses. Agronomy Journal, 2013, 105(3):764-772.
doi: 10.2134/agronj2012.0442 |
| [9] | 李双男, 郭慧娟, 王晶, 等. 不同盐碱胁迫对棉花种子萌发的影响. 种子, 2018, 37(1):38-45. |
| [10] | 刘建新, 王金成, 王瑞娟, 等. 盐、碱胁迫对燕麦幼苗光合作用的影响. 干旱地区农业研究, 2015, 33(6):155-160. |
| [11] |
Gong B, Wen D, Bloszies S, et al. Comparative effects of NaCl and NaHCO3 stresses on respiratory metabolism, antioxidant system, nutritional status,and organic acid metabolism in tomato roots. Acta Physiologiae Plantarum, 2014, 36(8):2167-2181.
doi: 10.1007/s11738-014-1593-x |
| [12] |
Gong B, Dan W, Vandenlangenberg K, et al. Comparative effects of NaCl and NaHCO3 stress on photosynthetic parameters, nutrient metabolism, and the antioxidant system in tomato leaves. Scientia Horticulturae, 2013, 157(3):1-12.
doi: 10.1016/j.scienta.2013.03.032 |
| [13] | 季平, 张鹏, 徐克章, 等. 不同类型盐碱胁迫对大豆植株生长性状和产量的影响. 大豆科学, 2013, 32(4):477-481. |
| [14] |
Lin J, Shao S, Wang Y, et al. Germination responses of the halophyte Chloris virgata to temperature and reduced water potential caused by salinity, alkalinity and drought stress. Grass and Forage Science, 2016, 71(3):507-514.
doi: 10.1111/gfs.2016.71.issue-3 |
| [15] | 张彦妮, 李博, 何淼. 盐胁迫对大花飞燕草种子萌发的影响. 草业科学, 2012, 29(8):1235-1239. |
| [16] |
Zhang H, Zhang G, Lü X, et al. Salt tolerance during seed germination and early seedling stages of 12 halophytes. Plant and Soil, 2015, 388(1/2):229-241.
doi: 10.1007/s11104-014-2322-3 |
| [17] |
He L, Lin B, Hong Y, et al. Assessing the moisture migration during microwave drying of coal using low-field nuclear magnetic resonance. Drying Technology, 2017, 36(5):1-11.
doi: 10.1080/07373937.2017.1326130 |
| [18] | 要世瑾, 杜光源, 牟红梅, 等. 基于核磁共振技术检测小麦植株水分分布和变化规律. 农业工程学报, 2014, 30(24):177- 186. |
| [19] | 牟红梅, 何建强, 邢建军, 等. 小麦灌浆过程籽粒水分变化的核磁共振检测. 农业工程学报, 2016, 32(8):98-104. |
| [20] |
Qiu Q Z, Wei L, Hao K L, et al. Low-field nuclear magnetic resonance for online determination of water content during sausage fermentation. Journal of Food Engineering, 2017, 212:291-297.
doi: 10.1016/j.jfoodeng.2017.05.021 |
| [21] |
Sandnes R, Simon S, Sjöblom J, et al. Optimization and validation of low field nuclear magnetic resonance sequences to determine low water contents and water profiles in W/O emulsions. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2014, 441(3):441-448.
doi: 10.1016/j.colsurfa.2013.09.030 |
| [1] | 张业猛, 卢柯欣, 毛文钰, 张晓燕, 李文静, 杨修. 藜麦LEA基因家族鉴定及盐胁迫下的表达分析[J]. 作物杂志, 2025, (6): 100–111 |
| [2] | 郭龙玉, 高欣梅, 福英, 张明伟, 王莹, 李乌日吉木斯, 乌日力格, 王英杰, 王靖宇, 杨凤婷, 谷艳茹, 全宇. 免疫诱抗剂ZNC对盐碱胁迫下燕麦幼苗生长发育及生理适应性的影响[J]. 作物杂志, 2025, (6): 203–209 |
| [3] | 陈奕, 陈潇, 张茂星, 李静, 常静静, 李嘉炜, 林海晴, 陈兴平, 邓晓亮, 谢大森, 郭少龙, 沈仲灯, 张白鸽. 不同冬瓜品种对低盐胁迫的响应及其耐盐性综合评价[J]. 作物杂志, 2025, (6): 58–66 |
| [4] | 滕文, 叶凡, 周舟, 王屿乐, 刘立军. 小麦和油菜秸秆还田处理对盐胁迫下水稻产量和品质的影响[J]. 作物杂志, 2025, (5): 11–18 |
| [5] | 徐明丽, 吴柏辰, 刘畅, 高鑫涵, 殷佳琪, 何雪, 刘莹, 尹泽群, 苗兴芬. 褪黑素浸种对盐碱胁迫下谷子萌发的影响[J]. 作物杂志, 2025, (5): 42–46 |
| [6] | 闫晶蓉, 庞春花, 张永清, 毋悦悦, 侯钰晨, 王嘉祺, 乔曼. 脱硫石膏与腐植酸配施对盐碱地土壤及藜麦生长的影响[J]. 作物杂志, 2025, (5): 47–53 |
| [7] | 张金东, 王成, 卢环, 曾玲玲, 张巩亮, 孙浩月, 刘悦, 杨贺麟, 侯晓敏. 盐碱胁迫下不同基因型绿豆对外源BR的响应[J]. 作物杂志, 2025, (5): 54–60 |
| [8] | 陈萍, 罗园园, 王娟, 孙权, 马玲芳, 马文礼, 谢静波. 燕麦萌发期对混合盐碱胁迫的响应及耐受性评价[J]. 作物杂志, 2025, (5): 61–66 |
| [9] | 王兖薇, 武俊喜, 汪艳, 牟涛, 朗卓玛, 苗彦军. 盐碱胁迫对异株荨麻种子萌发的影响[J]. 作物杂志, 2025, (5): 67–73 |
| [10] | 赵洲, 张丽, 高新磊, 邱鸿雨. 复合盐碱胁迫对燕麦生长及代谢的影响[J]. 作物杂志, 2025, (5): 74–85 |
| [11] | 景茂雅, 张子玉, 张萌, 合佳敏, 严翻翻, 高艳梅, 张永清. 水杨酸浸种对盐胁迫藜麦种子萌发及幼苗生长的影响[J]. 作物杂志, 2025, (1): 194–201 |
| [12] | 李峰, 高宏云, 张翀, 张宝英, 马建富, 郭娜, 白苇, 方爱国, 杨志敏, 李源. 盐胁迫对燕麦生长及生理指标的影响[J]. 作物杂志, 2024, (6): 140–146 |
| [13] | 鄂利锋, 徐金崇, 陈修斌, 权建华, 华军, 尹丽娟, 王舜奇, 赵文勤. 外源硅对盐胁迫下娃娃菜种子萌发及幼苗生理特性的影响[J]. 作物杂志, 2024, (6): 212–217 |
| [14] | 侯钰晨, 庞春花, 张永清, 康书瑜, 毋悦悦, 闫晶蓉, 王嘉祺. 施用生物炭与氮肥对盐碱胁迫下藜麦幼苗生理生长特性的影响[J]. 作物杂志, 2024, (4): 240–246 |
| [15] | 顾怀应, 胡诗钦, 赵晴, 刘长华, 孟丽君. 根际微生物增强水稻耐盐性研究进展[J]. 作物杂志, 2024, (4): 8–13 |
|
||