Crops ›› 2016, Vol. 32 ›› Issue (2): 43-49.doi: 10.16035/j.issn.1001-7283.2016.02.008
Previous Articles Next Articles
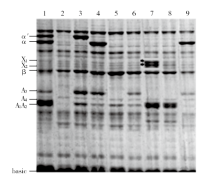
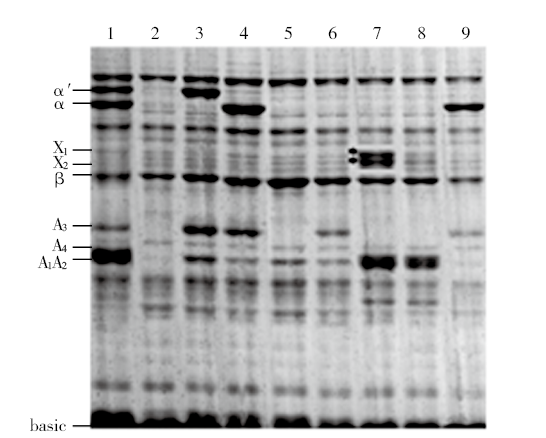
Development of a Set of 7S and 11S Multi-Subunit-Deficient Mutants with Chinese Soybean Genetic Background
Guo Bowen,Zhao Xue,Wei Xiaoshuang,Han Yanjing,Liang Qiangfei,Song Bo,Liu Shanshan
- Key Laboratory of Soybean Biology in Chinese Ministry of Education,Northeast Agricultural University,Harbin 150030,Heilongjiang,China
| [1] | Koshiyama I . Chemical and physical properties of a 7S protein in soybean globulins. Cereal Chemistry, 1968,45:394-404. |
| [2] |
Utsumi S, Kinsella J E . Forces involved in soy protein gelation: effects of various reagents on the formation,hardness and solubility of heat-induced gels made from 7S,11S and soy isolate. Journal of Food Science, 1985,50:1278-1282.
doi: 10.1111/jfds.1985.50.issue-5 |
| [3] | Salleh M R B, Maruyama N, Takahashi K , et al. Gelling properties of soybean beta-conglycinin having different subunits compositions.Bioscience,Biotechnology, and Biochemistry, 2004,68:1091-1096. |
| [4] | Aoyama T, Kohno M, Saito T , et al. Reduction by phytate-reduced soybean beta-conglycinin of plasma triglyceride level of young and adult rat.Bioscience,Biotechnology, and Biochemistry, 2001,65:1071-1075. |
| [5] |
Manzoni C, Lovati M R, Ggianazza E , et al. Soybean protein products as regulators of liver low-density lipoproteinreceptors Ⅱ.a α′ rich commercial soy concentrate and a deficient mutant differently affect low-density lipoproteinreceptor activation. Journal of Agricultural and Food Chemistry, 1998,46:2481-2484.
doi: 10.1021/jf980100c |
| [6] | Sirtori C R, Lovati M R, Manzoni C , et al. Soy and cholesterol reduction clinical experience. Journal of Nutrition, 1995,125:598-605. |
| [7] |
Schuler M A, Schmitt E S, Beachy R N . Closely related families of genes code for the α and α′ subunits of the soybean 7S storage protein complex. Nucleic Acids Research, 1982,10:8225-8243.
doi: 10.1093/nar/10.24.8225 |
| [8] |
Sebastiani F L, Farrell L B, Schuler M A , et al. Complete sequence of a cDNA of α subunit of soybean β-conglycinin. Plant Molecular Biology, 1990,15:197-201.
doi: 10.1007/BF00017745 |
| [9] |
Tierney M L, Bray E A, Allen R D , et al. Isolation and characterization of a genomic clone encoding the β-subunit of β-conglycinin. Planta, 1987,172:356-363.
doi: 10.1007/BF00398664 |
| [10] |
Than V H, Shibasaki K . Beta-conglycinin from soybean proteins.Isolation and immunological and physicochemical properties of the monomeric forms. Biochimica et Biophysica Acta, 1977,490:370-384.
doi: 10.1016/0005-2795(77)90012-5 |
| [11] | Staswick P E, Hermodson M A, Nielsen N C . Identification of the acidic and basic subunit complexes of glycinin. Biological Chemistry, 1981,256:8752-8755. |
| [12] | Kaviani B, Kharabian A . Improvement of the nutritional value of soybean [Glycine max (L.) Merr.]seed with alteration in protein subunit of glycinin (11S globulin) and beta-conglycinin (7S globulin). Turkish Journal of Biology, 2008,32:91-97. |
| [13] |
Tsukada Y, Kitamura K, Harada K , et al. Genetic analysis of subunits of two major storage protein (β-conglycinin and glycinin) in soybean seeds. Japanse Journal of Breed, 1986,36:390-400.
doi: 10.1270/jsbbs1951.36.390 |
| [14] | Vaintraub I A, Shutov A D . Moleeular weight of subunits of soybean 11S proteins. Biokhimiia, 1971,36(5):1086-1088. |
| [15] |
Adachi M, Takenaka Y, Gidamis A B . Crystal structure of soybean proglycinin AlaBlb homotrimer. Journal of Molecular Biology, 2001,305:291-305.
doi: 10.1006/jmbi.2000.4310 |
| [16] |
Adachi M, Kanalnori J, Masuda T . Crystal structure of soybean 11S globulin:glycinin A3B4 homohexamer. Proceedings of the National Academy of Science of the United States of America, 2003,100:7395-7400.
doi: 10.1073/pnas.0832158100 |
| [17] | Staswick P E, Hermodson M A, Nilsen N C . Identification of the cystines which link the acidic and basic components of the glycinin subunits. Biological Chemistry, 1984,259:13431-13435. |
| [18] | Ygasaki K, Kaizuma N, Kltamura K . Inheritance of glyeinin subunits and characterization of glyinin molecules lacking the subunits in soybean [Glyeine max(L.)Merr.]. Breeding Science, 1996,46:11-15. |
| [19] |
Nielsen N C, Diekinson C D, Cho T J . Characterization of the glycinin gene family in soybean. Plant Cell, 1989,1:313-328.
doi: 10.1105/tpc.1.3.313 |
| [20] |
Fukushina D . Recent progress of soybean protein foods. Food Reviews International, 1991,7:323-335.
doi: 10.1080/87559129109540915 |
| [21] |
Yamaauchi F . Molecular understanding of soybean protein. Food Reviews International, 1991,7:283-332.
doi: 10.1080/87559129109540914 |
| [22] |
Takahashi M, Uematsu Y, Kashiwaba K , et al. Accumulations of high levels of free amino acids in soybean seeds through integration of mutations conferring seed protein deficiency. Planta, 2003,217:577-586.
doi: 10.1007/s00425-003-1026-3 |
| [23] | Jennings A C, Morton R K . Amino acids and protein synthesis in developing wheat endosperm. Australia Journal of Boil Science, 1963,16:384-394. |
| [24] |
Mertz E T, Bates L S, Nelson O E . Mutant gene that changes protein composition and increased lysine content of maize endosperm. Scinence, 1964,145:279-280.
doi: 10.1126/science.145.3629.279 |
| [25] | Misra P S, Mertz E T, Glover D V . Studies on corn proteins:Ⅷ.Free amino acid content of opaque-2 and double mutants. Cereal Chemistry, 1975,52:844-848. |
| [26] | Mauri I, Maddaloni M, Lohmer S , et al. Functional expression of the transcriptional activator Opaque-2 of Zea mays in transformed yease. Molecular and General Genetics, 1993,241:319-326. |
| [27] | Or E, Boyer S K, Larkins B A . Opaque2 modifies act post-transcriptionally and in a polar manner on gamma-zein gene expression in maize endosperm. Plant cell, 1993,5:1599-1609. |
| [28] |
Gillikin J W, Zhang F, Coleman C E , et al. A defective sign peptide tethers the floury-2 zein to the endoplasmic reticulum membrane. Plant Physiology, 1997,114, 345-352.
doi: 10.1104/pp.114.1.345 |
| [29] |
Coleman C E, Lopes M A, Gillikin J W , et al. A defective signal peptide in the maize high-lysine mutant floury-2. Proceedings of National Academy Sciences USA, 1995,92:6828-6831.
doi: 10.1073/pnas.92.15.6828 |
| [30] | Mertz E T. Thirty years of opaque2 maize. In BA Larkins,ET Mertz,eds,Quality protein Maize 1964-1994.Purdue University Press,West Lafayette, 1997: 25-37. |
| [31] |
Kim C S, Gibbon B C, Gillikin J W , et al. The maize mucronate mutation is a deletion in the 16-kDa gamma-zein gene that induces the unfolded protein response. Plant Journal, 2006,48:440-451.
doi: 10.1111/tpj.2006.48.issue-3 |
| [32] | Geevers H O, Lake J K . Development of modified opaque2 maize in South Africa. American Association of Cereal Chemists, 1992: 49-78. |
| [33] | Glover D V . Corn protein-genetics,breeding,and value in foods and feeds. American Association of Cereal Chemists, 1992: 49-78. |
| [34] | Harada K, Hayashi M, Tsubokura Y . Genetic variation of globulin composition in soybean seeds. Agriculture Research Updates, 2013,5:101-116. |
| [35] |
Cantón F R, Suáez M F, Cánovas F M . Molecular aspects of nitrogen mobilization and recycling in trees. Photosynthesis Research, 2005,83:265-278.
doi: 10.1007/s11120-004-9366-9 |
| [36] |
Cheng L, Ma F, Ranwala D . Nitrogen storage and its interaction with carbohydrates of young apple trees in response to nitrogen supply. Tree Physiology, 2004,24:91-98.
doi: 10.1093/treephys/24.1.91 |
| [1] | Yun Zhao,Cailong Xu,Xu Yang,Suzhen Li,Jing Zhou,Jicun Li,Tianfu Han,Cunxiang Wu. Effects of Sowing Methods on Seedling Stand and Production Profit of Summer Soybean under Wheat-Soybean System [J]. Crops, 2018, 34(4): 114-120. |
| [2] | Mingjun Zhang,Zhongfeng Li,Lili Yu,Jun Wang,Lijuan Qiu. Identification and Screening of Protein Subunit Variation Germplasm from Both Mutants and Natural Population in Soybean [J]. Crops, 2018, 34(3): 44-50. |
| [3] | Jiani Zhu,Huiping Dai,Shuhe Wei,Genliang Jia,Dejing Chen,Jinjin Pei,Qing Zhang,Long Qiang. Effects of Applying Zn on the Growth and Zn Accumulation in Soybean at Flowering Stage [J]. Crops, 2018, 34(1): 152-155. |
| [4] | Tianle Ma,Jianxin Zhang. Effects of Different Multiple Cropping Methods on Dry Matter Accumulation, Distribution and Yield of Summer Soybean [J]. Crops, 2018, 34(1): 156-159. |
| [5] | Lina Li,Longguo Jin,Chuanxiao Xie,Changlin Liu. Determining Blind Samples of Transgenic Maize and Transgenic Soybean [J]. Crops, 2017, 33(6): 37-44. |
| [6] | Xuechao Zhou,Surong Ding,Yunshan Wei,Yanfang Zhou,Xue Wei,Rina Na,Feng Li. Evaluation of Adaptability of Different Vegetable Soybean Cultivars (Lines) in Chifeng Area [J]. Crops, 2017, 33(3): 44-48. |
| [7] | Guoyong Ren,Wei Li,Lifeng Zhang,Caijie Wang,Haiying Dai,Jinlong Wang,Ran Xu,Yanwei Zhang. The Resistance to Soybean Cyst Nematode Race 1 of Transgenic Soybean with hrf2 Encoding HarpinXooc [J]. Crops, 2017, 33(3): 49-53. |
| [8] | Xuli Zhang,Baolong Xing,Guimei Wang,Lili Yin. Effects of Planting Density on Agronomic Traits, Economic Traits and Yield of Soybean in North of Shanxi Province [J]. Crops, 2017, 33(3): 127-131. |
| [9] | Xiting Zhang,Liwei Cao,Shucai Lü,Guoxing Chen,Yongji Wang,Shuhan Yu,Zhenping Gong. Effects of Bulk Density on Nitrogen Absorption and Yield of Soybean on Black Soil [J]. Crops, 2017, 33(3): 132-137. |
| [10] | Yixin Tian,Fengju Gao. The Response of Growth and Dry Matter Accumulation and Distribution of High Protein Soybean to Plant Density [J]. Crops, 2017, 33(2): 121-125. |
| [11] | Li Yan,Qiang Yang,Yupeng Shao,Dandan Li,Zhikun Wang,Wenbin Li. Cloning and Sequence Analysis of GmWRI1a Gene Promoter in Soybean [J]. Crops, 2017, 33(2): 51-58. |
| [12] | Yu Gao,Yanchao Liu,Shusen Shi,Juan Cui,Jinfeng Xiong. Review on Soybean Thrips in China [J]. Crops, 2017, 33(1): 8-13. |
| [13] | Haiyan Li,Deli Cai,Jingsheng Chen,Yuxi Duan,Lijie Chen,Yingyu Shang. The Influence of Resistant and Susceptible Soybean Germplasm on Growth Dynamic Changes of Soybean Cyst Nematode Race 3(SCN3) [J]. Crops, 2017, 33(1): 144-149. |
| [14] | Qianxu Zhao,Xianrong Yue,Yunsheng Xia,Naiming Zhang,Fuzhao Nian,Yunqiang Yang,Yulin Ma. Effects of Arbuscular Mycorrhizal Fungus Inoculation on Growth and Nitrogen Utilization of Intercropped Maize and Soybean in Purple Soil under Facilitated Condition [J]. Crops, 2016, 32(5): 94-100. |
| [15] | Zhao Geng,Youbin Kong,Lili Zhao,Cui Liu,Hui Du,Xihuan Li,Caiying Zhang. Transformation of GmPHR1 and GmPAP4 Related to High Phosphorus Efficiency and Elite Germplasm Enhancement in Soybean (Glycine max) [J]. Crops, 2016, 32(3): 58-62. |
|